Sodium hydrogen difluoride
| Structural formula | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
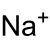 
|
|||||||||||||||||||
| General | |||||||||||||||||||
| Surname | Sodium hydrogen difluoride | ||||||||||||||||||
| other names |
|
||||||||||||||||||
| Molecular formula | NaHF 2 | ||||||||||||||||||
| Brief description |
white solid |
||||||||||||||||||
| External identifiers / databases | |||||||||||||||||||
|
|||||||||||||||||||
| properties | |||||||||||||||||||
| Molar mass | 61.99 g mol −1 | ||||||||||||||||||
| Physical state |
firmly |
||||||||||||||||||
| density |
2.08 g cm −3 |
||||||||||||||||||
| Melting point |
Decomposition from 160 ° C |
||||||||||||||||||
| solubility |
little in water (32.5 g l −1 at 20 ° C) |
||||||||||||||||||
| safety instructions | |||||||||||||||||||
|
|||||||||||||||||||
| MAK |
1 mg m −3 |
||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||||||||
Sodium hydrogen difluoride is a white, hygroscopic sodium salt of hydrofluoric acid , i.e. a chemical compound from the group of fluorides . It can be viewed as an addition compound of sodium fluoride and hydrogen fluoride (NaF · HF).
Appearance and properties
Sodium hydrogen difluoride crystallizes from solutions of sodium fluoride in the presence of excess hydrofluoric acid. The salt forms white, hygroscopic crystals that dissolve moderately in water (32.5 g · l −1 ). From 160 ° C the salt decomposes to sodium fluoride and hydrogen fluoride.
Presentation:
Decomposition by heating:
use
The substance is used as a preservative , etchant for glasses , enamel and ceramic products. On a large industrial scale, the salt is used as a flux in aluminum production, as a fungicide , insecticide , for the impregnation of timber and as a medicinal substance in the therapy of osteoporosis .
safety instructions
On the one hand, sodium hydrogen difluoride has a caustic and tissue-destructive effect by splitting off hydrogen fluoride. On the other hand, like all soluble fluorides, it is a potent poison. In the body, it binds calcium ions, which precipitate as water-insoluble calcium fluoride and are therefore no longer available for the metabolism. Symptoms of pulmonary edema , shock, tetany and cardiac arrhythmias appear after ingestion of larger amounts .
Individual evidence
- ↑ a b c d e f g h Entry on sodium hydrogen difluoride in the GESTIS substance database of the IFA , accessed on January 10, 2017(JavaScript required) .
- ↑ Entry on Sodium hydrogendifluoride in the Classification and Labeling Inventory of the European Chemicals Agency (ECHA), accessed on February 1, 2016. Manufacturers or distributors can expand the harmonized classification and labeling .



