2,4-dimethylpentan-3-one
| Structural formula | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
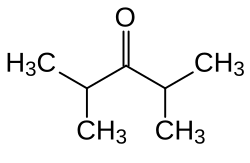
|
|||||||||||||||||||
| General | |||||||||||||||||||
| Surname | 2,4-dimethylpentan-3-one | ||||||||||||||||||
| other names |
|
||||||||||||||||||
| Molecular formula | C 7 H 14 O | ||||||||||||||||||
| Brief description |
colorless liquid with an odor similar to acetone |
||||||||||||||||||
| External identifiers / databases | |||||||||||||||||||
|
|||||||||||||||||||
| properties | |||||||||||||||||||
| Molar mass | 114.19 g mol −1 | ||||||||||||||||||
| Physical state |
liquid |
||||||||||||||||||
| density |
0.81 g cm −3 |
||||||||||||||||||
| Melting point |
−34 ° C |
||||||||||||||||||
| boiling point |
124 ° C |
||||||||||||||||||
| Vapor pressure |
27 hPa (30 ° C) |
||||||||||||||||||
| solubility |
|
||||||||||||||||||
| Refractive index |
1.3990 (20 ° C) |
||||||||||||||||||
| safety instructions | |||||||||||||||||||
|
|||||||||||||||||||
| Toxicological data | |||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . Refractive index: Na-D line , 20 ° C | |||||||||||||||||||
2,4-Dimethylpentan-3-one ( diisopropyl ketone ) is a chemical compound from the group of alkanones .
Extraction and presentation
2,4-Dimethylpentan-3-one can be obtained by passing 2,2,4-trimethyl-1,3-pentanediol over an aluminum oxide catalyst.
2,4-Dimethylpentan-3-one is also produced by oxidation of the corresponding secondary carbinol, which is an important by-product of methanol.
It can also be made from isobutyric acid and by the reaction of sodium amide and iodomethane with acetone , methyl ethyl ketone , diethyl ketone and methyl isopropyl ketone .
properties
2,4-Dimethylpentan-3-one is an easily flammable colorless liquid with an acetone-like odor that is sparingly soluble in water.
use
2,4-Dimethylpentan-3-one is used as an intermediate in the production of α-aryl ketones and in Grignard reactions .
safety instructions
The vapors of 2,4-dimethylpentan-3-one can form an explosive mixture with air ( flash point 18 ° C, ignition temperature 478 ° C).
Individual evidence
- ↑ a b c d e f g h i j k l m Entry on 2,4-dimethylpentan-3-one in the GESTIS substance database of the IFA , accessed on January 14, 2020(JavaScript required) .
- ↑ a b c data sheet diisopropyl ketone, 98% from AlfaAesar, accessed on August 12, 2018 ( PDF )(JavaScript required) .
- ^ William M. Haynes: CRC Handbook of Chemistry and Physics, 96th Edition . CRC Press, 2015, ISBN 978-1-4822-6097-7 , pp. 218 ( limited preview in Google Book search).
- ↑ Entry on 2,4-dimethylpentan-3-one in the Classification and Labeling Inventory of the European Chemicals Agency (ECHA), accessed on August 31, 2018. Manufacturers or distributors can expand the harmonized classification and labeling .
- ↑ Google Patents: US3660492A - Synthesis of diisopropyl ketone - Google Patents , accessed August 12, 2018.
- ^ A b Frank C. Whitmore: Organic Chemistry, Volume One Part I: Aliphatic Compounds Part II: Alicyclic Compounds . Courier Corporation, 2012, ISBN 978-0-486-60700-9 , pp. 222 ( limited preview in Google Book search).

