Terbium (III) oxide
| Crystal structure | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||
| __ Tb 3+ __ O 2− | ||||||||||||||||
| General | ||||||||||||||||
| Surname | Terbium (III) oxide | |||||||||||||||
| other names |
|
|||||||||||||||
| Ratio formula | Tb 2 O 3 | |||||||||||||||
| Brief description |
white solid |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 365.85 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| density |
7.91 g cm −3 |
|||||||||||||||
| Melting point |
2303 ° C |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Terbium (III) oxide is a chemical compound , more precisely the oxide of terbium .
Occurrence
Terbium (III) oxide does not occur, of course. However, it occurs bound as a highly complex silicate z. B. in gadolinite .
Extraction and presentation
Terbium (III) oxide cannot be obtained directly from terbium and oxygen , since brown terbium (III, IV) oxide Tb 4 O 7 is formed. Instead, it is produced from this through controlled reduction with hydrogen .
properties
Physical Properties
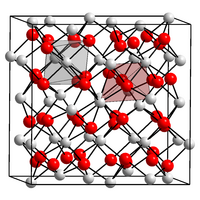
As with other heavy lanthanoid oxides, two crystal structures are known of terbium (III) oxide . The normally stable structure is a cubic defective fluorite structure ( bixbyite structure) with the lattice parameter a = 1072 pm . This also occurs with other lanthanide oxides. A monoclinic structure of terbium (III) oxide is also known. This is created when crystallizing from terbium oxide / magnesium oxide melts.
Terbium (III) oxide is pure white to cream-colored, depending on its purity. Luminesce glasses with additions of terbium (III) oxide.
use
Pure terbium (III) oxide is hardly used or not used at all. Terbium (III, IV) oxide Tb 4 O 7 is used as the starting material for the production of pure terbium .
See also
- Terbium (III, IV) oxide Tb 4 O 7 (black-brown powder, which serves as an activator for various fluorescent phosphors)
- Terbium (IV) oxide TbO 2
Individual evidence
- ↑ a b c David R. Lide (Ed.): CRC Handbook of Chemistry and Physics . 90th edition. (Internet version: 2010), CRC Press / Taylor and Francis, Boca Raton, FL, Properties of the Elements and Inorganic Compounds, pp. 4-94.
- ↑ a b Entry on diterbium trioxide in the GESTIS substance database of the IFA , accessed on February 10, 2017(JavaScript required) .
- ^ NN Greenwood, A. Earnshaw: Chemistry of the elements , 1st edition, VCH Verlagsgesellschaft, Weinheim 1988, ISBN 3-527-26169-9 .
- ↑ NC Baenzinger, HA Eick, HS Schuldt, L. Eyring: Terbium Oxides. III. X-Ray Diffraction Studies of Several Stable Phases . In: Journal of the American Chemical Society , 1961, 83, 10, pp. 2219-2223.
- ↑ E. Hubbert-Paletta, Hk. Müller-Buschbaum: X-ray investigation on single crystals of monoclinic Tb 2 O 3 . In: Journal for inorganic and general chemistry , 1968, 363, pp. 145-150, doi : 10.1002 / zaac.19683630306 .

