Articain
| Structural formula | ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||||||||
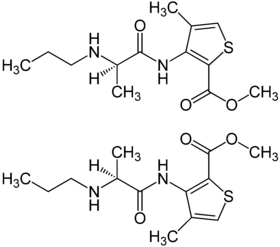
| 1: 1 mixture of ( R ) -form (top) and ( S ) -form (bottom) |
||||||||||||||||||||||
| General | ||||||||||||||||||||||
| Non-proprietary name | Articain | |||||||||||||||||||||
| other names |
|
|||||||||||||||||||||
| Molecular formula | C 13 H 20 N 2 O 3 S | |||||||||||||||||||||
| External identifiers / databases | ||||||||||||||||||||||
|
||||||||||||||||||||||
| Drug information | ||||||||||||||||||||||
| ATC code | ||||||||||||||||||||||
| Drug class | ||||||||||||||||||||||
| properties | ||||||||||||||||||||||
| Molar mass | 284.37 g · mol -1 | |||||||||||||||||||||
| Physical state |
firmly |
|||||||||||||||||||||
| Melting point |
177–178 ° C (articaine hydrochloride) |
|||||||||||||||||||||
| boiling point |
162-167 ° C (39 Pa ) (Articaine) |
|||||||||||||||||||||
| pK s value |
7.9 |
|||||||||||||||||||||
| safety instructions | ||||||||||||||||||||||
|
||||||||||||||||||||||
| Toxicological data | ||||||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||||||||
Articain (until 1984 Carticain ) is a drug from the group of local anesthetics that is mainly used in dentistry. Articain was patented by the then Farbwerke Hoechst in 1969 and 1974 .
application
Infiltration and conduction anesthesia (0.5–4%, hyperbaric up to 5%). Articain is the most frequently used local anesthetic in dentistry in Germany.
chemistry
Articaine is commonly classified as an amide- type local anesthetic because the intermediate chain contains such an amide bond. However, the thiophene ring also has a side chain with an ester bond, so that Articaine occupies a special position within the classification of local anesthetics. The cleavage of the ester bond by esterases in the plasma also leads to a metabolite without any local anesthetic effect.
effect
Articain changes the membrane permeability so that the sodium influx into the nerve fiber is inhibited and thus the formation of an action potential is prevented. The onset of action is 2 minutes and the duration of action is 0.5-3 hours, the duration of action being mature between 20 minutes and 75 minutes.
The drug penetrates well into bone tissue. It is inactivated by hydrolysis and exposure of a hydrophilic acid group, whereby it can be quickly eliminated from the body. The plasma half-life is 25 minutes. Articain has a high plasma protein binding and good general tolerance and is the drug of choice for pregnant women.
Unwanted side effects
In patients with a cholinesterase deficiency , the indication must be made strict, since a prolonged and possibly stronger effect can be expected. Articain should only be used with special caution in:
- Disorders of blood coagulation ,
- severe kidney or liver problems,
- simultaneous treatment with halogenated inhalation anesthetics and
- History of known epilepsy .
Stereoisomerism
Articaine contains a stereocenter , so it is chiral . The racemate [1: 1 mixture of ( R ) isomer and ( S ) isomer] is used as a local anesthetic .
Trade names
Ultracaine (D, A),
With epinephrine : Alphacain (CH), Rudocain (CH), Septanest (D, A, CH), Sopira Citocartin (D, A), Ubistesin (D, A, CH), Ultracain DS (D, A, CH)
Individual evidence
- ↑ a b c d Entry on Articain. In: Römpp Online . Georg Thieme Verlag, accessed on November 10, 2014.
- ↑ a b Data sheet Articaine hydrochloride, ≥98% (HPLC) from Sigma-Aldrich , accessed on October 31, 2016 ( PDF ).
- ↑ Data sheet ARTICAINE HYDROCHLORIDE CRS (PDF) at EDQM , accessed on August 7, 2008.
- ↑ Judith M. Jacob, Sieg über den pain , DENTAL MAGAZIN 6/2005, p. 50. Retrieved on July 3, 2016.
- ^ DE Becker & KL Reed: Essentials of Local Anesthetic Pharmacology . In: Anesth. Prog. , 2006; 53: 98-109, PMID 17175824 .
- ↑ Specialist information Ultracain