Prednicarbate
| Structural formula | ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
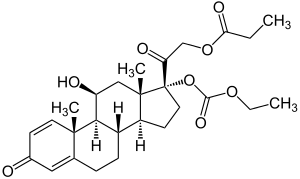
|
||||||||||||||||||||||
| General | ||||||||||||||||||||||
| Non-proprietary name | Prednicarbate | |||||||||||||||||||||
| other names | ||||||||||||||||||||||
| Molecular formula | C 27 H 36 O 8 | |||||||||||||||||||||
| Brief description |
white to almost white, crystalline powder |
|||||||||||||||||||||
| External identifiers / databases | ||||||||||||||||||||||
|
||||||||||||||||||||||
| Drug information | ||||||||||||||||||||||
| ATC code | ||||||||||||||||||||||
| Drug class |
Glucocorticoid |
|||||||||||||||||||||
| properties | ||||||||||||||||||||||
| Molar mass | 488.6 g · mol -1 | |||||||||||||||||||||
| Physical state |
firmly |
|||||||||||||||||||||
| density |
1.26 g cm −3 |
|||||||||||||||||||||
| Melting point |
110-122 ° C |
|||||||||||||||||||||
| boiling point |
640 ° C |
|||||||||||||||||||||
| solubility |
|
|||||||||||||||||||||
| safety instructions | ||||||||||||||||||||||
|
||||||||||||||||||||||
| Toxicological data | ||||||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||||||||
Prednicarbate is a drug from the group of topically applied glucocorticoids that is used to treat various skin diseases. It is a moderately potent glucocorticoid (" Class II").
Clinical information
Application areas (indications)
Prednicarbate is used for acute and chronic inflammatory skin diseases such as B. chronic eczema . If used on the face, care should be taken not to get the drug in the eyes. The application concentration is usually 0.25%.
Type and duration of application
As with all corticoids , long-term use should be avoided if possible, as this can lead to skin atrophy , telangiectasia , striae , a special form of acne , perioral dermatitis and hypertrichosis . A maximum duration of use of 2 weeks is recommended.
Contraindications (contraindications)
Extensive use of corticoids or those under an occlusive dressing should generally be avoided in order to avoid increased absorption of the active ingredient through the skin and thus the risk of systemic, i.e. H. prevent adverse drug reactions affecting the entire body. Further contraindications are hypersensitivity to the active ingredient and use on or in the eye.
Drug interactions
No interactions are known for prednicarbate.
Use during pregnancy and breastfeeding
Extensive application to more than 30% of the body surface is not indicated, as is application to the breast area in nursing mothers. In general, the indication for the use of prednicarbate should be strictly set during pregnancy and its use limited to a small area of skin.
Special patient groups (diabetics, kidney patients)
In the case of infants, particular care must be taken to establish the indication, as the absorption of the active ingredient is increased in them.
Adverse effects (side effects)
The side effects of prednicarbate are the same as other topical glucocorticoids; see Glucocorticoids: Side Effects .
Pharmacological properties
Mechanism of action (pharmacodynamics)
Prednicarbate intervenes in the arachidonic acid metabolism , from which a decrease in the production of inflammatory mediators such as z. B. prostaglandins or leukotrienes and thus an anti-inflammatory effect results.
There is also an antiallergic effect; this is based on influencing the lymphocytes . In particular, the substance acts on B and T lymphocytes, whose activity and number are reduced.
Finally, prednicarbate also has an antiproliferative effect by reducing the metabolism and DNA synthesis rate. Both lead to a slowdown in cell division. This property of the glucocorticoids explains their effectiveness in psoriasis .
Absorption and distribution in the body (pharmacokinetics)
Prednicarbate is in the skin of prednisolone 17-ethyl carbonate reacted . This has a much higher affinity for the corresponding receptors. Gradually it breaks down into prednisolone . The prednicarbate does not enter the bloodstream if it is used correctly.
toxicology
Prednicarbate has been shown to be "non-irritating to the skin" in animal experiments , and is also neither phototoxic nor photosensitizing .
Trade names
Prednitop, dermatop
See also
Glucocorticoids , prednisolone , skin disease
Individual evidence
- ↑ European Pharmacopoeia, Deutscher Apotheker Verlag Stuttgart, 6th edition, 2008, pp -3738-3739, ISBN 978-3-7692-3962-1 .
- ↑ a b c d Fagron: Safety data sheet Prednicarbate , accessed on December 21, 2019
- ↑ GWA Milne: Ashgate Handbook of Endocrine Agents and Steroids . Routledge, 2017, ISBN 978-1-351-74347-1 ( limited preview in Google Book Search).
- ↑ a b c Cayman Chemical: Prednicarbate (73771-04-7) , accessed December 21, 2019
- ↑ Glucorticoide topical In: Altmeyers Enzyklopädie Dermatologie online.
- ↑ a b c d e f g h Specialist information Dermatop ( Memento from May 13, 2018 in the Internet Archive ), August 2017.
- ↑ a b Aktories, Förstermann, Hofmann, Starke: General and special pharmacology and toxicology, Urban & Schwarzenberg, Munich 2009, ISBN 978-3-437-42522-6 .
- ↑ Red List 2013.