Choline
| Structural formula | ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
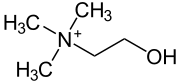
|
||||||||||||||||||||||
| General | ||||||||||||||||||||||
| Surname | Choline | |||||||||||||||||||||
| other names |
|
|||||||||||||||||||||
| Molecular formula |
|
|||||||||||||||||||||
| Brief description |
colorless, hygroscopic solid with a weak amine-like odor |
|||||||||||||||||||||
| External identifiers / databases | ||||||||||||||||||||||
|
||||||||||||||||||||||
| Drug information | ||||||||||||||||||||||
| ATC code | ||||||||||||||||||||||
| properties | ||||||||||||||||||||||
| Molar mass | 139.62 g mol −1 | |||||||||||||||||||||
| Physical state |
firmly |
|||||||||||||||||||||
| Melting point |
Decomposes at 180 ° C |
|||||||||||||||||||||
| solubility |
|
|||||||||||||||||||||
| safety instructions | ||||||||||||||||||||||
|
||||||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||||||||
Choline [ çoˈliːn ] (from ancient Greek χολή cholé , German ' bile ' ) is a primary monohydric alcohol and a quaternary ammonium compound . Choline chloride is usually commercially available .
history
Choline was discovered in pig bile by Adolph Strecker in 1849 and characterized and named in 1862. It was chemically synthesized for the first time in 1866 and 1867, independently of one another by Adolf Baeyer and Charles Adolphe Wurtz .
Occurrence
Choline is very common among living things. In the form of its acetic acid ester it forms the neurotransmitter acetylcholine , in the form of its phosphoric acid ester it is part of the lecithins (phosphatidylcholine) and also an intermediate product of metabolism.
Choline is found in food both in free form and in bound form as sphingomyelin or phosphatidylcholine (lecithin). It is found in smaller quantities in cereals (e.g. wheat germ), soybeans, vegetables and nuts. The highest levels are found in egg yolks, beef and pork liver.
Extraction and presentation
Choline can be produced by exhaustive methylation of the amine function of monoethanolamine . Another synthesis consists in the reaction of trimethylamine with ethylene oxide .
use
In food supplements , animal feed , preparations against liver damage ( fatty liver ).
In semiconductor production , choline is used as a cleaning agent for wafers .
In biotechnology , choline chloride is used in culture media for plants.
In the form of 18 F-choline, it is used in positron emission tomography as a tracer in the diagnosis of prostate cancer .
Choline chloride is an additive in fracking fluids .
Biological importance
The biosynthesis of the important neurotransmitter acetylcholine takes place through the transfer of an acetyl residue to the choline by the choline acetyl transferase ( EC 2.3.1.6 ) . In reverse of this reaction, acetylcholinesterase is responsible for the hydrolysis of this ester to acetic acid and choline.
Choline is phosphorylated by choline kinase ( EC 2.7.1.32 ) . The resulting zwitterionic O - phosphocholine serves as the starting material for the biosynthesis of phosphatidylcholines, which are an essential component of biomembranes .
Choline can also be oxidized to betaine , the zwitterionic trimethylglycine , which, in addition to folic acid , S- adenosylmethionine and vitamin B 12, is an important methyl group carrier in the metabolism .
Choline also acts as a lipotropic factor and can prevent fat from being stored in the liver. According to a scientific assessment by the European Food Safety Authority (EFSA) , choline contributes to normal lipid metabolism and normal liver function and a normal metabolism of homocysteine . Corresponding health-related advertising statements are permissible for foods with sufficient choline content throughout the European Union according to Regulation (EC) No. 1924/2006 (Health Claims) .
Various esters of choline, e.g. B. the known as suxamethonium succinyldicholine, act as agonists of the acetylcholine receptors , without being able to be quickly broken down by existing cholinesterases . After a short arousal, there is a long-lasting depolarization and thus muscle relaxation .
Vitamin-like substance
For some animals, dietary choline intake is essential . Choline is not a necessity for humans as long as their diet contains the amino acids methionine and folic acid . Choline was previously considered a vitamin and - like adenine - was called vitamin B 4 , although it was known that the compound can be synthesized by the human organism. Currently, choline is therefore often classified as a vitamin-like substance , including inositol . In monogastric animals, it is also ingested through food, since choline is present in the cell membrane as a component of phosphatidylcholines. Ruminants are an exception here, as choline is almost completely broken down in the rumen .
Adequate Intake and Tolerable Upper Intake Level
The US Institute of Medicine of the National Academy of Sciences gives the following values for Adequate Intake (AI) and Tolerable Upper Intake Level (UL) of choline:
| Infants and children |
AI (mg / day) | UL (mg / day) | Men | AI (mg / day) | UL (mg / day) | Women | AI (mg / day) | UL (mg / day) | Pregnant and breastfeeding women |
AI (mg / day) | UL (mg / day) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 0–6 months | 125 | - | 9–13 years | 375 | 2000 | 9–13 years | 375 | 2000 |
Pregnant women ≤ 18 years |
450 | 3000 |
| 7-12 months | 150 | - | 14-18 years | 550 | 3000 | 14-18 years | 400 | 3000 |
Pregnant women 19-30 years |
450 | 3500 |
| 1-3 years | 200 | 1000 | 19-30 years | 550 | 3500 | 19-30 years | 425 | 3500 |
Pregnant women aged 31–50 |
450 | 3500 |
| 4-8 years | 250 | 1000 | 31–50 years | 550 | 3500 | 31–50 years | 425 | 3500 |
Breastfeeding women ≤ 18 years |
550 | 3000 |
| 50–70 years | 550 | 3500 | 50–70 years | 425 | 3500 |
Breastfeeding 19-30 years |
550 |
3500 |
|||
| > 70 years | 550 | 3500 | > 70 years | 425 | 3500 |
Breastfeeding 31–50 years |
550 |
3500 |
|||
|
|||||||||||
See also
literature
- Wolfgang Herrmann, Rima Obeid (Ed.): Vitamins in the prevention of human diseases . Gruyter, 2011, ISBN 978-3-11-021448-2 , pp. 599-628 .
Web links
Individual evidence
- ↑ a b c Entry on choline. In: Römpp Online . Georg Thieme Verlag, accessed on May 29, 2014.
- ↑ External identifiers or database links for choline chloride : CAS number: 67-48-1, EC number: 200-655-4, ECHA InfoCard: 100.000.596 , GESTIS substance database : 27810 , PubChem : 6209 , ChemSpider : 5974 , Wikidata : Q2964153 .
- ↑ External identifiers or database links for choline hydroxide : CAS number: 123-41-1, EC number: 204-625-1, ECHA InfoCard: 100.004.206 , GESTIS substance database : 102101 , PubChem : 31255 , ChemSpider : 28995 , Wikidata : Q27268827 .
- ↑ Choline data sheet (PDF) from Merck , accessed on January 19, 2011.
- ↑ Entry on choline chloride in the GESTIS substance database of the IFA , accessed on May 9, 2017(JavaScript required) .
- ↑ Adolph von Strecker: Observations on the bile of various animals . In: Annals of Chemistry and Pharmacy . tape 70 , no. 2 , 1849, p. 149-197 , doi : 10.1002 / jlac.18490700203 .
- ↑ A. Strecker: About some new components of pig bile . In: Annals of Chemistry and Pharmacy . tape 123 , no. 3 , 1862, p. 353-360 , doi : 10.1002 / jlac.18621230310 .
- ^ Adolf Baeyer: Synthesis of Neurin . In: Annals of Chemistry and Pharmacy . tape 140 , no. 3 , 1866, pp. 359-364 , doi : 10.1002 / jlac.18661400308 .
- ↑ A. Wurtz: About a new class of composite ammonia . In: Annals of Chemistry and Pharmacy . tape 142 , no. 3 , 1867, p. 306-313 , doi : 10.1002 / jlac.18671420318 .
- ↑ a b Ibrahim Elmadfa, Claus Leitzmann: Human nutrition (= UTB for science ). Ulmer.
- ↑ DocMedicus Vitalstofflexikon: Choline content in foods
- ↑ Michael E. Horn, Joseph H. Sherrard, Jack M. Widholm: Photoautotrophic Growth of Soybean Cells in Suspension Culture: I. Establishment of Photoautotrophic Cultures . In: Plant Physiol. tape 72 , no. 2 , 1983, p. 426-429 (English, PDF [accessed August 7, 2009]).
- ↑ EFSA : Scientific opinion on the substantiation of health claims related to choline and contribution to normal lipid metabolism (ID 3186), maintenance of normal liver function (ID 1501), contribution to normal homocysteine metabolism (ID 3090), maintenance of. In: EFSA Journal , 9, No. 4, 2011, p. 2056, doi : 10.2903 / j.efsa.2011.2056 .
- ^ SH Zeisel, KA Da Costa, PD Franklin, EA Alexander, JT Lamont, NF Sheard, A. Beiser: Choline, an essential nutrient for humans. In: FASEB Journal , Vol. 5, No. 7, 1991, pp. 2093-2098.
- ↑ a b Hans Konrad Biesalski, Peter Grimm: Pocket Atlas of Nutrition . Georg Thieme Verlag, 2011, ISBN 3-13-167605-1 , p. 78 ( limited preview in Google Book search).
- ↑ Jürgen Stein: Practical Guide to Clinical Nutrition and Infusion Therapy . Springer DE, 2003, ISBN 3-642-55896-8 , pp. 388 ( limited preview in Google Book search).
- ↑ a b c National Academy of Medicine : Dietary Reference Intakes: Vitamins ( Memento of the original from September 1, 2010 in the Internet Archive ) Info: The archive link was inserted automatically and has not yet been checked. Please check the original and archive link according to the instructions and then remove this notice.
- ↑ a b usda.gov: Interactive DRI Glossary ( Memento of the original from December 21, 2014 in the Internet Archive ) Info: The archive link was inserted automatically and has not yet been checked. Please check the original and archive link according to the instructions and then remove this notice.