2-butanethiol
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|

|
||||||||||||||||
| Simplified structural formula without stereochemistry | ||||||||||||||||
| General | ||||||||||||||||
| Surname | 2-butanethiol | |||||||||||||||
| other names |
|
|||||||||||||||
| Molecular formula | C 4 H 1 0 S | |||||||||||||||
| Brief description |
colorless, malodorous liquid |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 90.18 g mol −1 | |||||||||||||||
| Physical state |
liquid |
|||||||||||||||
| density |
0.83 g cm −3 (20 ° C) |
|||||||||||||||
| Melting point |
-165 ° C |
|||||||||||||||
| boiling point |
82–88 ° C (1013 hPa) |
|||||||||||||||
| Vapor pressure |
108 hPa (25 ° C) |
|||||||||||||||
| solubility |
|
|||||||||||||||
| Refractive index |
1.436 (20 ° C) |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| Toxicological data | ||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . Refractive index: Na-D line , 20 ° C | ||||||||||||||||
2-Butanethiol is a chemical compound from the group of thiols .
Extraction and presentation
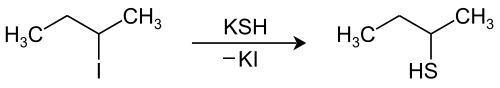
2-Butanethiol can be obtained by reacting 2-butene with hydrogen sulfide in the presence of a catalyst (usually a halide) or from 2-iodobutane or 2-bromobutane using an alcoholic potassium hydrogen sulfide solution.
This reaction produces a 1: 1 mixture of ( R ) -2-butanethiol and ( S ) -2-butanethiol, that is, racemic 2-butanethiol [synonym: ( RS ) -2-butanethiol]. If “2-butanethiol” is mentioned in the scientific literature or in this article without further details, the racemate is meant.
properties
2-Butanethiol is a colorless, malodorous liquid that is sparingly soluble in water.
use
2-Butanethiol is used as an odor additive for natural gas or as an intermediate in the manufacture of other chemical compounds (such as cadusafos ).
Individual evidence
- ↑ a b c d e f g h i j k l Data sheet 2-butanethiol (PDF) from Merck , accessed on January 27, 2019.
- ↑ a b c Entry on 2-butanethiol in the Hazardous Substances Data Bank , accessed January 27, 2019.
- ↑ Data sheet 2-butanethiol, 98% from Sigma-Aldrich , accessed on January 27, 2019 ( PDF ).