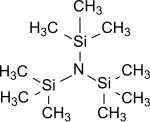
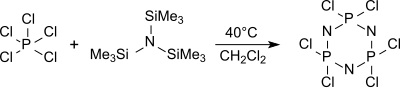Tris (trimethylsilyl) amine
| Structural formula | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||
| General | ||||||||||||||||
| Surname | Tris (trimethylsilyl) amine | |||||||||||||||
| other names |
|
|||||||||||||||
| Molecular formula | C 9 H 27 NSi 3 | |||||||||||||||
| Brief description |
crystalline, colorless or waxy solid |
|||||||||||||||
| External identifiers / databases | ||||||||||||||||
|
||||||||||||||||
| properties | ||||||||||||||||
| Molar mass | 233.57 g mol −1 | |||||||||||||||
| Physical state |
firmly |
|||||||||||||||
| density |
0.8635 g cm −3 at 20 ° C |
|||||||||||||||
| Melting point |
|
|||||||||||||||
| boiling point |
|
|||||||||||||||
| solubility |
Easily soluble in non-polar organic solvents, such as. B. benzene , carbon tetrachloride , diethyl ether , 1,4-dioxane , petroleum ether or tetrahydrofuran |
|||||||||||||||
| Refractive index |
1.4545 (20 ° C) |
|||||||||||||||
| safety instructions | ||||||||||||||||
|
||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . Refractive index: Na-D line , 20 ° C | ||||||||||||||||
Tris (trimethylsilyl) amine is the simplest representative of tris (trialkylsilyl) amines of the general formula (R 3 Si) 3 N, in which all three hydrogen atoms of ammonia have been replaced by trimethylsilyl groups (-Si (CH 3 ) 3 ). Tris (trimethylsilyl) amine is a stable intermediate in the chemical fixation of nitrogen , i.e. H. the introduction of atmospheric nitrogen N 2 into organic substrates under normal conditions , which has been the center of great scientific interest for years.
Occurrence and representation
Early attempts to prepare tris (trimethylsilyl) amine from the starting compounds ammonia and trimethylchlorosilane (TMS-Cl) were unsuccessful even at temperatures of 500 ° C and in the presence of the base pyridine . The reaction of ammonia and trimethylchlorosilane stops at the level of the doubly silylated product bis (dimethylsilyl) amine, usually referred to as hexamethyldisilazane (HMDS).
Nonamethyltrisilazane is formed in the reaction of the sodium salt of hexamethyldisilazane - from HMDS and sodium amide or from with HMDS, sodium and styrene - with trimethylchlorosilane in 80% yield.
The lithium salt of HMDS - from HMDS and n -butyllithium or from HMDS and phenyllithium - only reacts with trimethylchlorosilane to form tris (trimethylsilyl) amine in yields of 50–60%.
The reaction of lithium nitride with trimethylchlorosilane in THF can be carried out as a one-pot reaction with a 72% yield .
properties
Tris (trimethylsilyl) amine is a colorless, crystalline or waxy solid that is stable to water and alkalis , but decomposes when exposed to alcohols or acids, splitting the Si-N bond and forming ammonia.
Applications
Tris (trimethylsilyl) amine as a synthetic building block
Of antimony trichloride and tris (trimethylsilyl) amine, a complex Nitridoantimon heterojunction is formed almost quantitatively at -60 ° C cubane structure .
Ketones can be trifluoromethylated in the presence of P4-t-Bu and nonamethyltrisilazane under mild conditions in yields of up to 84% with the very inert fluoroform (HCF 3 , HFC-23).
From tris (trimethylsilyl) amine and phosphorus pentachloride in hexane, the monomer trichloro (trimethylsilyl) phosphoraneimine Cl 3 P = NSiMe 3 is formed at 0 ° C ,
which can be polymerized to linear polydichlorophosphazenes with defined molecular weights and polydispersities .
In boiling dichloromethane (approx. 40 ° C) the cyclic trimer (NPCl 2 ) 3 hexachlorocyclotriphosphane is formed from tris (trimethylsilyl) amine and phosphorus pentachloride, along with other oligomers, mainly (76%) , from which, when heated to over 250 ° C, high-molecular, but poorly defined polydichlorophosphazenes are obtained.
Nitrogen trifluoride NF 3 , which u. a. is used for plasma etching of silicon wafers , is accessible from tris (trimethylsilyl) amine and fluorine gas at −40 ° C in acetonitrile , whereby the formation of nitrogen N 2 and tetrafluorohydrazine N 2 F 4 , which are undesirable by-products in the standard synthesis from ammonia or ammonium fluoride incurred, is suppressed.
Tris (trimethylsilyl) amine as an intermediate in the chemical fixation of nitrogen
The technical nitrogen fixation was realized by the Haber-Bosch process , in which nitrogen is converted into ammonia by reductive protonation in the presence of iron catalysts under high pressures (> 150 bar) and temperatures (> 400 ° C).
In chemical nitrogen fixation, i. H. the conversion of atmospheric nitrogen under normal conditions into reactive starting materials for chemical syntheses - usually also ammonia - tris (trimethylsilyl) amine plays an important role in the so-called reductive silylation , as it is hydrolyzed to ammonia with water.
As early as 1895 it was observed that metallic lithium reacts with nitrogen to form lithium nitride even at room temperature. In 1972 K. Shiina observed that lithium (as an electron donor ) in the presence of chromium (III) chloride as a catalyst at room temperature with the nitrogen used for inerting forms small amounts of tris (trimethylsilyl) amine with a dark color with trimethylsilyl chloride.
More recently, sodium has been preferred as an electron donor instead of lithium for the reductive silylation of N 2 for nitrogen fixation under normal conditions , as well as molybdenum and iron compounds, such as. B. pentacarbonyl iron or ferrocenes are used as catalysts, with up to 34 equivalents of N (Me 3 Si) 3 per iron atom in the catalyst.
With a molybdenum-ferrocene complex as a catalyst has catalytic productivity (engl. Turnover number, TON ) can be achieved of up to 226.
Despite intensive research, the catalytic productivity of the catalysts developed to date for chemical nitrogen fixation is still orders of magnitude lower than z. B. the modern polymerization catalysts of the metallocene type or enzymes .
Individual evidence
- ↑ a b c d e data sheet Tris (trimethylsilyl) amine 98% from Sigma-Aldrich , accessed on December 28, 2016 ( PDF ).
- ↑ a b Data sheet nonamethyltrisilazane from AlfaAesar, accessed on December 28, 2016 ( PDF )(JavaScript required) .
- ↑ a b c d e C.R. Krüger, H. Niederprüm, M. Schmidt, O. Scherer: Sodium Bis (trimethylsilyl) amide and Tris (trimethylsilyl) amine, in Inorganic Syntheses . Ed .: HF Holtzlow. tape 8 . John Wiley & Sons, Inc., Hoboken, NJ, USA 1966, ISBN 978-0-470-13167-1 , pp. 15-19 , doi : 10.1002 / 9780470132395.ch5 .
- ↑ a b c d e f J. Goubeau, J. Jiminéz-Barberá: Tris (trimethylsilyl) amine . In: ZAAC . tape 303 , no. 5-6 , 1960, pp. 217-226 , doi : 10.1002 / zaac.19603030502 .
- ↑ a b E.H. Amonoo-Neizer, RA Shaw, DO Skovlin, BC Smith, JW Rosenthal, WL Jolly: Lithium Bis (trimethylsilyl) amide and Tris (trimethylsilyl) amine, in Inorganic Syntheses . Ed .: HF Holtzlow. tape 8 . John Wiley & Sons, Inc., Hoboken, NJ, USA 1966, ISBN 978-0-470-13167-1 , pp. 19-22 , doi : 10.1002 / 9780470132395.ch5 .
- ↑ a b W.L. Lehn: Preparation of tris (trimethylsilyl) and tris (trimethylstannyl) amines . In: J. Am. Chem. Soc. tape 86 , no. 2 , 1964, p. 305 , doi : 10.1021 / ja01056a057 .
- ↑ a b K. Shiina: Reductive silylation of molecular nitrogen via fixation to tris (trimethylsilyl) amine . In: J. Am. Chem. Soc. tape 94 , no. 26 , 1972, p. 9266-9267 , doi : 10.1021 / ja00781a068 .
- ↑ a b K.C. MacLeod, PL Holland: Recent developments in the homogeneous reduction of dinitrogen by molybdenum and iron . In: Nature Chemistry . tape 5 , 2013, p. 559-565 , doi : 10.1038 / nchem . 1620 .
- ↑ WI Dzik: silylation of dinitrogen catalyzed by hydridodinitrogen (triphenylphosphine) cobalt (I) . In: Inorganics . tape 4 , no. 3 , 2016, p. 21 , doi : 10.3390 / inorganics4030021 .
- ^ RO Sauer: Derivatives of the methylchlorosilanes. I. Trimethylsilanol and its simple ethers . In: J. Am. Chem. Soc. tape 66 , no. 10 , 1944, pp. 1707-1710 , doi : 10.1021 / ja01238a030 .
- ^ RO Sauer, RH Hasek: Derivatives of the methylchlorosilanes. IV. Amines . In: J. Am. Chem. Soc. tape 68 , no. 2 , 1946, p. 241–244 , doi : 10.1021 / ja01206a028 .
- ↑ a b U. Wannagat, H. Niederprüm: Contributions to the chemistry of silicon-nitrogen compounds, XIII. Silyl-substituted alkali amides . In: Chem. Ber. tape 94 , no. 6 , 1961, pp. 1540-1547 , doi : 10.1002 / cber.19610940618 .
- ↑ U. Wannagat, H. Niederprüm: triply silylated amines . In: ZAAC . tape 308 , no. 1-6 , 1961, pp. 337-351 , doi : 10.1002 / zaac.19613080135 .
- ↑ M. Rhiel, F. Weller, J. Pebler, K. Dehnicke: [SbN (SbCl) 3 (NSbCl 2 ) (NSiMe 3 ) 3 · SbCl 3 ], an unusual nitridoantimony complex with a heterocubane structure . In: Angew. Chem. Band 106 , no. 5 , 1994, pp. 599-600 , doi : 10.1002 / anie.19941060519 .
- ↑ S. Okusu, K. Hirano, E. Tokunaga, N. Shibata: Organocatalyzed trifluoromethylation of ketones and sulfonyl fluorides by fluoroform under a super base system . In: ChemistryOpen . tape 4 , 2015, p. 581-585 , doi : 10.1002 / open.201500160 .
- ↑ Patent US5698664 : Synthesis of polyphosphazenes with controlled molecular weight and polydispersity. Filed April 26, 1995 , published December 16, 1997 , Applicant: The Penn State Research Foundation, University of Toronto, Inventor: HR Allcock, CT Morrissey, I. Manners, CH Honeyman.
- ↑ Patent US8163262B1 : Method for production of nitrogen fluoride from trimethylsilylamines. Registered on January 4, 2011 , published on April 24, 2012 , inventor: BA Omotowa.
- ^ H. Deslandres: Absorption de l'azote par le lithium à froid . In: Comptes rendus . tape 121 , 1895, pp. 886-887 ( bnf.fr ).
- ↑ Q. Liao, N. Saffon-Merceron, N. Mézailles: N 2 reduction into Silylamines at tridentate phosphine / Mo center: catalysis and mechanistic study . In: ACS Catal. tape 5 , no. 11 , 2015, p. 6902-6906 , doi : 10.1021 / acscatal.5b01626 .
- ↑ M. Yuki, H. Tanaka, K. Sasaki, Y. Miyake, K. Yoshizawa, Y. Nishibayashi: Iron-catalyzed transformation of molecular dinitrogen into silylamine under ambient conditions . In: Nature Commun. tape 3 , 2012, p. 1254 , doi : 10.1038 / ncomms2264 .
- ↑ H. Tanaka et al .: Molybdenum-Catalyzed Transformation of Molecular Dinitrogen into Silylamine: Experimental and DFT Study on the Remarkable Role of Ferrocenyldiphosphine Ligands . In: J. Am. Chem. Soc. tape 133 , no. 10 , 2011, p. 3498-3506 , doi : 10.1021 / ja109181n .
- ^ Y. Nishibayashi: Recent progress in transition-metal-catalyzed reduction of molecular dinitrogen under ambient reaction conditions . In: Inorg. Chem. Band 54 , no. 19 , 2015, p. 9234-9247 , doi : 10.1021 / acs.inorgchem.5b00881 .