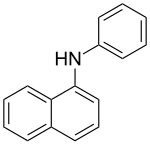
N- phenyl-1-naphthylamine
| Structural formula | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|||||||||||||||||||
| General | |||||||||||||||||||
| Surname | N- phenyl-1-naphthylamine | ||||||||||||||||||
| other names |
|
||||||||||||||||||
| Molecular formula | C 16 H 13 N | ||||||||||||||||||
| Brief description |
red-violet solid with a slightly aromatic odor |
||||||||||||||||||
| External identifiers / databases | |||||||||||||||||||
|
|||||||||||||||||||
| properties | |||||||||||||||||||
| Molar mass | 219.29 g mol −1 | ||||||||||||||||||
| Physical state |
firmly |
||||||||||||||||||
| density |
1.16 g cm −3 |
||||||||||||||||||
| Melting point |
56-60 ° C |
||||||||||||||||||
| boiling point |
224 ° C |
||||||||||||||||||
| Vapor pressure |
0.07 hPa (123 ° C) |
||||||||||||||||||
| solubility |
|
||||||||||||||||||
| safety instructions | |||||||||||||||||||
|
|||||||||||||||||||
| Toxicological data | |||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||||||||
N -Phenyl-1-naphthylamine is a chemical compound from the aminobenzenes group .
Extraction and presentation
N- phenyl-1-naphthylamine can be obtained by catalytic conversion of 1-naphthylamine with aniline at 200 ° C.
properties
As a technical product, N- phenyl-1-naphthylamine is a flammable, hardly inflammable, flaky, red-violet solid with a slightly aromatic odor, which is practically insoluble in water. The pure compound is in the form of lemon-yellow needles or prisms. Small amounts (<1%) of impurities in the technical product are 1-naphthylamine , 2-naphthylamine , aniline , 1-naphthol , 1,1-dinaphthylamine and N -phenyl -2-naphthylamine .
use
N- phenyl-1-naphthylamine is used as an antioxidant in lubricating, gear, hydraulic and bearing oils as well as in rubber compounds. It is also a precursor to the synthesis of the triarylmethane dye Victoria blue and other chemical compounds.
Risk assessment
N- phenyl-1-naphthylamine was included in the EU's ongoing action plan ( CoRAP ) in 2012 in accordance with Regulation (EC) No. 1907/2006 (REACH) as part of substance evaluation . The effects of the substance on human health and the environment are re-evaluated and, if necessary, follow-up measures are initiated. The reasons for the uptake of N- phenyl-1-naphthylamine were concerns about its widespread use and the dangers arising from a possible assignment to the group of PBT / vPvB substances. The re-evaluation has been running since 2012 and is carried out by Germany .
Individual evidence
- ↑ a b c d e f g h i j Entry on N-phenyl-1-naphthylamine in the GESTIS substance database of the IFA , accessed on September 10, 2017(JavaScript required) .
- ↑ a b c d Toxicological assessment of N-phenyl-1-naphthylamine (PDF) at the professional association raw materials and chemical industry (BG RCI), accessed on May 1, 2018.
- ↑ Patent EP0924191 : Production of N-phenyl-1-naphthylamine. Published June 23, 1999 .
- ^ Concise International Chemical Assessment Document (CICAD) for N-Phenyl-1-naphthylamine , accessed September 11, 2017.
- ↑ Entry on N-Phenylnaphthalenamine. In: Römpp Online . Georg Thieme Verlag, accessed on September 11, 2017.
- ↑ Community rolling action plan ( CoRAP ) of the European Chemicals Agency (ECHA): N-1-naphthylaniline , accessed on March 26, 2019.


