Natamycin
| Structural formula | ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||||||||
| General | ||||||||||||||||||||||
| Non-proprietary name | Natamycin | |||||||||||||||||||||
| other names |
|
|||||||||||||||||||||
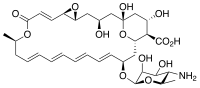
| Molecular formula | C 33 H 47 NO 13 | |||||||||||||||||||||
| Brief description |
white to light yellow powder |
|||||||||||||||||||||
| External identifiers / databases | ||||||||||||||||||||||
|
||||||||||||||||||||||
| Drug information | ||||||||||||||||||||||
| ATC code | ||||||||||||||||||||||
| Drug class | ||||||||||||||||||||||
| properties | ||||||||||||||||||||||
| Molar mass | 665.73 g · mol -1 | |||||||||||||||||||||
| Physical state |
firmly |
|||||||||||||||||||||
| Melting point |
280 ° C (decomposition) |
|||||||||||||||||||||
| solubility |
hardly soluble in water |
|||||||||||||||||||||
| safety instructions | ||||||||||||||||||||||
|
||||||||||||||||||||||
| Toxicological data | ||||||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||||||||
Natamycin is a fungistatic to fungicidal antimycotic from Streptomyces natalensis , an Actinobacterium of the genus Streptomyces . It is a macrolide - polyene - antifungal and is used in the food industry and as a drug . The use of natamycin as a medicine is very limited.
Application in the food industry
Natamycin ( E 235 ) is approved and used as a preservative for the surface treatment of the rind of hard cheese , semi- hard cheese and semi-hard cheese and for dried and cured sausages. According to food law, a maximum amount of 1 mg / dm 2 surface applies . Natamycin must no longer be detectable 5 mm below the cheese rind.
In some non-EU countries, the use of natamycin for other foods and luxury foods is permitted, e.g. B. for wine The natamycin serves on the one hand to interrupt fermentation at a certain residual sugar content and on the other hand as protection against undesired microbial influences after fermentation by harmful organisms. In order to achieve an effect, approximately an addition of 10 mg natamycin per liter is necessary. In the EU, use is not permitted except for the foods mentioned above (cheese, sausages), so imports must be free of natamycin.
The Bavarian State Ministry for the Environment and Consumer Protection declared in 2012: “Natamycin [...] is similar to penicillin. The formation of resistant microorganisms is therefore fundamentally to be feared ”, although in previous years the maximum permissible amount or penetration depth could never be verified in cheese bark samples.
Use as a medicine
Mechanism of action
Natamycin is an antifungal agent that is used against fungal infections ( mycoses ).
It attaches to the ergosterol , an important component of the cell membrane of the fungi, and thus causes pores to form in the cell membrane . Through these pores reach potassium - ions (K + ), and other cell components from the fungal cell. As a result, the fungal cell dies.
Admission and side effects
Natamycin is not absorbed (taken up) in the intestine . That is why natamycin is only used locally. A daily dose of 300–400 mg / kg - given over several days - causes nausea , vomiting and diarrhea .
Application areas (indications)
- Yeast infections ( candidosis ) of the intestine
- Fungal infections of the oral cavity (oral thrush): selective oral decontamination
- Fungal infections of the eyes , eyelids and tear ducts
- Relapse prophylaxis in chronically recurrent vaginal mycoses
Chemical properties
Natamycin is a white crystalline powder that hardly dissolves in water and alcohols. It is also almost insoluble in higher quality alcohols or oils. Natamycin is sensitive to light and is not absorbed.
Trade names
Pima-biciron (D), pimafucin (D), pimaricin
Veterinary medicine: Mycophyte ad us. vet. (CH)
literature
- Marianne Abele-Horn: Antimicrobial Therapy. Decision support for the treatment and prophylaxis of infectious diseases. With the collaboration of Werner Heinz, Hartwig Klinker, Johann Schurz and August Stich, 2nd, revised and expanded edition. Peter Wiehl, Marburg 2009, ISBN 978-3-927219-14-4 , p. 255.
Web links
- Vetpharm entry on Natamycin , accessed August 11, 2012.
- European Food Safety Authority : Scientific opinion on the use of natamycin (E 235) as a food additive , 14 December 2009.
Individual evidence
- ↑ Entry on E 235: Natamycin in the European database on food additives, accessed on August 11, 2020.
- ↑ Entry on NATAMYCIN in the CosIng database of the EU Commission, accessed on March 21, 2020.
- ↑ a b c d e Datasheet Natamycin from Sigma-Aldrich , accessed on May 13, 2017 ( PDF ).
- ^ Scientific Opinion on the use of natamycin (E 235) as a food additive. In: EFSA Journal. 7, 2009, p. 1412, doi : 10.2903 / j.efsa.2009.1412 .
- ↑ Lower Saxony State Office for Consumer Protection and Food Safety : Natamycin in cheese rind - everything safe?
- ↑ Why is natamycin used? Internet presence of the manufacturer DSM Food Specialties BV, Delf (NL), subsidiary of DSM
- ↑ R. Godelmann, K. Zur, M. Rupp: Verbotenes Antimykotikum Natamycin in Wein. The investigation offices for food control and animal health, October 26, 2010, accessed on October 4, 2016 .
- ↑ Food additives: pimaricin, natamycin .
- ^ Natamycin - Use of Natamycin. In: Natamycin.com. Retrieved October 4, 2016 .
- ↑ Natamycin on cheese rinds, Dr. Knut Werkmeister, Bavarian State Office for Health and Food Safety , on the Bavarian consumer portal of the Bavarian State Ministry for the Environment, Health and Consumer Protection . See also BfR statement January 2012: The area of application of natamycin as a food additive should not be expanded.
- ^ Joint FAO / WHO Expert Committee on Food Additives (JECFA), Monograph for Natamicin (Pimaricin) , accessed December 9, 2014.