Pyraclostrobin
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
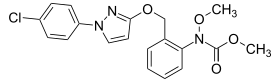
|
||||||||||||||||
| General | ||||||||||||||||
| Surname | Pyraclostrobin | |||||||||||||||
| other names |
|
|||||||||||||||
| Molecular formula | C 19 H 18 ClN 3 O 4 | |||||||||||||||
| Brief description |
yellow solid with a slightly aromatic odor |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 387.82 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| density |
1.29 g cm −3 |
|||||||||||||||
| Melting point |
59-62 ° C |
|||||||||||||||
| boiling point |
200 ° C (decomposition) |
|||||||||||||||
| Vapor pressure |
2.6 10 −10 hPa (20 ° C) |
|||||||||||||||
| solubility |
|
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| Toxicological data |
> 5,000 mg kg −1 ( LD 50 , rat , transdermal ) |
|||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Pyraclostrobin is a chemical compound from the group of strobilurins that is used in agriculture.
Extraction and presentation
Pyraclostrobin is chemically produced by reacting 4-chlorophenyl hydrazine with ethyl acrylate and oxygen to form pyrazole, alkylation with 2-nitrobenzyl chloride , catalytic hydrogenation to form hydroxylamine and subsequent reaction with methyl chloroformate and dimethyl sulfate .
properties
As a pure substance, pyraclostrobin is an odorless, colorless solid. The technical product is a flammable yellow solid with a weak aromatic odor, which is practically insoluble in water. In an aqueous solution in the dark it is stable at pH 4, 5 and 7 (25 ° C and 50 ° C). At a pH of 9, a very slow degradation is observed at normal ambient temperature.
use
Pyraclostrobin is used as an active ingredient in crop protection products. It is used as a fungicide and works by inhibiting mitochondrial respiration. This leads to a reduction in the amount of ATP available in the fungal cell. It has been marketed by BASF since 2002 .
Admission
The use of pyraclostrobin as a fungicide in plant protection products has been permitted in the European Union since 2004, and since 2009 its use as a growth regulator has also been permitted. In Germany, Austria and Switzerland, plant protection products with this active ingredient are approved.
Web links
- Joint Meeting on Pesticide Residues (JMPR), Monograph for PYRACLOSTROBIN
Individual evidence
- ↑ a b c d e f g h i Entry on pyraclostrobin in the GESTIS substance database of the IFA , accessed on January 8, 2020(JavaScript required) .
- ↑ a b c d e f FAO: PYRACLOSTROBIN (210) - First draft prepared by Bernard Declercq, Epinay sur Orge, France (PDF; 1.2 MB)
- ↑ Pyraclostrobin data sheet , PESTANAL at Sigma-Aldrich , accessed on May 19, 2017 ( PDF ).
- ↑ a b Wolfgang Krämer, Ulrich Schirmer: Modern Crop Protection Compounds . John Wiley & Sons, 2012, ISBN 978-3-527-32965-6 , pp. 618 (English, limited preview in Google Book search).
- ↑ Directive 2009/25 / EC of the Commission of April 2, 2009 amending Directive 91/414 / EEC of the Council with regard to an extension of the uses of the active ingredient pyraclostrobin
- ↑ General Directorate Health and Food Safety of the European Commission: Entry on pyraclostrobin in the EU pesticide database; Entry in the national registers of plant protection products in Switzerland , Austria and Germany ; accessed on March 13, 2016.



