1,1-dichloro-1-fluoroethane
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
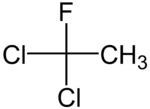
|
||||||||||||||||
| General | ||||||||||||||||
| Surname | 1,1-dichloro-1-fluoroethane | |||||||||||||||
| other names |
|
|||||||||||||||
| Molecular formula | C 2 H 3 Cl 2 F | |||||||||||||||
| Brief description |
colorless liquid with a slightly ethereal odor |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 116.95 g mol −1 | |||||||||||||||
| Physical state |
liquid |
|||||||||||||||
| density |
1.25 g cm −3 |
|||||||||||||||
| Melting point |
−103.5 ° C |
|||||||||||||||
| boiling point |
32 ° C |
|||||||||||||||
| Vapor pressure |
648 mbar (20 ° C) |
|||||||||||||||
| solubility |
sparingly soluble in water (4 g l −1 at 20 ° C) |
|||||||||||||||
| Refractive index |
1.3600 |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| Toxicological data | ||||||||||||||||
| Global warming potential |
938 (based on 100 years) |
|||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . Refractive index: Na-D line , 20 ° C | ||||||||||||||||
1,1-dichloro-1-fluoroethane is a chemical compound from the group of saturated halogenated hydrocarbons .
Extraction and presentation
1,1-Dichloro-1-fluoroethane can be obtained by fluorinating 1,1,1-trichloroethane with anhydrous hydrogen fluoride at 144 ° C, whereby 1-chloro-1,1-difluoroethane is also formed.
1,1-dichloro-1-fluoroethane is one of the chemical substances that are produced in large quantities (" High Production Volume Chemical ", HPVC) and for which the Organization for Economic Cooperation and Development (OECD) collects data on possible dangers (" Screening Information Dataset ", SIDS) was prepared.
properties
1,1-dichloro-1-fluoroethane is a very volatile, colorless liquid with a slightly ethereal odor that is sparingly soluble in water. At temperatures above 200 ° C the compound decomposes, producing hydrogen fluoride, hydrogen chloride and phosgene . The compound has a global warming potential of 938 and an ozone depletion potential of 0.11.
use
1,1-dichloro-1-fluoroethane is used as a cleaning agent and for plastic foaming for PUR foams.
Individual evidence
- ↑ a b c d e f g h i j k l Entry on 1,1-dichloro-1-fluoroethane in the GESTIS substance database of the IFA , accessed on February 1, 2016(JavaScript required) .
- ↑ WM Haynes (Ed.): CRC handbook of chemistry and physics. A ready-reference book of chemical and physical data . founded by David R. Lide. 93rd edition. CRC Press, Boca Raton 2012, ISBN 978-1-4398-8049-4 , pp. 3–162 (English, limited preview in Google Book Search).
- ↑ Entry on 1,1-dichloro-1-fluoroethane in the Classification and Labeling Inventory of the European Chemicals Agency (ECHA), accessed on August 1, 2016. Manufacturers or distributors can expand the harmonized classification and labeling .
- ↑ a b data sheet 1,1-dichloro-1-fluoroethane (R 141b) (PDF) at GisChem.
- ↑ G. Myhre, D. Shindell et al .: Climate Change 2013: The Physical Science Basis . Working Group I contribution to the IPCC Fifth Assessment Report. Ed .: Intergovernmental Panel on Climate Change . 2013, Chapter 8: Anthropogenic and Natural Radiative Forcing, pp. 24-39; Table 8.SM.16 ( PDF ).
- ↑ Entry on 1,1-dichloro-1-fluoroethane in the Hazardous Substances Data Bank , accessed on March 10, 2013.
- ↑ OECD : Screening Information Dataset (SIDS) Initial Assessment Report (SIAR) for Ethane, 1,1-dichloro-1-fluoro- , accessed on October 3, 2014.
