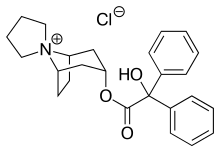
Trospium chloride
| Structural formula | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|||||||||||||||||||
| General | |||||||||||||||||||
| Non-proprietary name | Trospium chloride | ||||||||||||||||||
| other names |
|
||||||||||||||||||
| Molecular formula | C 25 H 30 ClNO 3 | ||||||||||||||||||
| Brief description |
white to almost white crystalline powder |
||||||||||||||||||
| External identifiers / databases | |||||||||||||||||||
|
|||||||||||||||||||
| Drug information | |||||||||||||||||||
| ATC code | |||||||||||||||||||
| Drug class | |||||||||||||||||||
| Mechanism of action | |||||||||||||||||||
| properties | |||||||||||||||||||
| Molar mass | |||||||||||||||||||
| Physical state |
firmly |
||||||||||||||||||
| Melting point |
255–257 ° C (decomposition) |
||||||||||||||||||
| solubility |
|
||||||||||||||||||
| safety instructions | |||||||||||||||||||
|
|||||||||||||||||||
| Toxicological data | |||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||||||||
Trospium is a drug from the group of tropane derivatives and belongs to the synthetic anticholinergics . Chemically, it is a quaternary 3 α - nortropanol , which is esterified with benzilic acid. It is used to treat urge and reflex incontinence .
Clinical information
The pharmacological effect comes about through competitive blocking of the effect of acetylcholine on the muscarinic acetylcholine receptors . This leads to an inhibition of the parasympathetic influence on the smooth muscles , the heart muscle , glands and ganglia of the peripheral nervous system . As a quaternary ammonium ion, trospium is poorly absorbed from the gastrointestinal tract and can hardly penetrate the blood-brain barrier , which is why it has hardly any effects on the central nervous system. In addition, trospium chloride is recognized and discharged by special transporter systems located at the blood-brain barrier.
Pharmacological properties
Contraindications
Contraindications for use are untreated glaucoma , obstructive prostate hyperplasia , intestinal obstruction , pyloric stenosis and other narrowing in the gastrointestinal tract, as well as cardiac arrhythmias with increased heart rate.
Side effects
In principle, the side effects also correspond to those of other anticholinergics; reddening of the skin, bloating , constipation , visual disturbances, reduced perspiration , rapid heartbeat ( tachycardia ), accommodation disorders and a dry mouth may occur.
Trade names
Inkontan (A), Rekont (A), Spasmex (D), Spasmolyt (D, A), Spasmo-Rhoival (D), Spasmo-Urgenin Neo (CH), Spasmo-Urgenin TC (D), Uraplex (A), Urivesc (D, A)
Spasmo Urgenin (A)
literature
- Ernst Mutschler et al .: Mutschler - drug effects textbook of pharmacology and toxicology . 9th edition. Scientific Verlagsgesellschaft, Stuttgart 2008, ISBN 978-3-8047-1952-1 .
- A. Wiedemann, PA Schwantes: Antimuscarinic drugs for the treatment of overactive bladder: are they really all the same? - A comparative review of data pertaining to pharmacological and physiological aspects . Eur J Ger 9 (1): 29-42, 2009.
Web links
- Trospium on www.urologielehrbuch.de
Individual evidence
- ↑ a b c d European Pharmacopoeia Commission (Ed.): European Pharmacopoeia 6th edition. 6.0-6.2, 2008.
- ↑ a b entry on trospium chloride. In: Römpp Online . Georg Thieme Verlag, accessed on May 30, 2014.
- ↑ a b Entry on trospium chloride in the GESTIS substance database of the IFA , accessed on January 8, 2020(JavaScript required) .
- ^ J. Geyer, O. Gavrilova, E. Petzinger: The role of P-glycoprotein in limiting brain penetration of the peripherally acting anticholinergic overactive bladder drug trospium chloride in Drug Metabolism and Disposition 37 (2009) 1371-1374. doi : 10.1124 / dmd.109.027144
