4- tert- butyltoluene
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
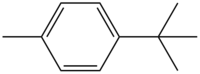
|
||||||||||||||||
| General | ||||||||||||||||
| Surname | 4- tert- butyltoluene | |||||||||||||||
| other names |
|
|||||||||||||||
| Molecular formula | C 11 H 16 | |||||||||||||||
| Brief description |
colorless liquid |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 148.25 g mol −1 | |||||||||||||||
| Physical state |
liquid |
|||||||||||||||
| density |
0.82 g cm −3 (20 ° C) |
|||||||||||||||
| Melting point |
−52 ° C |
|||||||||||||||
| boiling point |
193 ° C |
|||||||||||||||
| Vapor pressure |
0.867 hPa (25 ° C) |
|||||||||||||||
| solubility |
heavy in water (6 g l −1 at 20 ° C) |
|||||||||||||||
| Refractive index |
1.492 (20 ° C) |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| Toxicological data | ||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . Refractive index: Na-D line , 20 ° C | ||||||||||||||||
4- tert- butyltoluene is a chemical compound from the group of alkylbenzenes .
Extraction and presentation
4- tert -Butyltoluol can be prepared by alkylation of toluene with isobutylene can be obtained.
properties
4- tert -Butyltoluene is a flammable colorless liquid that is sparingly soluble in water.
use
4- tert- butyltoluene is used as a solvent for synthetic resins and as an intermediate in the manufacture of other chemical compounds.
4- tert -Butylbenzaldehyde can be obtained by oxidizing 4- tert -butyltoluene with hydrogen peroxide in glacial acetic acid , catalyzed by bromide ions in combination with cobalt (II) acetate or cerium (III) acetate .
The standard process for the production of 4- tert- butylbenzoic acid is the oxidation of 4- tert- butyltoluene in the liquid phase with atmospheric oxygen under catalysis with cobalt salts , preferably cobalt (II) acetate .
safety instructions
The vapors of 4- tert- butyltoluene can form an explosive mixture with air ( flash point > 55 ° C, ignition temperature 510 ° C).
Individual evidence
- ↑ a b c d e f g h i j k l m Entry on 4-tert-butyltoluene in the GESTIS substance database of the IFA , accessed on January 8, 2020(JavaScript required) .
- ↑ Data sheet 4-tert-butyltoluene, 95% from Sigma-Aldrich , accessed on August 21, 2017 ( PDF ).
- ↑ a b Entry on 4-tert-butyltoluene in the Hazardous Substances Data Bank , accessed on August 21, 2017.
- ^ Leon GA van de Water, Arati Kaza, James K. Beattie, Anthony F. Masters, Thomas Maschmeyer: Partial Oxidation of 4- tert -Butyltoluene Catalyzed by Homogeneous Cobalt and Cerium Acetate Catalysts in the Br - / H 2 O 2 / Acetic Acid System: Insights into Selectivity and Mechanism . In: Chemistry - A European Journal . tape 13 , no. 28 , 2007, pp. 8037-8044 , doi : 10.1002 / chem.200700371 .
- ↑ GW Hearne, TW Evans, VW Buls, CG Schwarzer: Preparation of tert-Alkylbenzoic Acids by Liquid Phase Catalytic Oxidation . In: Ind. Eng. Chem. Band 47 , no. 11 , 1955, pp. 2311-2313 , doi : 10.1021 / ie50551a035 .
- ↑ Patent US4603220 : Process for the preparation of aromatic monocarboxylic acids from toluene and substituted toluenes. Applied on July 18, 1984 , published July 29, 1986 , applicant: Dynamit Nobel AG, inventor: M. Feld.



