Tizanidine
| Structural formula | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
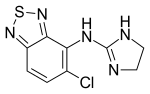
|
|||||||||||||
| General | |||||||||||||
| Surname | Tizanidine | ||||||||||||
| other names |
4-chloro- N - (4,5-dihydro-1 H -imidazol-2-yl) -8-thia-7,9-diazabicyclo [4.3.0] nona-2,4,6,9-tetraen-5 -amin |
||||||||||||
| Molecular formula |
|
||||||||||||
| External identifiers / databases | |||||||||||||
|
|||||||||||||
| Drug information | |||||||||||||
| ATC code | |||||||||||||
| Drug class | |||||||||||||
| Mechanism of action | |||||||||||||
| properties | |||||||||||||
| Molar mass | 253.71 g mol −1 | ||||||||||||
| Physical state |
firmly |
||||||||||||
| Melting point |
221-223 ° C |
||||||||||||
| solubility |
> 20 mg / ml in water |
||||||||||||
| safety instructions | |||||||||||||
|
|||||||||||||
| Toxicological data | |||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||
Tizanidine ( trade name Sirdalud ® , manufactured by Novartis ) is primarily an α 2 -adrenoceptor - agonist which the group of centrally acting muscle relaxants heard with main target in the spinal cord.
Clinical information
Application areas (indications)
The drug is used to treat:
- painful, peripheral muscle tension . This includes functional and static problems of the spine such as thoracic , lumbar and cervical syndromes ,
- after operations, for example because of a herniated disc ( disc herniation ) or degenerative disease of the hip joints ( hip arthrosis ).
- Muscle spasms that can result from multiple sclerosis or spinal cord injuries .
Adverse effects (side effects)
The side effects of tizanidine are diverse and often include dry mouth, fatigue, asthenia (weakness, drowsiness), dizziness , bradycardia (slow pulse) and a slight drop in blood pressure , rarely hallucinations , insomnia, sleep disorders, nausea, gastrointestinal disorders, and muscle weakness temporary increase in transaminases .
Drug interactions
In vitro studies of cytochrome P450 isoenzymes using human liver microsomes indicate that neither tizanidine, nor the major metabolite , is likely to affect the metabolism of other agents metabolized by cytochrome P450 isoenzymes.
- Paracetamol : Tizanidine delays T max of paracetamol by 16 minutes. Paracetamoldid notaffect the pharmacokinetics of tizanidine.
- Alcohol : Alcohol increasedtizanidine's AUC by approximately 20% and also its C max by approximately 15%. This has been linked to an increase in the side effects of tizanidine. The central nervous system suppressive effects of tizanidine and alcohol are additive.
- Fluvoxamine : Clinically significant hypotension (decreases in systolic and diastolic blood pressures ) has been reported with concomitant use of fluvoxamine after single 4 mg doses.
- Oral contraceptives : According to the results of a clinical study, oral contraceptives can possibly reduce the metabolism of tizanidine by inhibiting CYP1A2 isoenzymes and thus contribute to significantly increased plasma levels.
Caution is advised when co-administering tizanidine with other inhibitors of CYP1A2 such as B. Antiarrhythmics ( amiodarone , mexiletine , propafenone ), cimetidine , fluoroquinolones ( ciprofloxacin , norfloxacin ), rofecoxib , oral contraceptives and ticlopidine .
Pharmacological properties
Absorption and distribution in the body (pharmacokinetics)
After oral administration, tizanidine is rapidly absorbed . The maximum plasma concentration is reached after approximately 1 hour . The absolute bioavailability is an average of 34% . After intravenous administration the mean volume of distribution at steady state is 160 liters. The plasma protein binding is low and is 30%.
The drug is rapidly and extensively metabolized by the liver . Tizanidine is mainly broken down by the cytochrome P450 enzyme system (CYP 1A2). The metabolites are inactive and are mainly (70%) excreted renally . The unchanged substance is only excreted in small amounts (approx. 2.7%) via the kidneys. The average plasma half-life of the unchanged substance is 2 to 4 hours.
Individual evidence
- ↑ a b c Entry on tizanidine. In: Römpp Online . Georg Thieme Verlag, accessed on October 1, 2014.
- ↑ a b c data sheet Tizanidine hydrochloride from Sigma-Aldrich , accessed on April 24, 2011 ( PDF ).
- ↑ technical information Sirdalud®, Red List of 2008.
- ↑ Rote-Hand-Brief zu Sirdalud ( Memento of October 22, 2007 in the Internet Archive ).
- ↑ Granfors MT, Backman JT, Laitila J, Neuvonen PJ. Oral contraceptives containing ethinyl estradiol and gestodene markedly increase plasma concentrations and effects of tizanidine by inhibiting cytochrome P450 1A2. Clin Pharmacol Ther . 2005 Oct; 78 (4): 400-11, PMID 16198659 .
- ↑ Tizanidine hydrochloride at DailyMed .
- ↑ SPC for Sirdalud in drug compendium of Switzerland - 22nd of May 2008.