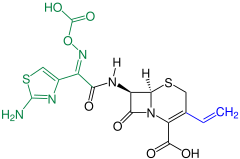
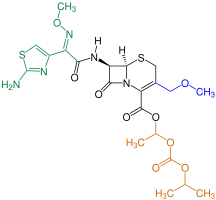
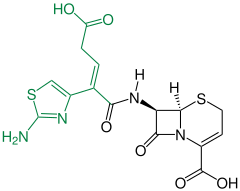
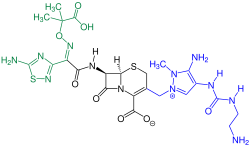
Cephalosporins
Cephalosporins (from kephale κέφαλη for “head” and sporos σπόρος for “spore”) form a group of broad spectrum antibiotics and, like penicillin , belong to the β-lactam antibiotics . They have a bacteriostatic effect by inhibiting the rebuilding of the cell walls in dividing bacteria (here the internal cross-linking of the murein layer ).
Cephalosporins occur naturally in the mold Acremonium chrysogenum (formerly Cephalosporium acremonium ) as Cephalosporin-C. Cephalosporin-C was first discovered in 1945 by Giuseppe Brotzu at the University of Cagliari and examined more closely by Edward Abraham and colleagues in Oxford. The chemical structure, which has similarities to the structure of penicillin G , was clarified in 1953. Because of its weak pharmacological effect, it is of no importance, but it still serves as the basic substance for the production of more effective, semi-synthetic cephalosporins. Biofermentation takes place today mainly in Hubei Province in China.
Classification and spectrum of activity
There is no internationally binding classification of cephalosporins. A classification can be made according to the spectrum of activity according to generations or groups , as well as according to the type of intake ( parenteral or perorally ).
None of the cephalosporins currently available on the market are not effective against enterococci because they have primary resistance (the so-called "enterococcal gap").
Division into groups
Often the division is made according to generations . Since the "replacement" of older cephalosporins by newer substances does not take place in all cases, the term group is used here for classification. “Classical” or cephalosporins of the first group are characterized by a good effectiveness against staphylococci , also against β-lactamase producers (see below). Their effectiveness against gram-negative pathogens is limited. In addition to the first generation cephalosporins, there are a number of modifications that are known as the second and third group cephalosporins . The advantage of the second group is better resistance to β-lactamase , an enzyme that some bacteria, especially gram-negative rod bacteria, can use to protect themselves against β-lactam antibiotics (see above). The third group has a broader spectrum of activity, i.e. it covers other bacteria. At least 18 substances were approved on the German market in the 1990s; After a consolidation phase, the Red List now contains nine cephalosporins with good tolerability and effectiveness. Cephalosporins of the fourth group also show an activity against Pseudomonas aeruginosa . In contrast to the other cephalosporins, those of the fifth group are also effective against MRSA .
Parenteral and peroral therapy
Cephalosporins can also be divided into whether they are administered parenterally or are suitable for peroral therapy. There are also cephalosporins that are only approved for veterinary medicine (e.g. cefalonium).
Group 1 cephalosporins (or classic cephalosporins)
These basic cephalosporins have the highest activity against staphylococci compared to others. They show weak activity against gram-negative bacteria and are unstable against many β-lactamases from gram-negative bacteria.
- Parenteral therapy: Cefazolin , Cefacetril , Cefapirin , Ceftezol , Cefazaflur , Cefalotin , Cefaloridin
- Oral therapy: Cefadroxil , Cefalexin , Cefradin , Cefaloglycine (no longer available in stores ), Cefatrizine , Cefazedon , Cefroxadin
- Exclusively in veterinary medicine: Cefalonium
2nd group cephalosporins
These transitional cephalosporins are sufficient compared to group 1, but less effective against gram-positive bacteria. But they show an improved effect in the gram-negative range. They are stable to penicillinases from staphylococci and to a limited extent stable to β-lactamases from gram-negative bacteria.
- Parenteral therapy: cefuroxime sodium , cefotiam (no longer available)
- Oral therapy: cefuroxime (as cefuroxime axetil ) , cefaclor , loracarbef (no longer available)
3rd group cephalosporins
These broad-spectrum cephalosporins are less effective against staphylococci, but significantly more effective against gram-negative bacteria than groups 1 and 2. They are stable against numerous β-lactamases of gram-negative bacteria.
- Parenteral therapy: cefotaxime , ceftriaxone , ceftazidime , ceftazidime can also be used inhalatively. Ceftazidime is also the only cephalosporin of this generation that is also effective against pseudomonads .
- Oral therapy: cefixime , cefpodoxime (as cefpodoxime proxetil ), ceftibutene
- Exclusively in veterinary medicine: Cefovecin , Ceftiofur
4th group cephalosporins
These cephalosporins are also known as broad-spectrum cephalosporins . Ceftolozane shows high activity against Pseudomonas aeruginosa and, when combined with tazobactam, also detects ESBL-producing pathogens.
- Parenteral therapy: cefepime , ceftolozane (with tazobactam)
5th group cephalosporins
These cephalosporins are also referred to as broad-spectrum cephalosporins . Ceftaroline Fosamil is not effective against Pseudomonas aeruginosa . Both show an effectiveness against MRGN and other gram-positive as well as gram-negative pathogens.
- Parenteral therapy: Ceftobiprole , Ceftaroline Fosamil
Inhibitory concentration
The minimum inhibitory concentration (MIC) against microorganisms is the lowest concentration of the antibiotic at which the growth of a microbial pathogen is barely inhibited over a certain period of time. The microbiological examination of new β-lactam antibiotics usually involves an MIC examination with a large number of mostly freshly isolated bacteria against the new substance compared to older substances.
| General structure | Surname | S. aureus | E. coli |
|---|---|---|---|

|
Cefalotin | 0.2-0.4 | 6.2 |
| Cephaloridin | 0.02 | 3.2-6.4 | |
| Cefapirin | 0.1-0.4 | 12 | |
| Cefamandol | 0.2-0.8 | 0.4-0.8 | |
| Cefuroxime | 0.8 | 0.8-6.2 | |
| Cefotaxime | 3.2 | 0.04-0.10 | |
| Cefoperazone | 1.6 | 0.04-0.40 |
literature
- Gunter Schmidt: Cephalosporins . In: Chemistry in Our Time . tape 10 , no. 6 , 1976, p. 189-195 , doi : 10.1002 / ciuz.19760100605 .
- Mutschler: drug effects - textbook of pharmacology and toxicology . 8th edition. Wissenschaftliche Verlagsgesellschaft, Stuttgart 2001, ISBN 3-8047-1763-2 , p. 793-797 .
- Klaus Aktories, Ulrich Förstermann, Franz Bernhard Hofmann, Klaus Starke: General and special pharmacology and toxicology . 12th edition. Munich 2017, ISBN 978-3-437-42525-7 , 33.2.2.
Individual evidence
- ^ A. Riva, B. Orrù, E. Fruciano: Giuseppe Brotzu and the Discovery of Cephalosporines , 8th European Conference of Medical and Health Libraries, Cologne, September 16-21, 2002.
- ↑ According to management consultant Morris Hosseini, quoted by Tobias Lau: So far we have always got away, maybe this time too , in: Apotheke ad hoc, February 12, 2020, accessed on February 15, 2020
- ↑ a b c d e f g Klaus Aktories, Ulrich Förstermann, Franz Bernhard Hofmann, Klaus Starke: General and special pharmacology and toxicology . 12th edition. Munich 2017, ISBN 978-3-437-42525-7 , 33.2.2.
- ^ Hahn H et al .: Medical microbiology and infectious diseases. 6th edition. Springer, Heidelberg 2009.
- ↑ Entry on Cefalonium in the DrugBank of the University of Alberta , accessed June 18, 2019.
- ↑ Qin Lu, Nebulized ceftazidime and amikacin in ventilator-associated pneumonia , Am J Respir Crit Care Med 2011; 184: 106-115; PMID 21474643 ; doi: 10.1164 / rccm.201011-1894OC .
- ^ H. Lüllmann, K. Moh, L. Hein, Taschenatlas Pharmakologie , 6th edition, Georg Thieme Verlag, Stuttgart 2008, p. 255.
- ↑ MSD (Ed.): Specialist information ZERBAXA (Ceftolozane and Tazobactam) .
- ↑ Specialist information Zavicefta 2g / 0.5g: Specialist information Zavicefta 2g / 0.5g . Ed .: Pfizer.
- ↑ Nils-Olaf Hübner, Katharina Sciermoch, Axel Kramer: Comparison of methods for determining the minimum inhibitory concentration and conclusions for the further development of the methods . In: GMS Hospital Hygiene Interdisciplinary . tape 2 , no. 2 , December 28, 2007, p. Doc34 ( egms.de [accessed December 6, 2018]).
- ↑ B. Wiedemann: In vitro assessment parameters of beta-lactam ring antibiotics . In: Infection . tape 6 , no. 2 , March 1, 1978, p. S203-S206 , doi : 10.1007 / BF01638974 .
- ^ Gerhard G. Habermehl, Peter E. Hammann, Hans C. Krebs, W. Ternes: Naturstoffchemie: An introduction . Springer, Berlin; 3rd, completely revised u. exp. Edition 2008; ISBN 978-3-540-73732-2 ; P. 301.