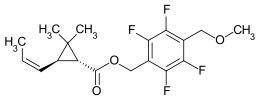
Metofluthrin
| Structural formula | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|

|
|||||||||||||||||||
| Structural formula without specifying the stereochemistry | |||||||||||||||||||
| General | |||||||||||||||||||
| Surname | Metofluthrin | ||||||||||||||||||
| other names |
(1 RS , 3 RS ; 1 SR , 3 SR ) -2,2-dimethyl-3- ( EZ ) - (prop-1-enyl) cyclopropanecarboxylic acid-2,3,5,6-tetrafluoro-4- (methoxymethyl) benzyl ester |
||||||||||||||||||
| Molecular formula | C 18 H 20 F 4 O 3 | ||||||||||||||||||
| Brief description |
yellow liquid |
||||||||||||||||||
| External identifiers / databases | |||||||||||||||||||
|
|||||||||||||||||||
| properties | |||||||||||||||||||
| Molar mass | 360.35 g mol −1 | ||||||||||||||||||
| Physical state |
liquid |
||||||||||||||||||
| density |
1.21 g cm −3 |
||||||||||||||||||
| Melting point |
−54 ° C |
||||||||||||||||||
| boiling point |
334 ° C |
||||||||||||||||||
| Vapor pressure |
1.96 mPa (25 ° C) |
||||||||||||||||||
| solubility |
|
||||||||||||||||||
| safety instructions | |||||||||||||||||||
|
|||||||||||||||||||
| Toxicological data | |||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||||||||
Metofluthrin is a chemical compound belonging to the group of pyrethroids . It consists of a total of eight different isomers , with the RTZ isomer (epsilon-metofluthrin) making up the majority with more than 75%.
Extraction and presentation
Metofluthrin can be obtained by reacting 2,3,5,6-tetrafluoro-4-methoxymethylbenzyl alcohol and a norchrysanthemum acid derivative .
properties
Metofluthrin is a yellowish liquid that is practically insoluble in water. An unspecified mixture of the eight isomers (2,3,5,6-tetrafluoro-4- (methoxymethyl) benzyl- ( EZ ) - (1 RS , 3 RS ; 1 SR , 3 SR ) -2,2- is used as metofluthrin. dimethyl-3-prop-1-enylcyclopropanecarboxylate), the technical product containing a specific isomer profile in which the RTZ isomer (2,3,5,6-tetrafluoro-4- (methoxymethyl) benzyl- (1 R , 3 R ) -2,2-dimethyl-3 - [( Z ) -prop-1-enyl] cyclopropanecarboxylate) has the largest proportion with more than 75%.
use
Metofluthrin is used as an insecticide . At the end of 2005 , Sumitomo Chemical (UK) applied for the new active ingredient to be included in Appendix I of the European Biocide Directive 98/8 / EC. On November 4, 2010, the EU Commission decided to include metofluthrin in the list of approved biocidal active ingredients. It is intended for household use. The slow evaporation using an electric vaporizer is intended to combat mosquitoes indoors. It acts as a contact poison that blocks the voltage-dependent sodium channels in the nerve membranes so that they cannot be closed again from the open state.
safety instructions
Metofluthrin has neurotoxic potential and causes tremors in rats and dogs, but not in mice or rabbits.
Web links
- EPA: Factsheet Metofluthrin
- EU: Assessment Report Metofluthrin , June 2010.
Individual evidence
- ↑ a b c d e f g h i j k BAuA : Metofluthrin ( Memento from February 26, 2012 in the Internet Archive )
- ↑ a b Sumitomo: Discovery and Development of a Novel Pyrethroid Insecticide 'Metofluthrin (SumiOne®, Eminence®) (PDF file; 232 kB).
- ↑ Entry on epsilon-metofluthrin in the GESTIS substance database of the IFA , accessed on January 2, 2020 (JavaScript required)
- ↑ Entry on 2,3,5,6-tetrafluoro-4- (methoxymethyl) benzyl (1R, 3R) -2,2-dimethyl-3 - [(1Z) -prop-1-en-1-yl] cyclopropanecarboxylate; epsilon-metofluthrin in the Classification and Labeling Inventory of the European Chemicals Agency (ECHA), accessed on January 2, 2020. Manufacturers or distributors can expand the harmonized classification and labeling .
- ↑ External identifiers of or database links to Trans- (Z) -metofluthrin : CAS number: 240494-71-7, ECHA InfoCard: 100.278.770 , GESTIS substance database : 536376 , PubChem : 12128785 , ChemSpider : 28534042 , Wikidata : Q27159726 .
- ↑ ECHA (Ed.): CLH report - Proposal for Harmonized Classification and Labeling: Substance Name: Epsilon-metofluthrin . 2015, p. 12 ( europa.eu [PDF]).
- ↑ Directive 2010/71 / EU of the Commission of November 4, 2010 amending Directive 98/8 / EC of the European Parliament and of the Council to include the active ingredient metofluthrin in Appendix I (PDF)