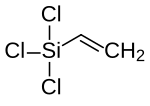
Trichloro (vinyl) silane
| Structural formula | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|||||||||||||||||||
| General | |||||||||||||||||||
| Surname | Trichloro (vinyl) silane | ||||||||||||||||||
| other names |
|
||||||||||||||||||
| Molecular formula | C 2 H 3 Cl 3 Si | ||||||||||||||||||
| Brief description |
colorless liquid with a pungent odor |
||||||||||||||||||
| External identifiers / databases | |||||||||||||||||||
|
|||||||||||||||||||
| properties | |||||||||||||||||||
| Molar mass | 161.49 g mol −1 | ||||||||||||||||||
| Physical state |
liquid |
||||||||||||||||||
| density |
1.27 g cm −3 |
||||||||||||||||||
| Melting point |
−95 ° C |
||||||||||||||||||
| boiling point |
91 ° C |
||||||||||||||||||
| Vapor pressure |
74 hPa (20 ° C) |
||||||||||||||||||
| solubility |
|
||||||||||||||||||
| Refractive index |
1.436 (20 ° C) |
||||||||||||||||||
| safety instructions | |||||||||||||||||||
|
|||||||||||||||||||
| Toxicological data | |||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . Refractive index: Na-D line , 20 ° C | |||||||||||||||||||
Trichloro (vinyl) silane is a chemical compound from the group of organosilicon compounds .
Extraction and presentation
Trichlor (vinyl) silane can be obtained by reacting trichlorosilane with ethyne or vinyl chloride .
properties
Trichloro (vinyl) silane is a highly flammable, volatile, colorless liquid with a pungent odor that decomposes in water, releasing hydrogen chloride and hydrogen . The resulting aqueous solution reacts strongly acidic.
use
Trichloro (vinyl) silane is used as a monomer for copolymers in water-repellent materials, electrical insulating resins and high-temperature resins for paints, as well as an intermediate product for silicones and as an adhesion promoter in adhesives (e.g. for glass fiber reinforced plastics).
safety instructions
The vapors of trichloro (vinyl) silane can form an explosive mixture with air ( flash point 10 ° C, ignition temperature 280 ° C).
Individual evidence
- ↑ a b c d e f g h i j k l m Entry on vinyltrichlorosilane in the GESTIS substance database of the IFA , accessed on November 3, 2018(JavaScript required) .
- ↑ David R. Lide: CRC Handbook of Chemistry and Physics A Ready-reference Book of Chemical and Physical Data . CRC Press, 1995, ISBN 978-0-8493-0595-5 , pp. 542 ( limited preview in Google Book search).
- ↑ Data sheet Trichlorovinylsilane, 97% from Sigma-Aldrich , accessed on November 4, 2018 ( PDF ).
- ↑ a b Entry on trichloro (vinyl) silane in the Hazardous Substances Data Bank , accessed on November 4, 2018.
- ↑ Google Patents: EP0456901B1 - Process for the preparation of vinyltrichlorosilane - Google Patents , accessed November 4, 2018
- ↑ Manfred Flemming, Gerhard Ziegmann, Siegfried Roth: Fiber composite construction methods fibers and matrices . Springer-Verlag, 2013, ISBN 978-3-642-57776-5 , p. 176 ( limited preview in Google Book search).


