Selenium tetrafluoride
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
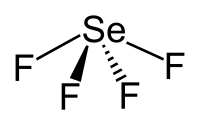
|
||||||||||||||||
| General | ||||||||||||||||
| Surname | Selenium tetrafluoride | |||||||||||||||
| other names |
Selenium (IV) fluoride |
|||||||||||||||
| Molecular formula | SeF 4 | |||||||||||||||
| Brief description |
colorless liquid |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 154.95 g mol −1 | |||||||||||||||
| Physical state |
liquid |
|||||||||||||||
| density |
2.72 g cm −3 |
|||||||||||||||
| Melting point |
−9.5 ° C |
|||||||||||||||
| boiling point |
106 ° C |
|||||||||||||||
| solubility |
violent decomposition in water |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||
Selenium tetrafluoride is an inorganic chemical compound of selenium from the group of fluorides .
Extraction and presentation
Selenium tetrafluoride can be obtained by reacting selenium with fluorine .
The preparation by halogenation of selenium dioxide with sulfur tetrafluoride , boron trifluoride or hydrogen fluoride is also possible .
properties
Selenium tetrafluoride is a colorless liquid that is miscible with sulfuric acid , ethanol , ether and iodine pentafluoride . It decomposes violently on contact with water. Metal fluorides such as sodium fluoride , potassium fluoride , rubidium fluoride , cesium fluoride and thallium (I) fluoride dissolve in selenium tetrafluoride with the formation of the metal-SeF 5 complexes. It also attacks glass.
Individual evidence
- ↑ a b c d e f g h Georg Brauer (Ed.), With the collaboration of Marianne Baudler u. a .: Handbook of Preparative Inorganic Chemistry. 3rd, revised edition. Volume I, Ferdinand Enke, Stuttgart 1975, ISBN 3-432-02328-6 , p. 195.
- ↑ This substance has either not yet been classified with regard to its hazardousness or a reliable and citable source has not yet been found.
- ^ AF Holleman , E. Wiberg , N. Wiberg : Textbook of Inorganic Chemistry . 101st edition. Walter de Gruyter, Berlin 1995, ISBN 3-11-012641-9 , p. 620.
- ^ Dale L. Perry: Handbook of Inorganic Compounds, Second Edition . Taylor & Francis US, 2011, ISBN 1-4398-1462-7 , pp. 360 ( limited preview in Google Book Search).
