Boron tribromide
| Structural formula | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
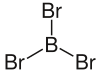
|
|||||||||||||||||||
| General | |||||||||||||||||||
| Surname | Boron tribromide | ||||||||||||||||||
| other names |
Tribromoborane |
||||||||||||||||||
| Molecular formula | BBr 3 | ||||||||||||||||||
| Brief description |
colorless liquid with a pungent odor |
||||||||||||||||||
| External identifiers / databases | |||||||||||||||||||
|
|||||||||||||||||||
| properties | |||||||||||||||||||
| Molar mass | 250.54 g mol −1 | ||||||||||||||||||
| Physical state |
liquid |
||||||||||||||||||
| density |
2.64 g cm −3 |
||||||||||||||||||
| Melting point |
−46 ° C |
||||||||||||||||||
| boiling point |
91 ° C |
||||||||||||||||||
| Vapor pressure |
72 h Pa (20 ° C) |
||||||||||||||||||
| solubility |
violent decomposition in water |
||||||||||||||||||
| Refractive index |
1.5312 (16 ° C) |
||||||||||||||||||
| safety instructions | |||||||||||||||||||
|
|||||||||||||||||||
| MAK |
Switzerland: 1 ml m −3 or 10 mg m −3 |
||||||||||||||||||
| Thermodynamic properties | |||||||||||||||||||
| ΔH f 0 |
−239.7 kJ / mol |
||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . Refractive index: Na-D line , 20 ° C | |||||||||||||||||||
Boron tribromide is a chemical compound consisting of the elements boron and bromine with the empirical formula BBr 3 .
presentation
Boron tribromide can be produced by implementing:
1. Boron trifluoride with aluminum bromide
2. Boron carbide with elemental bromine in the quartz tube at elevated temperature
3. Boron with elemental bromine in a quartz tube at around 700 ° C
4. Potassium tetrafluoroborate with anhydrous aluminum bromide
properties
Boron tribromide is a very toxic compound that smokes in moist air and is a liquid at room temperature . It is commercially available and is a strong Lewis acid .
Upon contact with water, a violent decomposition reaction occurs through hydrolysis to boric acid and hydrobromic acid :
use
Boron tribromide can be used for the cleavage of ethers under mild conditions, in particular for the gentle cleavage of alkylaryl ethers . In addition, it will also help the olefin - Polymerization and reactions Friedel-Crafts used. In the electronics industry it serves as Borlieferant in the doping of semiconductors .
Individual evidence
- ↑ a b c d e f data sheet boron tribromide (PDF) from Merck , accessed on January 19, 2011.
- ↑ David R. Lide (Ed.): CRC Handbook of Chemistry and Physics . 90th edition. (Internet version: 2010), CRC Press / Taylor and Francis, Boca Raton, FL, Index of Refraction of Inorganic Liquids, pp. 4-140.
- ↑ a b Entry on boron tribromide in the GESTIS substance database of the IFA , accessed on January 8, 2018(JavaScript required) .
- ↑ Entry on boron tribromide in the Classification and Labeling Inventory of the European Chemicals Agency (ECHA), accessed on February 1, 2016. Manufacturers or distributors can expand the harmonized classification and labeling .
- ↑ Schweizerische Unfallversicherungsanstalt (Suva): Limits - Current MAK and BAT values (search for 10294-33-4 or boron tribromide ), accessed on November 2, 2015.
- ↑ David R. Lide (Ed.): CRC Handbook of Chemistry and Physics . 90th edition. (Internet version: 2010), CRC Press / Taylor and Francis, Boca Raton, FL, Standard Thermodynamic Properties of Chemical Substances, pp. 5-5.
- ↑ a b Georg Brauer (Ed.), With the collaboration of Marianne Baudler u a .: Handbook of Preparative Inorganic Chemistry. 3rd, revised edition. Volume II, Ferdinand Enke, Stuttgart 1978, ISBN 3-432-87813-3 , p. 800.
- ↑ E. Lee Gamble: Boron chloride and bromide . In: Ludwig F. Audrieth (Ed.): Inorganic Syntheses . tape 3 . McGraw-Hill, Inc., 1950, pp. 27-30 (English).
- ^ Arnold F. Holleman, Egon Wiberg: Textbook of inorganic chemistry . Walter de Gruyter, 1995, ISBN 978-3-11-012641-9 , pp. 1890 ( limited preview in Google Book Search).
- ^ JJ Zuckerman: Inorganic Reactions and Methods, The Formation of Bonds to Hydrogen . John Wiley & Sons, 2009, ISBN 0-470-14536-6 , pp. 92 ( limited preview in Google Book search).
- ^ Wiley-VCH: Ullmann's Fine Chemicals, 3 Volume Set . John Wiley & Sons, 2014, ISBN 978-3-527-68359-8 , pp. 406 ( limited preview in Google Book search).






