Diaminopyrimidines
Diaminopyrimidines are basic organic compounds with two amino groups on one pyrimidine ring . Four isomers can arise depending on the arrangement of the amino groups on the ring.
Representative
| Diaminopyrimidines | ||||||||
| Surname | 2,4-diaminopyrimidine | 2,5-diaminopyrimidine | 4,5-diaminopyrimidine | 4,6-diaminopyrimidine | ||||
| other names | Pyrimidine-2,4-diamine | Pyrimidine-2,5-diamine | Pyrimidine-4,5-diamine | Pyrimidine-4,6-diamine | ||||
| Structural formula |

|

|

|
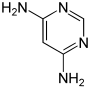
|
||||
| CAS number | 156-81-0 | 22715-27-1 | 13754-19-3 | 2434-56-2 | ||||
| PubChem | 67431 | 229075 | 83703 | 79608 | ||||
| Molecular formula | C 4 H 6 N 4 | |||||||
| Molar mass | 110.12 g mol −1 | |||||||
| Physical state | firmly | |||||||
| Brief description | light yellow solid | orange to brown solids | ||||||
| Melting point | 143-147 ° C | 204-206 ° C | ||||||
| solubility | soluble in water | |||||||
|
GHS labeling |
|
|
|
|
||||
| H and P phrases | 315-319-335 | see above | no H-phrases | 315-319 | ||||
| no EUH phrases | see above | no EUH phrases | no EUH phrases | |||||
| 261-305 + 351 + 338 | see above | no P-phrases |
264-280-305 + 351 + 338-337 + 313- 302 + 352-332 + 313-362 + 364 |
|||||
use
Derivatives of diaminopyrimidines are used as pharmacological agents. They often act as folic acid antagonists and inhibit the bacterial dihydrofolate reductase . Since folic acid is an important growth substance for many bacteria and the diaminopyrimidines block the synthesis of folic acid within a bacterium, they act as an antibiotic .
Representatives of this group are for example:
- Aminopterin
- Iclaprim
- Kopexil (used as "Aminexil" to treat hair loss)
- Methotrexate
- Pyrimethamine
- Minoxidil (used to treat hair loss)
- Trimethoprim (most important representative of the group)
- 2,5-diaminopyrimidine nucleoside triphosphate (occurs in the biosynthesis of biopterin from GTP )
The structurally similar diaminopyridine derivatives, e.g. B. 3,4-diaminopyridine are used in neurological diseases such as Lambert-Eaton syndrome . Their influence on presynaptic potassium channels and the increased release of transmitters mediated by them is used.
Related links
- Aminopyrimidines Pyrimidines with an amino group
- Triaminopyrimidines Pyrimidines with three amino groups
- Diaminopyridines Pyridines with two amino groups
Web links
- Freepatentsonline: 2,4-Diaminopyrimidines
- Freepatentsonline: Diaminopyrimidines as modulators for an EP2 receptor
Individual evidence
- ↑ a b c data sheet 2,4-diaminopyrimidine from Sigma-Aldrich , accessed on November 13, 2017 ( PDF ).
- ↑ a b data sheet 4,5-diaminopyrimidine from Sigma-Aldrich , accessed on November 5, 2016 ( PDF ).
- ↑ Entry on 4,6-diaminopyrimidines at TCI Europe, accessed on November 13, 2017.
- ↑ Entry on diaminopyrimidine in Flexikon , a wiki from DocCheck , accessed on November 26, 2015.
