Inulin
| Structural formula | ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
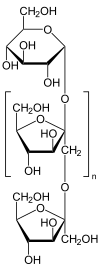
|
||||||||||||||||||||||
| n = about 35 | ||||||||||||||||||||||
| General | ||||||||||||||||||||||
| Surname | Inulin | |||||||||||||||||||||
| other names |
|
|||||||||||||||||||||
| Molecular formula | C 6n H 10n + 2 O 5n + 1 | |||||||||||||||||||||
| Brief description |
white, crystalline solid |
|||||||||||||||||||||
| External identifiers / databases | ||||||||||||||||||||||
|
||||||||||||||||||||||
| Drug information | ||||||||||||||||||||||
| ATC code | ||||||||||||||||||||||
| properties | ||||||||||||||||||||||
| Molar mass | varies with the chain length of the polymer | |||||||||||||||||||||
| Physical state |
firmly |
|||||||||||||||||||||
| Melting point |
178-181 ° C |
|||||||||||||||||||||
| solubility |
soluble in hot water, slightly soluble in organic solvents |
|||||||||||||||||||||
| safety instructions | ||||||||||||||||||||||
|
||||||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | ||||||||||||||||||||||
Inulin is a mixture of polysaccharides made from fructose building blocks with a chain length of up to 100 monomers and a terminal glucose residue . It is one of the fructans .
Occurrence and discovery

Many plants store inulin as a reserve substance , especially types of the sunflower and umbelliferous plants . Examples include yacon , Jerusalem artichoke , chicory , dahlia , artichoke , Dandelion , salsify , chicory , and parsnips . Inulin was discovered in 1807 by Valentin Rose the Younger in the alanten root ( Inula helénium L. ).
use
In nutrition
Nowadays, inulin is often used as an ingredient in food production , for example in yogurt as a fat substitute and to improve the taste, texture and mouthfeel . In sausages it is used to increase the fiber content. Inulin is one of the prebiotic food additives. It also serves as a base for the hydrolytic production of fructose and oligofructose .
Inulin can be metabolized by bifidobacteria as an energy source . This means that inulin uptake can be used for the selective accumulation of bifidobacteria in the human intestine. Excessive intake can lead to flatulence and diarrhea in people with a diet that favors unbranched and simple carbohydrates, but intestinal flora .
In lesser amounts, inulin plays a role as an additive in animal nutrition.
In the medicine
Low molecular weight inulin is soluble in warm water . It is used in physiological research to determine extracellular space because it easily penetrates the interstitium but not the cells . The measurement of the inulin clearance can be used (instead of the usual creatinine clearance ) to determine the glomerular filtration rate (GFR) of the kidneys more precisely. This is possible because inulin is completely filtered in the glomerulum , but is neither secreted nor reabsorbed in the tubule system .
Inulin can be used as a starch substitute in the therapy of diabetes mellitus , as it does not affect the blood sugar level. Inulin is not absorbed in the small intestine because humans lack the degrading enzyme ( inulinase ). Instead, it is fermentatively broken down in the large intestine by bacteria ( bifidus bacteria , Bakteroides strains and Eubacteriacae ) into short-chain fatty acids . Due to the increased acid production, the pH value in the colon drops . The gases formed in the process can lead to flatulence in sensitive people (especially irritable bowel syndrome ) - the only known side effect of consuming inulin-containing plant parts, which is why inulin should also be counted among the FODMAPs . The u. a. The resulting propionates can also act as a natural appetite suppressant .
Starting material for other compounds
When roasting of chicory roots for the production of coffee substitute inulin is partially hydroxymethylfurfural converted, which provides for the coffee-like aroma.
See also
literature
Individual evidence
- ↑ a b c d data sheet inulin at AlfaAesar, accessed on February 13, 2013 ( PDF )(JavaScript required) . .
- ↑ Inulin data sheet at Acros, accessed on November 22, 2007.
- ^ The Merck Index: An Encyclopedia of Chemicals, Drugs, and Biologicals. 14th edition. Merck & Co., Whitehouse Station, NJ, USA 2006, ISBN 0-911910-00-X , p. 870 #.
- ↑ Article on reduced- fat sausage products in www.foodaktuell.ch ( Memento of the original from September 16, 2011 in the Internet Archive ) Info: The archive link was inserted automatically and has not yet been checked. Please check the original and archive link according to the instructions and then remove this notice.
- ↑ Example of a poultry pie ( Memento of the original from April 18, 2010 in the Internet Archive ) Info: The archive link was inserted automatically and has not yet been checked. Please check the original and archive link according to the instructions and then remove this notice.
- ↑ opinion on fructooligosaccharides and inulin , food Chemical Society - Division of the German Chemical Society
- ↑ Glenn R. Gibson, Emily R. Beatty, Xin Wang, John H. Cummings: Selective stimulation of bifidobacteria in the human colon by oligofructose and inulin. In: Gastroenterology. 108, No. 4, 1995, pp. 975-982. doi: 10.1016 / 0016-5085 (95) 90192-2 .
- ↑ Nutritional advice Rhineland-Palatinate: Forgotten vegetables: black salsify . Last visited on January 29, 2018.
- ^ Kurt Widhalm: Nutritional Medicine . Ed .: Kurt Widhalm. 2nd Edition. Verlagshaus der Ärzte, Vienna 2005, ISBN 3-901488-51-0 , p. 137 .
- ↑ A. Pedersen, B. Sandström, JM Van Amelsvoort: The effect of ingestion of inulin on blood lipids and gastrointestinal symptoms in healthy females. In: Br J Nutr. 78 (2), Aug 1997, pp. 215-222. PMID 9301412 .
- ↑ L. Sobotka, M. Brátova, M. Slemrová, J. Manák, J. Vizd'a, Z. Zadák: Inulin as the soluble fiber in liquid enteral nutrition. In: Nutrition. 13 (1), Jan 1997, pp. 21-25. PMID 9058443 .
- ↑ MVZ Institute for Microecology GmbH: FODMAP-poor diet for irritable bowel patients. Retrieved June 3, 2020.
- ↑ Harald Frater: scinexx - Help against food cravings: A degradation product of dietary fiber inhibits the desire to feast. In: scinexx.de. July 5, 2016, accessed July 5, 2016 .
- ↑ Ben-Erik van Wyk: Food Plants of the World. Timber Press, 2006, ISBN 0-88192-743-0 , p. 134.

