Metamitron
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|

|
||||||||||||||||
| General | ||||||||||||||||
| Surname | Metamitron | |||||||||||||||
| other names |
|
|||||||||||||||
| Molecular formula | C 10 H 10 N 4 O | |||||||||||||||
| Brief description |
yellowish solid |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 202.22 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| density |
1.350 g cm −3 |
|||||||||||||||
| Melting point |
166.6 ° C |
|||||||||||||||
| Vapor pressure |
8.6 10 −9 hPa (20 ° C) |
|||||||||||||||
| solubility |
heavy in water (1700 mg l −1 at 20 ° C) |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| Toxicological data | ||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Metamitron is a chemical compound from the triazinone group .
Extraction and presentation
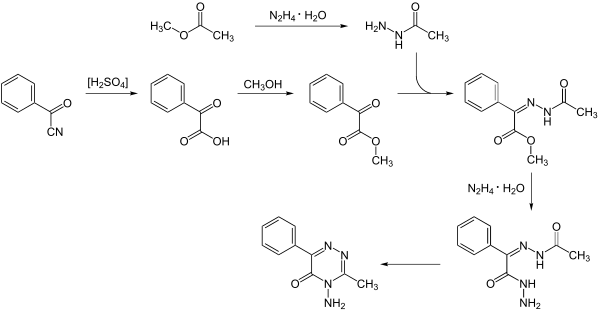
Metamitron can be obtained through a multi-stage reaction. Initially, benzoyl cyanide is reacted with sulfuric acid to form phenylglyoxylic acid and further with methanol . The intermediate product formed is reacted with acetylhydrazine , which in turn can be prepared from methyl acetate . In the further synthesis, the product is reacted with and hydrazine hydrate . Metamitron is finally formed through ring closure .
properties
Metamitron is a colorless solid that is very sparingly soluble in water. It rapidly decomposes through photolysis with deamination , is stable to acids and decomposes when exposed to bases with a pH value greater than 10.
use
Metamitron is used as a herbicide on beets against dicotyledon seed weeds in pre- and post-emergence. It was brought onto the market by Bayer AG in the mid-1970s under the name Goltix. Metamitron is also used to thin apples and pears. A short-term reduction in photosynthesis increases the fall of less developed fruits.
Admission
The active ingredient Metamitron was approved for use as a herbicide in the European Union with effect from September 1, 2009. In Germany, Austria and Switzerland, plant protection products with this active ingredient are approved.
Individual evidence
- ↑ a b c d e f g h Entry on 4-Amino-3-methyl-6-phenyl-1,2,4-triazin-5-one in the GESTIS substance database of the IFA , accessed on February 9, 2017(JavaScript required) .
- ↑ Metamitron data sheet , PESTANAL at Sigma-Aldrich , accessed on March 9, 2013 ( PDF ).
- ^ Müfit Bahadir, Harun Parlar, Michael Spiteller: Springer Umweltlexikon . Springer DE, 2000, ISBN 3-642-56998-6 , pp. 758 ( limited preview in Google Book search).
- ↑ Entry on 4-amino-3-methyl-6-phenyl-1,2,4-triazin-5-ones in the Classification and Labeling Inventory of the European Chemicals Agency (ECHA), accessed on August 1, 2016. Manufacturers or distributors can expand the harmonized classification and labeling .
- ↑ Thomas A. Unger: Pesticide Synthesis Handbook . William Andrew, 1996, ISBN 0-08-095716-1 , pp. 671 ( limited preview in Google Book search).
- ^ Terence Robert Roberts, David Herd Hutson: Metabolic Pathways of Agrochemicals: Part 1: Herbicides and Plant Growth ... Royal Society of Chemistry, 1998, ISBN 0-85404-494-9 , pp. 660 ( limited preview in Google Book search).
- ↑ Rudolf Heitefuss: Plant protection . Georg Thieme Verlag, 2000, ISBN 3-13-513303-6 , p. 219 ( limited preview in Google Book search).
- ↑ Janis Mc Farland, Orvin Burnside: The Triazine Herbicides . Elsevier, 2011, ISBN 0-08-055963-8 , pp. 39 ( limited preview in Google Book search).
- ↑ Product description from ADAMA Austria , accessed on May 23, 2018.
- ↑ Directive 2008/125 / EC of the Commission of December 19, 2008 amending Directive 91/414 / EEC of the Council to include aluminum phosphide, calcium phosphide, magnesium phosphide, cymoxanil, dodemorph, 2,5-dichlorobenzoate, metamitron, sulcotrione, tebuconazole and Triadimenol as active ingredients (PDF)
- ↑ General Directorate Health and Food Safety of the European Commission: Entry on Metamitron in the EU pesticide database; Entry in the national registers of plant protection products in Switzerland , Austria and Germany ; accessed on March 13, 2016.