Nitroxylenes
The nitroxylenes (also called dimethylnitrobenzenes or nitroxylenes ) form a group of substances that are derived from toluene or xylenes as well as from nitrobenzene . The structure consists of a benzene ring with two attached methyl groups (-CH 3 ) and a nitro group (-NO 2 ) as substituents . Their different arrangement results in six constitutional isomers with the empirical formula C 8 H 9 NO 2 . They are obtained by nitrating xylene and were first systematically studied by Emilio Noelting in 1885 . Depending on the degree of nitration, they are divided into:
- Nitroxylenes (mononitroxylenes): six constitutional isomers
- Dinitroxylenes
- Trinitroxylenes
Extraction and properties
The nitroxylenes be nitriding (for example, with a. Nitric acid - sulfuric acid mixture) of xylene recovered; the conversion of m-xylene produces an isomer mixture of 75% 2,4-dimethylnitrobenzene (4-nitro- m -xylene), 25% 2,6-dimethylnitrobenzene (2-nitro- m -xylene), and a little 3.5 -Dimethylnitrobenzene (5-nitro- m -xylene). They occur here as yellowish liquids that are poorly soluble in water, but soluble in organic solvents. The average density is 1.12 g / cm 3 ; the nitroxylenes are therefore all heavier than water.
The nitroxylenes have a low acute oral toxicity ( LD 50 µm or above 2000 mg / kg in female rats), there is evidence of a mutagenic effect for most of the isomers.
| Nitroxylenes | |||||||||||||
| Surname | 2,3-dimethyl- nitrobenzene |
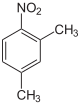
2,4-dimethyl- nitrobenzene |
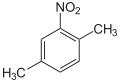
2,5-dimethyl- nitrobenzene |
2,6-dimethyl- nitrobenzene |
3,4-dimethyl- nitrobenzene |
3,5-dimethyl- nitrobenzene |
|||||||
| other names | 3-nitro- o- xylene | 4-nitro- m- xylene | 2-nitro- p- xylene | 2-nitro- m- xylene | 4-nitro- o- xylene | 5-nitro- m- xylene | |||||||
| Structural formula |

|
|
|

|

|

|
|||||||
| CAS number | 83-41-0 | 89-87-2 | 89-58-7 | 81-20-9 | 99-51-4 | 99-12-7 | |||||||
| 25168-04-1 (mixture of isomers) | |||||||||||||
| PubChem | 6739 | 6991 | 6974 | 6672 | 7440 | 7426 | |||||||
| Molecular formula | C 8 H 9 NO 2 | ||||||||||||
| Molar mass | 151.16 g mol −1 | ||||||||||||
| Physical state | liquid | firmly | |||||||||||
| Brief description | yellowish liquid | yellow, crystalline solids | |||||||||||
| Melting point | 15 ° C | 2 ° C | 2 ° C | 14-16 ° C | 30.5-32 ° C | 72-74 ° C | |||||||
| boiling point | 240-245 ° C | 245 ° C | 240 ° C | 222-224 ° C | 255-257 ° C | 273 ° C (985 hPa) | |||||||
| solubility | poorly soluble in water, soluble in ethanol, ether and chloroform | ||||||||||||
|
GHS labeling |
|
|
|
|
|
|
|||||||
| H and P phrases | 312-319 | 302 | no H-phrases | 312-332-411 | 302 | 302-312-332 | |||||||
| no EUH phrases | no EUH phrases | no EUH phrases | no EUH phrases | no EUH phrases | no EUH phrases | ||||||||
|
261-280-302 + 352 304 + 340-322-501 |
no P-phrases | no P-phrases |
261-280-302 + 352 304 + 340-322-501 |
no P-phrases |
261-280-302 + 352 304 + 340-322-501 |
||||||||
use
Nitroxylenes are used as intermediates in the manufacture of dyes (e.g. xylidines ), insecticides, herbicides, and pharmaceuticals. Nitro o -xylene is used to produce 3,4- xylidine used in the several stages of riboflavin (vitamin B 2 is produced). Since nitroxylenes are also used in the manufacture of explosives , they, like nitrotoluenes and 2,4-dinitrotoluene, can be detected as old armaments in the soil and groundwater.
Web links
Individual evidence
- ↑ a b Production of nitroxylenes ( Memento from March 9, 2012 in the Internet Archive ).
- ↑ Comparative consideration of the toxicological effects of five different nitroxylenes ( page no longer available , search in web archives ) Info: The link was automatically marked as defective. Please check the link according to the instructions and then remove this notice. (PDF; 58 kB), accessed on August 22, 2012.
- ↑ a b c Entry for CAS no. 83-41-0 in the GESTIS substance database of the IFA , accessed on October 14, 2012(JavaScript required) .
- ↑ a b c Entry for CAS no. 89-87-2 in the GESTIS substance database of the IFA , accessed on October 14, 2012(JavaScript required) .
- ↑ a b c Entry for CAS no. 89-58-7 in the GESTIS substance database of the IFA , accessed on October 14, 2012(JavaScript required) .
- ↑ a b c Entry for CAS no. 81-20-9 in the GESTIS substance database of the IFA , accessed on October 14, 2012(JavaScript required) .
- ↑ a b c Entry for CAS no. 99-51-4 in the GESTIS substance database of the IFA , accessed on October 14, 2012(JavaScript required) .
- ↑ a b c Entry for CAS no. 99-12-7 in the GESTIS substance database of the IFA , accessed on October 14, 2012(JavaScript required) .
- ↑ Toxicological assessment of 4-nitro-1,3-dimethylbenzene (PDF) at the professional association raw materials and chemical industry (BG RCI), accessed on August 22, 2012.
- ↑ Toxicological evaluation from page no longer available , search in web archives: 4-Nitro-1,2-dimethylbenzene ( page no longer available , search in web archives ) Info: The link was automatically marked as defective. Please check the link according to the instructions and then remove this notice. (PDF) from the trade association raw materials and chemical industry (BG RCI), accessed on August 22, 2012.

