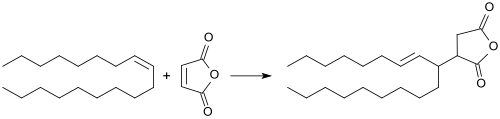
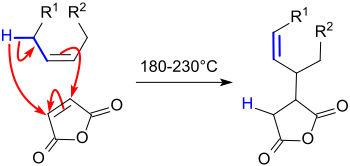Alkenyl succinic anhydrides
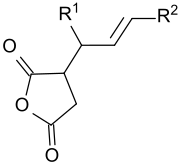
Alkenyl succinic anhydrides ( ASA ) are derived from the five-membered ring system of succinic anhydride (dihydrofuran-2,5-dione or oxolane -2,5 -dione), which has a long-chain (C 14 to C 22 ) branched iso - alkene residue carries. The liquid compounds are used in a variety of ways, in particular in surface and bulk sizing - also referred to as sizing or sizing - of paper , cardboard and cardboard , as well as in the hydrophobization of cellulose fibers . The products treated therewith have reduced penetration of aqueous media, such as. B. ink , printing ink or beverages, such as milk or fruit juices .
In the USA, alkenylsuccinic anhydrides have established themselves as the preferred paper sizing agents, while in Europe the use of alkyl ketene dimers (AKD) predominates.
history
The reaction of maleic anhydride (MAN) with aliphatic simply unsaturated n - and iso -alkenes in the so-called "cracked distillate" - one at the cracking of petroleum resulting distillate fraction having a high content of unsaturated hydrocarbons - has been described in a patent already 1936th
The olefins were then reacted in excess with maleic anhydride at 200 ° C. in an autoclave , then the excess alkene was distilled off in vacuo, the alkenylsuccinic anhydride obtained was hydrolyzed with dilute sodium hydroxide solution and the disodium salt reacted with acid to form alkenylsuccinic acid. Among the "many useful applications" of the products obtained, however, the use as a size has not yet been mentioned.
With a pre-cleaned "cracked petroleum distillate", 30% higher reaction yields were achieved at 210 ° C. in the autoclave and it was found that the hydrolysis of the succinic anhydride can be carried out using steam.
While the dark brown color and black, tar-like polymer residues in the alkenylsuccinic anhydrides produced according to the processes at that time were of little importance in the early technical uses as lubricants and rust inhibitors, clear, i.e. H. polymer-free, and less dark-colored ASA is required.
The first patented use of alkenylsuccinic anhydrides in 1959 for the water-repellent finish of cellulose-based ( cotton) textiles and the transfer of this concept to the hydrophobization of paper using iso- octadecenylsuccinic anhydrides (C 18 -ASA) in 1963 required the provision of liquid, particle-free and as light as possible ASA -Variants.
After initial difficulties with the use of ASA for paper sizing, especially the rapid hydrolysis to alkenylsuccinic acids, the formation of poorly soluble calcium salts in hard water and deposits in the paper machine,
the process parameters could be adjusted so that alkenylsuccinic anhydrides could establish themselves as currently the most important sizing agents in the USA.
Manufacturing
In the ene reaction of alkenes with maleic anhydride at high temperatures (> 200 ° C) competing reactions such. B. oxidation, condensation and (alternating co-) polymerization, undesired secondary products are formed which reduce the yield of ASA and negatively affect the color, consistency and processability of the end products.
The 1-alkenylsuccinic anhydrides obtained from unbranched 1-alkenes and solid at room temperature are not suitable for bulk sizing from aqueous emulsions.
The even-numbered 1-alkenes CH 2 = CH- (CH 2 ) n -CH 3 with terminal double bonds, which are accessible by the so-called SHOP process by oligomerization of ethylene, are isomerized on magnesium oxide catalysts with the double bond being shifted into internal positions along the carbon chain and by fractional distillation into desired sections within the preferred ASA for C 14 C - 22 separated fraction.
The alkenylsuccinic anhydrides are produced with an excess of iso- alkenes at temperatures> 200 ° C. under nitrogen for more than 3 hours; the excess iso- alkene is distilled off under reduced pressure.
Extensive patent literature is available on methods for suppressing the side reactions in the production of ASA from iso- alkenes and MAN. Thus, by adding antioxidants or polymerization inhibitors, such as. B. hydroquinone or phenothiazine reduces the polymer formation and the maleic anhydride can be converted quantitatively. Later it was also possible, for. B. by washing with water to improve the color of the solid ASA obtained in the reaction of 1-alkenes with maleic anhydride from dark brown to amber. By combining a reducing agent , such as. B. a trialkyl phosphite with a phenolic antioxidant, a substantial lightening and reduction of tar formation is achieved.
The fact that the problems of discoloration and tar formation in the ASA synthesis are still only inadequately solved is shown by recent patents z. B. use a sterically hindered phenol ( BHT as primary antioxidant), a thioether (as secondary antioxidant) and N , N ′ -disalicylidene-1,2-diaminopropane as a deactivator for metal ions in a “synergistic mixture”. The C 12 -ASA formed from dodecene and MAN after six hours at 220 ° C. is pale yellow in color but still contains considerable amounts of black, tar-like decomposition products.
A dramatic improvement (around 600%) in sizing efficiency is found with alkenyl succinic anhydrides based on symmetrical alkenes such as e.g. B. the C 22 -alkene 11-docoses (by alkene metathesis from 1-dodecene) are based.
use
Alkenylsuccinic anhydrides - and the alkenylsuccinic acids accessible from them by hydrolysis - are found due to their reactive and surface-active properties and the like. a. Used as hardeners for epoxy resins , as corrosion inhibitors in lubricating oils , as reactants in alkyd resins and unsaturated polyester resins , as additive in motor oil and fuels , as components in plasticizers , as additives in toner resins , as surfactants , as water-binding and moisture- controlling additives, as metal cleaners and as chemical agents Intermediates .
In terms of quantity, by far the most important use of alkenylsuccinic anhydrides is surface and ingot sizing of paper, cardboard and cardboard, with a global consumption in 2005 of approx. 47,000 tons.
Alkenyl succinic anhydrides as paper sizing agents
The solid alkyl ketene dimers (AKD) introduced in the 1950s as water repellants for cellulose-based products were followed in the 1960s by the liquid alkenylsuccinic anhydrides. Both substance classes are hydrophobic and therefore practically insoluble in water - the solubility of the standard compound iso-octadecenylsuccinic anhydride C 18 -ASA is only 5.33 × 10 −5 mgl −1 . Because of their shorter chain length, ASA are less hydrophobic and therefore less water-repellent than AKD. Its vapor pressure is higher than that of AKD, which means that it diffuses faster into and through the paper layer, but also forms deposits on machine covers. AKD are in water at room temperature as dispersions , ASA on the other hand as oil-in-water emulsions . The most important difference, however, lies in the significantly higher reactivity of the cyclic carboxylic acid anhydride structure in the ASA compared to the diketene structure in AKD. This is accompanied by the much faster hydrolysis in an aqueous, especially alkaline, medium. Therefore, alkenyl succinic anhydrides must be converted into an emulsion immediately before they are used on the paper machine, while AKD can be delivered and stored as a stable emulsion.
ASA emulsions, like AKD dispersions, are carried out with polycations as protective colloids and retention aids, especially with cationic starch or cationic polyacrylamides (C-PAM) in a ratio of ASA to cation of approx. 2: 1, and with approx. 1% anionic or nonionic surfactants high shear produced in an optimal particle size of approx. 1 μm.
The benefits of using ASA for paper sizing include:
- Can be used in the neutral to weakly alkaline pH range (optimally pH 6–8) - AKD even pH 5–9)
- High reactivity leads to hydrophobization already in the so-called wet end of the paper process - papers treated with AKD have to mature for a while
- Easily adjustable degree of hydrophobization - not with AKD
- Lower dosage with ASA (0.1%, based on paper pulp) than with AKD (0.2%)
- Hardly any influence on paper properties - AKD can lead to sticky paper surfaces at high dosages
- Good surface sizing
- The treated paper is highly resistant to aging
The disadvantages of ASA are in particular:
- Rapid hydrolysis of the AKD after the emulsion has been produced → very low storage stability
- Emulsion production only at the paper machine with high investment and operating costs
- Risk of sticky deposits forming in the machine
- Oily deposits in the dryer area
Faster reactions and lower quantities used (and thus material costs in one of the most cost-intensive sub-steps in paper production) speak in favor of using ASA, while AKD provides a more sustainable hydrophobization and better adhesion in material composites with e.g. B. cause polyethylene films for water and acid-resistant beverage packaging.
Paper sizing with alkenyl succinic anhydrides
As with AKD, paper sizing with ASA essentially takes place in three steps
- the retention or fixation of the ASA emulsion droplets on the moist paper pulp
- the breaking of the emulsion droplets with the spreading on the surface and penetration into the paper pulp
- the formation of a hydrophobic paper surface with contact angles> 100 ° through covalent linkage ( esterification ) of the ASA molecules with the hydroxyl groups of the cellulose
Studies published by Japanese authors as early as the 1990s raise considerable doubts about this idealized representation of the hydrophobization of cellulose fiber surfaces. It could be demonstrated that no or only very little ASA is bound to cellulose via covalent ester bonds.
This suggests that the hydrophobization brought about by ASA is more likely due to an adsorptive and associative interaction of the alkenylsuccinic acids formed by rapid hydrolysis in the aqueous medium with the constituents of the paper pulp (cellulose fibers, fillers such as calcium carbonate as PCC ( precipitated) calcium carbonate ) or cationic polymers), whereby even small (<0.15%) amounts (quote from MA Hubbe: "It doesn't take much") and also inhomogeneous distribution ("It doesn't have to be uniform") sufficient for a high degree of hydrophobicity.
The shift in the paper raw material base to recycling material - the so-called secondary fibers - the rapid increase in the volumes of cardboard and cardboard at the expense of printing paper, the growing speeds (> 120 km / h) and production volumes (max. Daily capacity of a single machine> 4,500 tons or .> 1.5 million tons per year) pose enormous challenges for the further development of ASA-based sizing agents for cellulose and their formulations, also in view of the understanding of the fundamental processes of hydrophobing cellulose fibers, which is still strongly supported by empirical evidence.
literature
- MA Hubbe: Acidic and Alkaline Sizings for Printing, Writing, and Drawing Papers . Ed .: G. Edwards. The Book and Paper Group Annual 23, Charlottesville, VA, USA 2004, p. 139-151 ( ncsu.edu ).
- P. Bajpai: Pulp and Paper Chemicals, 1st edit. Elsevier, Amsterdam 2015, ISBN 978-0-12-803408-8 .
- T. Arnson et al .: The Sizing of Paper, 3rd edit. Eds .: JM Gess, JM Rodriguez. TAPPI Press, Atlanta, GA, USA 2005, ISBN 1-59510-073-3 .
- MA Hubbe: Paper's Resistance to Water - A Review of Internal Sizing Chemicals and their Effects . In: BioResources . tape 2 , no. 1 , 2006, p. 106-145 ( ncsu.edu ).
- C. Hagiopol, JW Johnston: Chemistry of Modern Papermaking . CRC Press, Boca Raton, FL, GA, USA 2011, ISBN 978-1-4398-5646-8 .
Individual evidence
- ↑ Patent US2055456 : Process and product relating to olefin derivatives. Applied October 6, 1931 , published September 22, 1936 , Applicant: Shell Development Co., Inventor: E. Eichwald.
- ↑ Patent US2230005 : Process for the production of valuable products from cracked petroleum distillates. Applied January 4, 1938 , published January 28, 1941 , Applicant: Shell Development Co., Inventor: FR Moser.
- ↑ Patent US2903382 : Treatment of fabric with alkenyl succinic acids and anhydrides to impart water repellency. Applied on June 23, 1958 , published September 8, 1959 , Applicant: Armor and Co., Inventor: RJ Berls.
- ↑ Patent US3102064 : Novel paper sizing process. Registered September 8, 1961 , published August 27, 1963 , Applicant: National Starch and Chemical Corp., Inventor: OB Wurzburg, ED Mazzarella.
- ↑ Patent US3821069 : Process of sizing paper with a reaction product of maleic anhydride and an internal olefin. Applied January 2, 1973 , published June 28, 1974 , applicant: National Starch and Chemical Corp., inventor: OB Wurzburg.
- ^ PB Sellars, L. Lue, IS Burns, DN Work: Freezing properties of alkenyl succinic anhydrides derived from linear isomerized olefins . In: Ind. Eng. Chem. Res. Volume 55 , no. 8 , 2016, p. 2287-2292 , doi : 10.1021 / acs.iecr.5b04769 .
- ↑ Patent US5104486 : Alkenyl succinic anhydride compositions. Applied on February 27, 1987 , published April 14, 1992 , applicant: Chevron Research Co., inventor: WA Sweeney.
- ↑ Patent US3412111 : Process for reacting an olefin with maleic anhydride to obtain an alkenyl succinic anhydride. Applied on June 2, 1965 , published November 19, 1968 , Applicant: Gulf Research & Development Co., Inventor: PG Irwin, CM Selwitz.
- ↑ Patent US4158664 : Process for improving color of certain alkenyl succinic ahydrides. Registered June 17, 1977 , published June 19, 1979 , Applicant: Gulf Research & Development Co., Inventor: CM Selwitz, HI Thayer.
- ↑ Patent US5021169 : Alkenyl succinic anhydrides process. Applied on November 13, 1989 , published June 4, 1991 , Applicant: Ethyl Corp., Inventor: K.-H. Shin, PS Hale.
- ↑ Patent US8350058B2 : Method for producing alkenyl succinic anhydrides. Registered on December 3, 2005 , published on January 8, 2013 , applicant: Clariant Produkte (Deutschland) GmbH, inventor: D. Leinweber, T. Rau.
- ↑ Patent US7455751B2 : Use of alkenyl succinic anhydride compounds derived from symmetrical olefins in internal sizing for paper production. Filed April 15, 2005 , published November 25, 2008 , Applicant: Nalco Co., Inventors: WJ Ward, K. Andruszkiewicz, RT Gray, TP McGinnis, RW Novak.
- ↑ a b c S. Porkert: Physico-chemical processes during reactive paper sizing with alkenyl succinic anhydride (ASA) . Technical University of Dresden, 2016, urn : nbn: de: bsz: 14-qucosa-219620 (dissertation).
- ↑ Patent US2627477 : Higher alkyl ketene dimer emulsion. Filed October 6, 1949 , published February 3, 1953 , applicant: Hercules Powder Co., inventor: WF Downey.
- ^ J. Lindfors, J. Salmi, J. Laine, P. Stenius: AKD and ASA model surfaces: Preparation and characterization . In: BioResources . tape 2 , 2007, p. 652-670 ( ncsu.edu ).
- ↑ Patent US5606773 : Emulsification of alkenyl succinic anhydride sizing agents. Applied on December 10, 1984 , published on August 19, 1986 , applicant: Nalco Chemical Co., inventor: RW Novak.
- ^ E. Martorana, J. Belle, S. Kleemann: ASA optimization - Control of particle size, stability and hydrolysis . In: Professional Papermaking . tape 5 , no. 2 , 2008, p. 34–42 , urn : nbn: de: bvb: m347-opus-172 .
- ↑ PM 2- the largest paper machine in the world . In: Voith Paper . tape 31 , 2010, p. 16-19 ( blogus.de [PDF]).