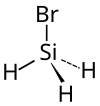
Monobromosilane
| Structural formula | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||
| General | ||||||||||
| Surname | Monobromosilane | |||||||||
| other names |
Bromosilane |
|||||||||
| Molecular formula | SiH 3 Br | |||||||||
| Brief description |
colorless gas |
|||||||||
| External identifiers / databases | ||||||||||
|
||||||||||
| properties | ||||||||||
| Molar mass | 111.01 g mol −1 | |||||||||
| Physical state |
gaseous |
|||||||||
| density |
4.538 g l −1 |
|||||||||
| Melting point |
−94 ° C |
|||||||||
| boiling point |
1.9 ° C |
|||||||||
| safety instructions | ||||||||||
|
||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||
Monobromosilane is a chemical compound from the group of silanes .
Extraction and presentation
Monobromosilane can be obtained by reacting monosilane with hydrogen bromide .
It can also be obtained by reacting phenylsilane with hydrogen bromide.
properties
Monobromosilane is a colorless gas that can spontaneously ignite in air but is thermally stable in its pure state. In the solid state it has an orthorhombic crystal structure with the space group Cmc 2 1 (space group no. 36) .
Individual evidence
- ↑ a b c d e f William M. Haynes: CRC Handbook of Chemistry and Physics, 93rd Edition . CRC Press, 2016, ISBN 978-1-4398-8050-0 , pp. 87 ( limited preview in Google Book search).
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.
- ^ A b E. G. Rochow: The Chemistry of Silicon Pergamon International Library of Science, Technology, Engineering and Social Studies . Elsevier, 2013, ISBN 978-1-4831-8755-6 , pp. 1366.1371 ( limited preview in Google Book search).
- ↑ Georg Brauer (Ed.), With the collaboration of Marianne Baudler a . a .: Handbook of Preparative Inorganic Chemistry. 3rd, revised edition. Volume II, Ferdinand Enke, Stuttgart 1978, ISBN 3-432-87813-3 , p. 683.
- ↑ SiH3Br (H3SiBr lt) Crystal Structure , accessed December 11, 2016.

