Homochirality
The term homochirality is used to describe a group of molecules that share the same sense of chirality . This means that similar substituents are arranged in the same way around a central atom, the center of chirality . Homochirality is mainly observed in living organisms. For example, all naturally occurring amino acids have the L form . Most of the biologically relevant sugars are also in the D form. The mirror-image enantiomers of these molecules are usually biologically inactive and in some cases even toxic for organisms. The origin of this phenomenon is not fully understood.
One theory is that homochirality lowers the entropy barrier when large, organized molecules form. It has been experimentally proven that amino acids form large aggregates in greater yield when they are enantiomerically pure and not as a racemate .
Homochirality should develop in three steps: The breaking of the mirror symmetry creates a tiny, enantiomeric imbalance, this is the key to homochirality. The Chiral amplification leads to an enantiomeric enrichment. Chirality transfer allows the transfer of chirality from one group of molecules to another.
Break of the mirror symmetry
The most difficult question to answer is how an enantiomeric imbalance is originally created. Some scientists support the theory of an extraterrestrial origin, which is based on the discovery of an enantiomeric excess in various, especially alpha-methylated amino acids in the Murchison meteorite . There is evidence for the existence of circularly polarized light in space, which is believed to have been caused by Mie scattering on aligned interstellar dust particles. The interaction of this circularly polarized light with racemic organic compounds could have triggered the formation of a small enantiomeric excess. Under simulated space conditions, it was possible to induce an enantiomeric excess of 2.6% in the amino acid leucine with the help of circularly polarized synchrotron radiation .
A classic study includes a laboratory experiment. If sodium chlorate crystallizes out of water and the crystals obtained are examined in a polarimeter , it is found that every crystal is chiral and consists exclusively of the L or D form. In a normal experiment, the respective amounts of L and D crystals are the same when statistical effects are corrected. However, if the sodium chlorate solution is stirred during the crystallization process, only L or D crystals are obtained. Out of 32 crystallization experiments, 14 experiments yielded D crystals and the remaining 18 experiments yielded L crystals. There is no clear explanation for this break in symmetry, but it is believed to be related to the autocatalysis that occurs in the nucleation process .
Chiral amplification
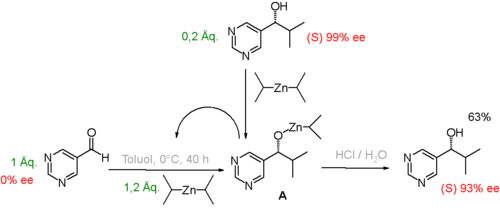
In laboratory experiments it could be demonstrated how in certain autocatalytic reaction systems the presence of small amounts of the reaction product with enantiomeric excess at the beginning of the reaction can lead to a much larger enantiomeric excess at the end of the reaction. In a seminal study was pyrimidine-5- carbaldehyde with (Fig. 1) diisopropylzinc to the corresponding pyrimidyl - alcohol alkylated. The initial reaction product is also an efficient catalyst, so the reaction is autocatalytic. The presence of only 0.2 equivalents of the ( S ) - enantiomer of the alcohol formed at the start of the reaction is sufficient to increase the enantiomeric excess to 93%.
Another study concerns the proline -catalyzed aminoxylation of propanal with nitrosobenzene (Fig. 2). In this system, the presence of an enantiomerically enriched catalyst also leads to one of the two possible optical isomers.
Serine octamer clusters are another example. These clusters, which consist of eight serine molecules, show an unusual homochiral preference in mass spectrometry , but there is no evidence that such clusters exist under non-ionizing conditions. In addition, the behavior of the amino acids at phase boundaries is far more relevant from a prebiotic point of view. The observation that the partial sublimation of a 10% enantiomerically enriched sample of leucine leads to an enrichment of up to 82% in the sublimate shows that an enantiomeric enrichment of amino acids could also take place in space. Partial sublimation processes can take place on the surface of meteors , where large temperature variations occur. This discovery could have consequences for the development of the Mars Organics Detector , the launch of which is scheduled for 2013. This probe is supposed to extract small amounts of amino acids from the surface of Mars using a sublimation technique.
A high amplification of the enantiomeric excess of sugars is also observed in the amino acid-catalyzed, asymmetric formation of hydrocarbons .
Chirality transfer
Many strategies in stereoselective synthesis are based on transfer of chirality. The so-called organocatalysis of organic reactions by proline, for example in Mannich reactions, is particularly important .
Resolution of racemic amino acids
There is no theory that explains the correlation between L- amino acids. If, for example , one compares alanine , which has a small methyl group , and phenylalanine, which has a large benzyl group , the simple question arises, in which aspect L -alanine resembles L -phenylalanine more than D -phenylalanine and what kind of mechanism is the selection of all L -amino acids. It would just as well be possible for natural alanine to have an L configuration, whereas natural phenylalanine has a D configuration.
In 2004 it was reported that an excess of racemic asparagine (= DL -asparagine), which spontaneously forms crystals of the pure isomers during recrystallization, leads to the resolution of a racemic amino acid that is also in solution, such as arginine (Arg), aspartic acid (Asp), glutamine (Gln), histidine (His), leucine (Leu), methionine (Met), phenylalanine (Phe), serine (Ser), valine (Val), tyrosine (Tyr) and tryptophan (Trp) . The enantiomeric excess of these second amino acids correlates almost linearly with the corresponding value of the inducing amino acid, i.e. asparagine. When a mixture of 12 DL -amino acids (Ala, Asp, Arg, Glu, Gln, His, Leu, Met, Ser, Val, Phe and Tyr) and an excess DL -Asn were recrystallized, it was found that all amino acids were the same Configuration like Asn preferably with crystallized out. It depended on chance whether the enrichment took place with the L or D asparagine. However, as soon as the selection was made, the coexisting amino acid with the same configuration at the α-carbon atom was preferred, which can be explained by the thermodynamic stability of the crystal structures formed. The highest reported enantiomeric excess was 100%. Based on these results, it is assumed that a mixture of racemic amino acids can be spontaneously and effectively resolved even if the asymmetric synthesis of a single amino acid cannot take place without the aid of an optically active molecule.
This is the first study to explain the emergence of chirality from racemic amino acids with experimental evidence.
history
The term homochirality was introduced by Kelvin in 1904, the same year he published his 1884 Baltimore Lecture. Recently, however, the term homochiral has also been used in the same sense as enantiomerically pure . This is allowed in some journals, but is not recommended. In these journals, however, the term means that a single of two optical isomers is preferred in a process or system.
See also
Individual evidence
- ^ RR Julian, S. Myung, DE Clemmer: Do Homochiral Aggregates Have an Entropic Advantage? In: J. Phys. Chem. B 2005 , 109 , 440-444; doi : 10.1021 / jp046478x .
- ↑ UJ Meierhenrich: Asymmetric vacuum UV photolysis of the amino acid leucine in the solid phase . In: Angewandte Chemie 2005 , 117 , 5774-5779; doi : 10.1002 / anie.200501311 .
- ↑ DK Kondepudi, RJ Kaufman, S. Singh: Chiral Symmetry Breaking in Sodium Chlorate Crystallization . In: Science 1990 , 250 , 975-976.
- ↑ T. Shibata, H. Morioka, T. Hayase, K. Choji, K. Soai: Highly Enantioselective Catalytic Asymmetric Automultiplication of Chiral Pyrimidyl Alcohol . In: J. Am. Chem. Soc. 1996 , 118 , 471-472. doi : 10.1021 / ja953066g
- ↑ PM Suju, H. Iwamura, DG Blackmond: Amplification of Enantiomeric Excess in a Proline-Mediated Reaction . In. Angew. Chem. Int. Ed. 2004 , 43 , 3317-3321.
- ^ RG Cooks, D. Zhang and KJ Koch: Chiroselective Self-Directed Octamerization of Serine: Implications for Homochirogenesis . In. Anal. Chem. 2001 , 73 , 3646-3655; doi : 10.1021 / ac010284l .
- ↑ SC Nanita, RG Cooks: Serine octamers: clustering, reactions and effects on the homochirality of biomolecules . In: Angew. Chem. 2006 , 118 , 568-583. doi : 10.1002 / anie.200501328
- ^ DG Blackmond, M. Klussmann: Spoilt for choice: assessing phase behavior models for the evolution of homochirality . In: Chem. Commun. 2007 , 3990-3996; doi : 10.1039 / b709314b .
- ↑ SP Fletcher, R: BC Jagt, BL Feringa: An astrophysically relevant mechanism for amino acid enantiomer enrichment . In: Chem. Commun. 2007 , 2578-2580; doi : 10.1039 / b702882b .
- ↑ a b A. Córdova, M. Engqvist, I. Ibrahem, J. Casas, H. Sundén: Plausible origins of homochirality in the amino acid catalyzed neogenesis of carbohydrates . In: Chem. Commun. 2005 , 15 , 2047-2049.
- ↑ a b S. Kojo, H. Uchino, M. Yoshimura, K. Tanaka: Racemic D, L-asparagine causes enantiomeric excess of other coexisting racemic D, L-amino acids during recrystallization: a hypothesis accounting for the origin of L- amino acids in the biosphere , in: Chem. Commun. 2004 , 2146-2147; doi : 10.1039 / b409941a .
- ^ DG Morris: Stereochemistry . Royal Society of Chemistry, Cambridge 2001, p. 30.
Web links
- On the Genesis of Homochirality . A. Maureen Rouhi, Chemical & Engineering News, June 17, 2004.
- Scienceweek digest 1998
- How left-handed amino acids got ahead: a demonstration of the evolution of biological homochirality in the lab . Press release, Imperial College London, 2004.