Colistin
| Structural formula | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
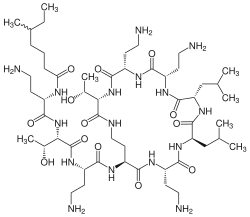
|
|||||||||||||
| General | |||||||||||||
| Non-proprietary name | Colistin | ||||||||||||
| other names |
Polymyxin E. |
||||||||||||
| Molecular formula | C 52 H 98 N 16 O 13 | ||||||||||||
| External identifiers / databases | |||||||||||||
|
|||||||||||||
| Drug information | |||||||||||||
| ATC code | |||||||||||||
| Drug class | |||||||||||||
| Mechanism of action |
Disturbance of the permeability of the cell membrane |
||||||||||||
| properties | |||||||||||||
| Molar mass | 1155.43 g · mol -1 | ||||||||||||
| safety instructions | |||||||||||||
|
|||||||||||||
| Toxicological data | |||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||
Colistin (also Polymyxin E ) is an antibiotic from the group of polymyxins . Polymyxin, first extracted from Bacillus colistinus by S. Kayama in 1950 , has been available for therapy since 1959.
application
Because of its toxicity when used systemically, colistin has long been used mainly locally as an ointment additive, orally for intestinal treatment or as an aerosol for inhalation therapy, especially in cystic fibrosis with chronic Pseudomonas colonization.
For parenteral systemic therapy in humans, there have been approved finished medicinal products in Germany again since 2012. The background for the renaissance of the antibiotic, which is primarily considered to be nephrotoxic, is the increased occurrence of carbapenem- resistant enterobacteria (so-called "CRE"; currently mainly in the USA, Israel, Greece, Turkey and North African countries). Treatment options for CRE are mostly limited to colistin, fosfomycin, and tigecycline .
The active ingredient is also used parenterally in veterinary medicine in pigs and cattle with severe septicemia . In the microbiological laboratory, the substance is used in culture media to select gram-positive bacteria (often in combination with nalidixic acid ), since colistin inhibits the growth of many gram-negative bacteria, but not that of gram-positive bacteria.
Analytics
Since colistin is considered a reserve antibiotic, the reliable qualitative and quantitative detection of the substance is of particular importance. For reliable detection in plasma and urine, the couplings of HPLC with mass spectrometry are available after adequate sample preparation . For very complex examination materials, such as B. Pig excrement are particularly complex preparations required. Highly specific evidence with appropriate sample preparation is also available for the examination of muscle tissue.
Mechanism of action
Colistimethate sodium (CMS) is a so-called prodrug which, after parenteral administration, is first converted into the pharmacologically active colistin base by hydrolysis . From 80 mg CMS (= 1 million IU ). approx. 33 mg colistin base are released. Colistin acts on gram-negative bacteria by damaging the outer membrane .
- sensitive: Salmonella , Shigella , Haemophilus influenzae , Pasteurella , Acinetobacter
- mostly sensitive: Pseudomonas aeruginosa , Escherichia coli , Enterobacter , Klebsiella
- resistant: Proteus , gonococci , meningococci and gram-positive bacteria
With polymyxin B is a cross-resistance .
resistance
Colistin is considered a reserve antibiotic for the treatment of multi-resistant Acinetobacter baumannii . Resistance is described more and more frequently in intensive care units, although there is no global limit value ( MIC ) for describing resistance. One form of resistance was initially found in farm animals, but now also in human E. coli bacteria. The resistance is triggered by the MCR-1 gene .
Side effects
- Neurotoxicity and nephrotoxicity, especially when used systemically
- Contact dermatitis when used as an ointment
- Inhalation asthma attacks
Interactions
Enhancement of side effects with nephrotoxic agents (such as aminoglycosides , vancomycin, and loop diuretics ) and agents with neuromuscular blockade (such as muscle relaxants )
Contraindications
Hypersensitivity to colistin or polymyxin B.
In the case of kidney damage, a dose adjustment (based on the measured creatinine clearance ) is necessary.
Legal
Colistin requires a prescription and therefore also requires a pharmacy.
Trade names
Human medicine:
- For oral administration: Diarönt mono (D), as a generic (D, CH)
- For inhalation: Colistin CF (D), Promixin (D), ColiFin (D), Colistineb (D)
- For intravenous administration: Promixin (D), colistimethate sodium INFECTOPHARM 1 million IU (D)
- For inhalation and intravenous administration: ColistiFlex (D)
Veterinary medicine: Animedistin , Belacol , Carbophen , Coliplus , Colipur , Colivet , Enteroxid , Klato Col , Colistinsulfat 100%
literature
- ME Falagas, SK Kasiakou, S. Tsiodras, A. Michalopoulos: The use of intravenous and aerosolized polymyxins for the treatment of infections in critically ill patients: a review of the recent literature . In: Clin Med Res . tape 4 , no. 2 , June 2006, p. 138-146 , PMID 16809407 , PMC 1483888 (free full text).
- ME Falagas, SK Kasiakou: Toxicity of polymyxins: a systematic review of the evidence from old and recent studies . In: Crit Care . tape 10 , no. 1 , February 2006, p. R27 , doi : 10.1186 / cc3995 , PMID 16507149 , PMC 1550802 (free full text).
Individual evidence
- ↑ a b Data sheet Colistin sulfate salt from Sigma-Aldrich , accessed on March 23, 2011 ( PDF ).
- ↑ Entry on colistin in the ChemIDplus database of the United States National Library of Medicine (NLM) .
- ↑ Karl Wurm, AM Walter: Infectious Diseases. In: Ludwig Heilmeyer (ed.): Textbook of internal medicine. Springer-Verlag, Berlin / Göttingen / Heidelberg 1955; 2nd edition, ibid. 1961, pp. 9–223, here: p. 55.
- ↑ Brief description Colistin In: Zeitschrift für Chemotherapie. May 2005.
- ↑ H. Heijerman, E. Westerman, S. Conway, D. Touw, G. Döring: Inhaled medication and inhalation devices for lung disease in patients with cystic fibrosis: A European consensus . In: J. Cyst. Fibros. tape 8 , no. 5 , September 2009, p. 295-315 , doi : 10.1016 / j.jcf.2009.04.005 , PMID 19559658 .
- ↑ Specialist information Promixin; Information for professionals colistimethate sodium INFECTOPHARM 1 million IU
- ^ J. Li: Colistin-Induced Apoptosis in Rat Renal Proximal Tubular Cells: Concentration- and Time-dependence and Protection by Melatonin. Poster A2-1183, ICAAC 2011.
- ↑ A. Lerner: Environmental Spread of Carbapenem-Resistant Enterobacteriaceae (CRE) by Patients Known to be Colonized with CRE. Lecture, ICAAC 2011
- ↑ W. Löscher et al. (Ed.): Pharmacotherapy in domestic and farm animals. 7th edition. Paul Parey Verlag, 2006, ISBN 3-8304-4160-6 , p. 266.
- ↑ K. Bihan, Q. Lu, M. Enjalbert, M. Apparuit, O. Langeron, JJ Rouby, C. Funck-Brentano, N. Zahr: Determination of colistin and colistimethate levels in human plasma and urine by high-performance liquid chromatography-tandem mass spectrometry. In: Ther Drug Monit. 38 (6), Sep 27, 2016, pp. 796-803. PMID 27684296
- ↑ M. Zhao, XJ Wu, YX Fan, BN Guo, J. Zhang: Development and validation of a UHPLC-MS / MS assay for colistin methanesulphonate (CMS) and colistin in human plasma and urine using weak-cation exchange solid-phase extraction. In: J Pharm Biomed Anal. 124, May 30, 2016, pp. 303-308. PMID 26970986
- ↑ T. Van den Meersche, E. Van Pamel, C. Van Poucke, L. Herman, M. Heyndrickx, G. Rasschaert, E. Daeseleire: Development, validation and application of an ultra high performance liquid chromatographic-tandem mass spectrometric method for the simultaneous detection and quantification of five different classes of veterinary antibiotics in swine manure. In: J Chromatogr A. 1429, Jan 15, 2016, pp. 248-257. PMID 26739912
- ^ JO Boison, S. Lee, J. Matus: A multi-residue method for the determination of seven polypeptide drug residues in chicken muscle tissues by LC-MS / MS. In: Anal Bioanal Chem. 407 (14), May 2015, pp. 4065-4078. PMID 25832483
- ↑ K. de With, H. Stocker, J. Borde, WV Kern: Antibiotics - new and old substances, rational regulation. In: Dtsch med Wochenschr. 137, 2012, pp. 186-189.
- ↑ NP Mortensen, JD Fowlkes, CJ Sullivan et al .: Effects of colistin on surface ultrastructure and nanomechanics of Pseudomonas aeruginosa cells . In: Langmuir . tape 25 , no. 6 , April 2009, p. 3728-3733 , doi : 10.1021 / la803898g , PMID 19227989 .
- ↑ Anastasia Antoniadou, Flora Kontopidou, Garifalia Poulakou, Evangelos Koratzanis, Irene Galani, Evangelos Papadomichelakis, Petros Kopterides, Maria Souli, Apostolos Armaganidis, Helen Giamarellou: Colistin-resistant isolates of Klebsiella pneumoniae emerging in intensive a unit patients: first report cluster . In: J. Antimicrob. Chemother. 59 (4), 2007, pp. 786-790. doi: 10.1093 / jac / dkl562
- ↑ Yun Cai, Dong Chai, Rui Wang, Beibei Liang, Nan Bai: Colistin resistance of Acinetobacter baumannii: clinical reports, mechanisms and antimicrobial strategies. In: J. Antimicrob. Chemother. 2012. doi: 10.1093 / jac / dks084
- ^ Robert Koch Institute: Acinetobacter baumannii - study
- ↑ Transferable colistin resistance in germs of farm animals in Germany. BfR, 7 January 2016.
- ↑ That means the "super-pathogen" find. MCR-1 turns off antibiotics. In: Spiegel online. May 27, 2015, accessed August 16, 2017 .
- ↑ a b c d Red List online, as of February 2016.
- ↑ AM comp. d. Switzerland, as of October 2009.