Triaminotrinitrobenzene
| Structural formula | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
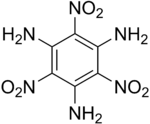
|
|||||||||||||||||||
| General | |||||||||||||||||||
| Surname | Triaminotrinitrobenzene | ||||||||||||||||||
| other names |
|
||||||||||||||||||
| Molecular formula | C 6 H 6 N 6 O 6 | ||||||||||||||||||
| Brief description |
yellow crystals |
||||||||||||||||||
| External identifiers / databases | |||||||||||||||||||
|
|||||||||||||||||||
| properties | |||||||||||||||||||
| Molar mass | 258.15 g mol −1 | ||||||||||||||||||
| Physical state |
firmly |
||||||||||||||||||
| density |
1.94 g cm −3 |
||||||||||||||||||
| Melting point |
different information: 330–480 ° C , often 448–449 ° C |
||||||||||||||||||
| safety instructions | |||||||||||||||||||
|
|||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||||||||
Triaminotrinitrobenzene ( TATB for short ) is an aromatic nitro compound used as an explosive .
history
TATB was first synthesized by Jackson and Wing in 1888. In the 1950s, the Naval Ordnance Laboratories (USA) recognized the stability and insensitivity of TATB and developed efficient, high-yield synthesis methods. In the 1970s, TATB was extensively investigated and characterized, especially in the USA.
presentation
There are various ways to synthesize triaminotrinitrobenzene:
- Nitration of 1,3,5-trichlorobenzene (C 6 H 3 Cl 3 ) with nitrating acid and subsequent treatment with ammonia in alcohol
- Reduction of trinitrotoluene (C 7 H 5 N 3 O 6 ) with hydrogen sulfide (H 2 S) in dioxane . This produces 6-amino-2,4-dinitrotoluene (C 7 H 7 N 3 O 4 ). This is then converted to pentanitroaniline with nitrating acid , then to TATB with ammonia in dichloromethane .
- In the VNS (Vicarious Nucleophilic Substitution) process, picramide ( trinitroaniline ) (C 6 H 4 N 4 O 6 ) is mixed with 1,1,1-trimethylhydrazinium iodide (TMHI) (C 3 H 11 N 2 I) in the presence of sodium methylate in dimethyl sulfoxide implemented directly to TATB. Instead of TMHI, the use of 1,1,1-triethylhydrazinium chloride or 4-amino-1,2,4-triazole is also described.
For purification, it is recrystallized from dimethyl sulfoxide and diphenyl ether .
properties
TATB forms yellow, triclinic crystals .
The stability of TATB is explained by intermolecular and intramolecular hydrogen bonds , which are formed around the benzene ring as a result of the alternating nitro and amino groups.
use
Despite its high manufacturing costs of around € 100 / kg, TATB is used as an explosive because the connection is stable and easy to handle on the one hand, but has a high explosive power on the other. The resistance of TATB to impact and heat is greater than that of any other known material with comparable energy density. TATB is not detonated even by the impact of a plane crash, fire, explosion or the impact of projectiles from small arms. Because of these properties, TATB is e.g. B. used in nuclear weapons or for insensitive ammunition (e.g. for ships and submarines).
Detonations in explosives based on TATB have a different course than detonations in sensitive explosives: They are quick, but not without delay and show a comparatively broad, three-dimensional reaction zone behind the detonation front. Such non-ideal detonations cannot be calculated with sufficient accuracy on the basis of simple theories. The use of TATB in weapon systems therefore requires a sophisticated understanding of the physics and chemistry of initiation and detonation.
- Detonation velocity at maximum density: 7619 m · s −1
- Detonation pressure : 25.9 G Pa
- Friction sensitivity :> 353 N
- Impact sensitivity : 50 J
TATB is also part of various mixed explosives, such as LX-17 (LX = Livermore explosive)
Legal provisions
The manufacture and processing of TATB are regulated in Germany by the Explosives Act.
Individual evidence
- ↑ a b c d e f g h i Entry on 1,3,5-triamino-2,4,6-trinitrobenzene. In: Römpp Online . Georg Thieme Verlag, accessed on June 1, 2014.
- ↑ There is not yet a harmonized classification for this substance . A labeling of 2,4,6-trinitrobenzene-1,3,5-triamine in the Classification and Labeling Inventory of the European Chemicals Agency (ECHA), accessed on May 10, 2018, is reproduced from a self-classification by the distributor .
- ↑ a b Klapötke, TM : Chemistry of High-Energy Materials , 2nd Edition, 2012 Walter de Gruyter GmbH & Co. KG, Berlin / Boston, ISBN 978-3-11-027358-8 , p. 118, (accessed from De Gruyter Online).
literature
- Philip F. Pagoria, Gregory S. Lee, Alexander R. Mitchell, Robert D. Schmidt: A Review of Energetic Materials Synthesis , in: Thermochimica Acta , 2002 , 384 (1-2), pp. 187-204; doi: 10.1016 / S0040-6031 (01) 00805-X .
- Roland Ionas Bialke: The demolition master's textbook. Collected knowledge about explosives from the clandestine hobby demolition master scene, Books on Demand GmbH, Norderstedt 2008, ISBN 978-3-8370-4729-5 .
See also
Web links
- Activities involving explosives (accessed on May 15, 2020)
- Contributions to the chemical-biological surface modification of nanodiamonds from detonation synthesis (accessed on May 15, 2020)
- Explosives (accessed May 15, 2020)
- Use of a Composition Comprising a Polymer and an Ionic Liquid (accessed May 15, 2020)

