Ehlers-Danlos Syndrome
| Classification according to ICD-10 | |
|---|---|
| Q79.6 | Ehlers-Danlos Syndrome |
| ICD-10 online (WHO version 2019) | |
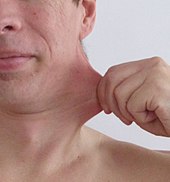
The Ehlers-Danlos syndrome ( EDS ) is a heterogeneous group of congenital disorders in the connective tissue , which is mainly characterized by a hyperextensibility skin and through movable joints. But it also influences vessels, muscles, ligaments, tendons and internal organs.
To date, 19 gene mutations are known to cause EDS. The various mutations lead to a change in the structure, production or processing of collagen or of proteins that interact with collagen. The frequency of occurrence in the population is assumed to be 1: 5,000 to 1: 10,000, so EDS is a rare disease. There are no differences in occurrence between different ethnic groups or the sexes.
history
The syndrome is one of the oldest known causes of effusions and bleeding, which was reported as early as 400 BC by Hippocrates. Were recognized. The first case analysis of a Spanish man with abnormal skin elasticity is known from the Dutch surgeon Job Janzoon von Meerkerin from 1668. The first comprehensive description of the syndrome with its many facets (skin, joints, scars, etc.) was made in 1891 by the Russian dermatologist Chernogubov. However, the isolated tsarist empire prevented the study from becoming generally known, so that Edvard Ehlers , who described the essential connections in 1901 as Cutis laxa, and Henri-Alexandre Danlos in 1908, with the suggestion that overstretchability and tearability of the skin be named as cardinal symptoms, gave the syndrome its name .
Other mainly outdated names of the EDS were:
- Ehlers-Danlos-Meekeren syndrome, Meekeren-Ehlers-Danlos syndrome, Van Meekeren syndrome
- Danlos syndrome
- Fibrodysplasia elastica generalisata, cutis hyperelastica
- Dermatolysis
- Rubber skin, Indian rubber skin
- Chernogubov syndrome
- Sack-Barabas Syndrome
classification
An EDS classification was attempted from the late 1960s. In 1986 ten types were defined, which were published in 1988 at a conference in Berlin. With advancing knowledge in the molecular and biochemical field, EDS was subdivided in 1997. The Villefranche classification served a clinically simplified diagnosis of the Ehlers-Danlos syndrome and to differentiate between diseases that overlap with the EDS (see table). The disease could now be described by six types with their main and secondary criteria. Other, exotic forms of EDS have also been identified in individual cases. The Villefranche classification has been replaced by the new diagnostic criteria since 2017. The criteria have been completely revised and updated and now differentiate into 13 types. According to the new diagnostic criteria, the following symptoms are no longer assigned to the EDS spectrum: occipital horn syndrome, fibronectin deficiency (EDS type X), familial hypermobility syndrome (EDS type XI), X-linked EDS (EDS type V) and filamin A EDS.
There is a great deal of variability and overlap in symptoms between types. A clear classification is therefore only possible on the basis of clinical diagnostics and genetic tests or skin biopsies.
The causative mutations were identified. Only in the hypermobile type is the triggering gene not yet known.
New diagnostic criteria 2017
At the same time as the publication of 18 articles on the subtypes of Ehlers-Danlos syndrome in the American Journal of Medical Genetics , new diagnostic criteria were also published in March 2017. These are far more comprehensive and specific than the previous Villefranche criteria. An international committee has brought together the experience of clinical examinations and the results of genetic studies in various working groups, also in order to improve doctors' sensitivity to this clinical picture and to improve diagnostics and thus patient care through cross-border criteria. For this purpose, in addition to the more specific possibilities for diagnostics, guidelines for treatment should also be developed.
The new diagnostic criteria replace the Villefranche classification. The Ehlers-Danlos syndrome is now divided into 13 types.
The distinction between EDS types
Hypermobile EDS (hEDS, formerly type 3): The genetic cause is not yet known. The diagnosis is made clinically after a detailed examination. hEDS is primarily characterized by the hypermobility of large and small joints. Subluxations and dislocations can occur regularly. HEDS sufferers often suffer from joint instability and often have soft, velvety skin that can be easily damaged. Very often patients suffer from chronic pain. The expression can be moderate to very strong. Around 90% of those affected by EDS have chronic pain, with the highest pain scores being found in hEDS patients.
Classic EDS (cEDS, formerly type 1 and 2): Around 90% of the genetic cause for this EDS type is found in the genes COL5A2 and COL5A. Mutations in COL1A1 are rare. Major criteria are extremely elastic skin that is fragile and easily vulnerable, atrophic scarring and general joint hypermobility. There are also nine "minor" criteria, such as velvety skin, muscle hypotonia or pseudotumors.
Vascular EDS (vEDS, formerly type 4): The genetic cause can be localized in variations of the COL3A1 gene. There are five major criteria and 12 "minor" criteria that provide clinical evidence of vEDS. Patients often have thin, translucent skin that is very fragile and easily vulnerable. Veins and organs are also easily vulnerable. The joints are hypermovable; mostly these are small joints like fingers or toes. Club feet or torn ligaments and muscles may occur. VEDS is life threatening; the median age is 51 years. The life span is wide and ranges from around ten to around 80 years. The most common cause of death are arterial dissections or ruptures.
Kyphoscoliotic EDS (kEDS, formerly type 6): The trigger is variants in the PLOD1 gene, less often in the FKBP 14 gene. The major criteria are congenital muscle hypertension, congenital or early onset kyphoscoliosis and generalized hypermobility with (sub) dislocations. There are also minor criteria. Kyphoscoliosis is usually severe and progressive. Other symptoms can include soft, pasty skin, atrophic scarring, and poor wound healing.
Arthrochalasia EDS (aEDS, formerly Type 7 A and B): aEDS is caused by mutations in the genes COL1A1 and COL1A2. Around 30 cases are currently known. Those affected suffer from pronounced joint hypermobility and congenital, bilateral hip dislocation. In addition, (sub-) dislocations of the large and small joints, foot deformities and scoliosis, lordosis and kyphoscoliosis can occur. The skin can be overstretchable, velvety and easily vulnerable.
Dermatosparaxis EDS (formerly type 7C): Mutations in the ADAMTS2 gene trigger dEDS. Symptoms include extremely fragile skin that scars quickly. The skin is loose and in excess. The joint hypermobility ranges from mild to severe. dEDS is very rare - around ten cases are known to date.
Brittle cornea syndrome (BCS): BCS comes in two types. Variant 1 results from a mutation in the ZNF469 gene, variant 2 from a mutation in the PRDM5 gene. BCS is characterized by a progressive thinning of the cornea of the eye. A keratoglobus or keratoconus can appear early , as well as nearsightedness, hearing loss, and blue sclera. Classic EDS symptoms such as hypermobile joints and overstretching skin are often observed.
Classical-like EDS (clEDS): Mutations in the TNXB gene lead to clEDS. The skin is usually overstretchable and velvety, but does not show any atrophic scarring. There is often general joint hypermobility, (sub) dislocations can occur, especially the shoulders and ankles are affected.
Spondylodysplastic EDS (spEDS): spEDS is triggered by mutations in the genes B4GALT7, B3GALT6 and SLC39A13. Affected people are often small and have hypotonia.
Musculocontractural EDS (mcEDS): Mutations in the genes CHST14 and DSE are responsible for mcEDS. Characteristic are skin overstretchability, increased skin vulnerability, atrophic scarring and increased wrinkling in the palms of the hands. mcEDS leads to congenital contractures .
Myopathic EDS (mEDS): Mutations in the COL12A1 gene lead to mEDS. People with mEDS suffer from congenital hypotonia and / or muscular atrophy that progresses with age. Proximal joint contractures and joint hypermobility of distal joints can occur.
Periodontal EDS (pEDS): pEDS is triggered by mutations in the C1R gene. pEDS leads to severe, persistent periodontal disease, which occurs in childhood or adolescence. The gums often do not fit well against the tooth.
Cardio-valvular EDS (cvEDS): COL1A2 mutations can lead to cvEDS. Seriously progressive cardio-valvular symptoms, such as those of the mitral valve, and an overstretchable, easily vulnerable skin that tends to develop atrophic scarring characterize cvEDS. Joint hypermobility also occurs, which can either be generalized or limited to the small joints.
The distinction between groups
The EDS types can also be sorted according to groups. In this case, the classification is based on similarities in the triggering genes.
Group A: Disorders of the collagen primary structure and collagen processing, this includes cEDS, vEDS, aEDS, dEDS and cvEDS.
Group B: Disorders of collagen folding and collagen crosslinking, this includes kEDS-PLOD1 and kEDS-FKB14.
Group C: Disorders of the structure and function of the myomatrix, this includes clEDS and mEDS.
Group D: disorders of the glycosaminoglycan biosynthesis, this includes spEDS-B4GALT7, spEDS-b3GALT6, mcEDS-CHST14 and mcEDS-DSE.
Group E: Defects in the complement system, this includes pEDS.
Group F: Disorders of intracellular processes, this includes spEDS-SLC39A13 and BCS.
Group G: Currently unclear, this includes hEDS.
| Former classification according to "Villefranche" | Former "Berlin" classification | known genes ("Villefranche" classification) | Characteristics |
|---|---|---|---|
| Classic guy | EDS type I (gravis) and type II (mitis) | COL5A1 / COL5A2 and COL1A1 mutations | highly overstretchable and easily injured skin; Hematoma tendency; abnormal wound healing ; Hypermobility of the joints; internal organs and vessels also affected (symptoms in type II as in type I, only less pronounced) ( autosomal dominant inheritance ) Gorlin's sign, the nose can be touched with the tongue. |
| Hypermobile type | Hypermobile - EDS Type III | COL5A3 , TNXB; <5% tenaskin X deficiency, COL3A1 | only slightly overstretchable, silky skin; pronounced hypermobility of the joints; frequent (sub) dislocations of the joints, tendinitis, herniated discs etc .; Chronic limb and joint pain, family history ( autosomal dominant inheritance ) Reverse Namaskar sign, palms of the hands can be pressed against each other behind the back. |
| Vascular type | Arterial Form - EDS Type IV | COL3A1 | thin, translucent skin; pronounced tendency to hematoma ; Hypermobility of the small joints; Involvement of internal organs and vessels; spontaneous rupture of arteries and vessels, aneurysms (autosomal dominant inheritance) |
| Kyphoscoliosis type | Ocular Scoliosis - EDS Type VI | PLOD1 | Overstretchability of the skin medium to strong; abnormal wound healing; severe hypermobility of the joints; Eye involvement; Involvement of the internal organs, outdated term: Nevo syndrome (autosomal recessive inheritance) |
| Arthrochalasia type | Arthrochalasia multiplex - EDS types VIIA and VIIB | COL1A1 / COL1A2, partial or complete absence of exon 6 | Overstretchability of the skin (slight to medium), thin skin, hip dislocation , pronounced hypermobility of the joints (autosomal recessive inheritance) |
| Dermatosparaxis type | Dermatosparaxis - EDS type VIIC | COL1 pNPI mutation | Skin very slack, excessive mobility of the joints, involvement of the internal organs (autosomal recessive inheritance) |
| Other, exotic forms | X-linked EDS - EDS type V (X-linked recessive inheritance) | ||
| Periodontitis type - EDS type VIII (autosomal dominant inheritance) | |||
| Familial Hypermobility Syndrome - EDS Type XI | |||
| Fibronectin-deficient type - EDS type X (autosomal recessive inheritance) | |||
| Progeroid EDS | B3GALT6 mutation | ||
| Non-specific forms |
Disease incidence
A disease or illness is defined as rare in Europe when it affects less than one in two thousand people. EDS as a group is one of the rare diseases with a rate of 1: 5,000 to approx. 1: 10,000.
For the classic type of EDS, the occurrence is estimated at around 1: 25,000. The hypermobile type of EDS is the most common type of EDS, with an estimated frequency of 1 in 10,000. For the vascular type, an occurrence of 1: 50,000 is expected. The other types of EDS are even rarer and occur only sporadically (fewer than a hundred known cases per type worldwide).
The classic (cEDS), the hypermobile (hEDS), the vascular (vEDS), the arthrochalasia (aEDS) and the periodontal (pEDS) Ehlers-Danlos syndrome can be inherited in an autosomal dominant manner, which means that a child has a 50 percent Is likely to be inherited if one parent has the disease. The other Ehlers-Danlos syndromes (clEDS, cvEDS, dEDS, kEDS, BCS, spEDS, mcEDS) can be inherited in an autosomal recessive manner . Their children can only inherit the disease if both parents have this genetic defect. Myopathic EDS (mEDS) can be inherited in an autosomal dominant or an autosomal recessive manner. Every Ehlers-Danlos syndrome has its own malformation, which means that those affected can only pass on the EDS that they themselves suffer from. A person with vascular EDS can tell his child B. do not inherit a classic EDS. Each of the Ehlers-Danlos syndromes can also be caused de novo, i.e. by a new mutation.
It should be noted that all frequency numbers are pure hypotheses. They are only based on registered cases. There are currently around 5,000 known people with EDS in Germany, but a high number of unreported cases is expected.
Diagnosis and symptoms
EDS diagnostics begin with a detailed medical examination. The joint hypermobility should first be tested with the help of the Beighton Score. The skin is examined for feel (like doughy or velvety) and overstretchability. Any scars should also be examined. In addition, patients should be questioned extensively about symptoms and the medical record should be observed. The results obtained are then compared with the clinical criteria of the Ehlers-Danlos syndromes. A final diagnosis is made through a genetic test. The diagnosis of hypermobile Ehlers-Danlos syndrome (hEDS) is made clinically. The diagnostic criteria are divided into three criteria areas, all of which must be met. Criterion 1 is a positive result with regard to the Beighton Score (at least 5/9 for adults). Criterion 2 is divided into areas A, B and C. In order to meet criterion 2, two of the three areas must apply. A comprises the presence of at least 5/12 systematic manifestations, B comprises a familial occurrence of hEDS and C comprises pain and instability. Criterion 3 provides for the exclusion of other diseases.
Differential diagnosis should u. a. the following diseases are clarified:
- Marfan syndrome , Loeys-Dietz syndrome Q87.4
- Osteogenesis imperfecta Q87.0
- Stickler syndrome Q87.8
- Achondroplasia (chondrodysplasia) Q77.4
- Rheumatoid arthritis M06
- Fibromyalgia M79.7
- Multiple sclerosis G35
- Hypermobility syndrome
- Growing pains
- Acrogeria
The diagnosis of rare diseases in general, and EDS in particular, poses problems in practice. According to a study in 2005, z. For example, around 25% of those affected do not make their EDS diagnosis until 28 years after the first symptoms appear.
The initial formulation of a clinical suspicion is crucial for an EDS diagnosis. The decisive factor is the presence of major and minor criteria, which represent a high level of sensitivity for the disease. A positive family history can substantiate the suspicion of EDS. In practice, however, the diagnosis is often different. Usually there are different symptoms that are unusual in combination, severity and frequency of occurrence and thus initiate the search for an explanatory cause. Sick people often encounter hurdles in the health system. The ignorance of large parts of the medical profession about EDS often allows those affected to experience an odyssey of years leading up to the diagnosis.
Symptoms by area (incomplete):
| cardiology | Aortic aneurysm ; Vasodilatation; enlarged coronary artery; Mitral valve prolapse ; Chronic Cerebrospinal Venous Insufficiency ; Postural orthostatic tachycardia syndrome POTS |
| Rheumatology / Orthopedics | Hypermobility (Beighton score ≥ 5); Arches , arches , splayfoot ; spontaneous sub / dislocations; Chronic joint and muscle pain; Scoliosis / kyphosis ; Leg length difference; Hip dysplasia ; Hernia ; Keel breast (pectus carinatum), early osteoarthritis ; Degeneration of the intervertebral discs ; Cysts on joints, arachnodactyly , clinodactyly of the little finger. |
| Dermatology / Sports Medicine | soft, velvety skin ; thin, translucent skin; easy vulnerability; wide scars; small semolina knots ; delayed wound healing; Hematoma tendency ; frequent injuries; rapid fatigue; delayed motor development; Hypotonia |
| Dentistry | high palate ; Misaligned teeth ; Receding gums; Temporomandibular joint pain, craniomandibular dysfunction ; hypermobile tongue; Bleeding in the mouth / throat |
| Gastroenterology / Internal Medicine | Hernias ( hernia ); Gastroparesis ; Nausea; Abdominal pain; Indigestion caused by contractile disorders; Heartburn , Irritable Bowel Syndrome, Colon Enlargement, Hemorrhoids , Uterine Depression , Mast Cell Activity Disorder ; chronic exhaustion |
| neurology | Chiari malformation ; Tethered cord ; Dysautonomy ; Neuralgia ; Migraines ; cervical spondylolisthesis ; Cervical spine disease; Headache ; general coordination disorders; poor sense of balance; spinal stenosis ; decreased effectiveness of local anesthetic ; Weakness; Fatigue; Sleep disorder |
| psychology | Depression ; Anxiety ; Withdrawal from society through lack of understanding and pain; |
| Ophthalmology | Photosensitivity ; Squint ; Myopia ; dry eyes; Retinal detachment ; Lens shift; Keratoconus ; Macular degeneration |
Pain
Pain as a diagnostic criterion requires a more detailed consideration because of its relevance. Severe pain, which can be both chronic and acute, is common in all types of Ehlers-Danlos syndrome. This means that the pain can have a significant impact on those affected and be severely impairing. It can be related to hypermobility, the frequency of (sub) dislocations, injuries to the connective tissue and previous operations. The pain can affect the musculoskeletal system, be localized in several parts of the body ("widespread pain") and occur as myalgia , arthralgia and / or neuralgia . Since Ehlers-Danlos syndromes are diseases of the connective tissue that is found throughout the body, numerous processes can lead to pain. In hypermobile Ehlers-Danlos syndrome (hEDS), a. the loss of proprioception is seen as an important factor in the development of chronic pain. The movements of the joints above normal can lead to recurring tissue injuries.
Usually the pain treatment of the patient is not sufficient. This is also due to the complex formation of the pain, because it is often not about a pain that has lost its function, but about actual, constantly recurring injuries to various structures.
therapy
There is no cure for Ehlers-Danlos syndromes. In general it can be said that a multimodal therapy adapted to the patient is urgently recommended. This should consist of physiotherapy, pain therapy and psychological support. Patients with the vascular type are considered to be most at risk and should be under constant medical supervision. Medical intervention for all EDS types is limited to symptomatic therapy, which can be listed in a series of recommendations.
Monitoring of the cardiovascular system, physiotherapy, professional consideration (if it is possible to pursue a professional activity), orthopedic aids such as orthotics, bandages, walking sticks, walking frames or wheelchairs can be helpful. Activities that involve overstretching or blocking the joints should be avoided. Any necessary surgical interventions should be carried out with caution. There are various possible difficulties for the anesthesia technique that may be necessary, such as non-response to local anesthetics, difficult airway conditions, tendency to tear vessels when placing central venous catheters and massive bleeding events, especially during minor operations in EDS patients with fragile vessels. Surgical interventions should only be performed in centers with sufficient expertise in the treatment of EDS. Ruffles or similar joint stabilization often does not lead to the desired success. In physiotherapy, posture training and stability exercises with building up the small muscles responsible for them should be in the foreground. Bandages can be used to protect the most sensitive areas from injuries. Some patients react to the administration of vitamin C with a reduced tendency to swell and improved wound healing. Trials with biotechnical skin substitutes for non-healing wounds have been successful in individual patients.
Children should be provided with information about EDS so that they can understand why contact sports or other stressful leisure activities should be avoided. It is also important to encourage posture control at an early stage to prevent damage from poor posture. Family members, teachers and friends should also be actively informed so that they can accept the child and, if necessary, encourage them.
Emotional support, behavioral therapy and psychological support help those affected of all subtypes to accept the impairment or to cope better with it. Patient organizations can help with this (see web links).
Listing by subject area:
| cardiology | Ultrasound examination annually, every 3 years if the results are normal; Beta blockers for enlarged aorta and / or high blood pressure; Classical treatment of POTS and cardiovascular problems (as in patients without EDS) |
| Rheumatology / Orthopedics | physiotherapeutic accompaniment; Lifelong training for stability and strength (functional rehab program), exercises with minimal joint involvement and little strength: balance exercises, isometric exercises, water aerobics, tai chi, pilates, avoidance of contact sports, repetitive movement sequences and overstretching when stretching; Joint protection: avoidance of excess weight, shoe insoles; Bandages, kinesio taping for most affected joints; Adaptation of lifestyle and movement style; Ergonomic work and aids: supporting mattresses and pillows, writing utensils with reinforced handles, household aids; Avoidance of stabilizing operations; Joint replacement if necessary; periodic bone density measurement |
| Dermatology / Sports Medicine | Avoidance of trauma / injuries, if necessary daily administration of vitamin C; sticky bandages can crack the skin; Wound closure without tension and in two layers with the sutures left in place for a longer period of time, plasters against wide scars |
| Dentistry | Examinations and prophylaxis every 6 months, note reduced response to local anesthesia |
| Gastroenterology / Internal Medicine | small meals; Treatment of gastrointestinal problems classic; special monitoring during pregnancy |
| neurology | Pain treatment with steroid and non-steroid analgesics, other non-opioid analgesics and opioid analgesics, anticonvulsants, cannabinoids, low-dose naltrexone (LDN) |
| psychology | psychological support; Pain therapy; Mediation of patient organizations |
| Ophthalmology | regular eye exams (retina) |
Others
EDS hypermobile type vs. Hypermobility Syndrome :
The clinical criteria that applied to the hypermobility syndrome and the hypermobile variant of EDS were unspecific and not exclusive to both sides. Therefore, some doctors and scientists have suggested that hypermobility syndrome is a mild variant of the hypermobile type of EDS. As part of the new diagnostic criteria from 2017, the criteria for hypermobile Ehlers-Danlos syndrome (hEDS) have been significantly revised. For patients who do not meet these requirements and who do not otherwise explain the symptoms, the term hypermobility spectrum disorders (HSD) was introduced as part of the update. HSD bridges the gap between asymptomatic joint hypermobility and hEDS.
Methylene tetrahydrofolate reductase
Methylene tetrahydrofolate reductase (MTHFR for short) plays a major role in the methylation of the body and is particularly involved in the metabolism of B6, B12 and folic acid. Mutations in this gene have been linked to several clinical pictures, particularly neural tube defects, homocysteinuria, and it is speculated that it may also lead to a phenotype of Ehlers-Danlos syndrome. Details are not known, but mutations in this gene have been found in some families with Ehlers-Danlos syndrome.
forecast
The outlook for people with EDS depends on the type they have been diagnosed with. Symptoms vary even within the subtypes and the frequency of complications differs from patient to patient. Some sufferers have only minor restrictions, while others are severely restricted by the severity of their daily life. Extreme joint instability , pain, and spinal deformation can severely limit mobility. Most people have a normal life expectancy. However, patients with vascular involvement are at an increased risk of serious complications.
EDS is a lifelong condition that is usually progressive . Those affected are confronted with social obstacles because of their illness. Some patients report fears of severe and painful ruptures, of deterioration, of unemployment due to their physical and emotional burdens, and of social exclusion in general. A gene therapy or other approaches for a cure are not in sight, studies are not yet known.
Ehlers-Danlos syndrome in animals
Ehlers-Danlos syndrome also occurs in dogs and cats. Affected animals show a greatly increased vulnerability of the skin, with the skin wounds scarring quickly. The elasticity of the skin is greatly increased. The disease is inherited recessively. A thinning of the skin as a result of high cortisol levels ( Cushing's syndrome ) or long-term therapy with glucocorticoids must be clarified in the differential diagnosis . The diagnosis is confirmed using an electron microscope. Treatment is not possible.
EDS in films and series
- The seemingly alcoholic teenage Emma in episode 4 of the 13th season ( Free Fall ) of the series Grey's Anatomy has EDS.
- In episode 18 of season 7 of Dr House , Nina has messie syndrome as a result of miscarriage from EDS.
- In episode 3 of Season 3 of Atlanta Medical , the injured criminal is in constant pain from EDS.
- Yvie Oddly , winner of the 11th season of the American reality TV series RuPaul's Drag Race , suffers from EDS.
literature
- Andreas Luttkus: The Ehlers-Danlos syndrome: an interdisciplinary challenge, Walter de Gruyter, 2011, ISBN 978-3-11-024955-2 limited preview in the Google Book Search
- Brat T. Tinkle, Carrie L. Atzinger: 24 Ehlers-Danlos Syndromes in: Suzanne B. Cassidy, Judith E. Allanson: Management of Genetic Syndromes, John Wiley & Sons, ISBN 978-0-470-19141-5, limited preview in Google Book Search
- Brat T. Tinkle: Issues and Management of Joint Hypermobility: A Guide for the Ehlers-Danlos Syndrome Hypermobility Type and the Hypermobility Syndrome, Left Paw Press, 2008, ISBN 978-0-9818360-1-0
Web links
- via EDS (PDF; 270 kB) In: Der Orthopäde. ( Memento from March 6, 2006 in the Internet Archive )
- Recommended action for performing anesthesia in EDS patients (English)
- Help, I have crooked fingers - or the solution to a number of medical phenomena - EDS case study (PDF)
- Ehlers-Danlos syndrome Genetic Home Reference. Federal Government . (English)
- Hypermobility due to weak connective tissue
Patient organizations:
- The Ehlers-Danlos Society
- German Ehlers-Danlos-Initiative e. V.
- Association Suisse des Syndromes d'Ehlers-Danlos
- Federal Association of Ehlers-Danlos Self-Help eV
- Ehlers-Danlos network Switzerland
Individual evidence
- ↑ a b Genetics Home Reference: Ehlers-Danlos syndrome. Retrieved September 23, 2019 .
- ↑ Rare diseases. Federal Ministry of Health , May 17, 2019, accessed on September 24, 2019 .
- ↑ LA Parapia, C. Jackson: Ehlers-Danlos syndrome - a historical review. In: British Journal of Hematology . Volume 141, Number 1, April 2008, pp. 32-35, ISSN 1365-2141 . doi: 10.1111 / j.1365-2141.2008.06994.x . PMID 18324963 .
- ↑ J. v. Meekeren: Een rekkelijke spanjert, Hooft-stuk 29. In Heel en Geneeskonstige Aanmerkingen, van Job vall Meekeren, in sijn leven Heelmeester der Stadt, Admiraliteyt en 't Gasthuys in Amsterdam. 1668, pp. 170-172.
- ↑ TO Chernogubov : A case of cutis laxae. In: Protokoly Moskowskawo Venere-ologitscheskawo i Dermatologitscheskawo Obtschestwa. 1891/1892, 1, pp. 23-29 (cited by Steinmann et al [21]).
- ^ Edvard Ehlers: Cutis laxa. Tendency to hemorrhage in the skin, loosening of several articulations. In: Dermatologische Zeitung , Volume 8, 1901, p. 173 f.
- ↑ H. Danlos: Un cas de cutis laxa avec tumeurs par contusion chronique des coudes et des genoux (xanthome juvénile pseudo-diabétique de MM Hallopeau et Macé de Lépinay) . In: Bull Soc Franc Dermatol Syphiligraph (Paris) . tape 19 , 1908, pp. 70-72 .
- ↑ Peter J. De Coster (Ed.): Oral health in prevalent types of Ehlers-Danlos syndromes. In: Oral Pathol Med. 2005, 34, pp. 298-307.
- ↑ 2017 EDS International Classification. In: The Ehlers Danlos Society. Retrieved September 23, 2019 (American English).
- ↑ a b The Types of EDS. In: The Ehlers Danlos Society. Retrieved September 23, 2019 (American English).
- ↑ HEDGE Study. In: The Ehlers Danlos Society. Retrieved September 23, 2019 (American English).
- ↑ a b hEDS Diagnostic Checklist. In: The Ehlers Danlos Society. Retrieved September 23, 2019 (American English).
- ^ Tinke et al .: Hypermobile Ehlers-Danlos syndrome (aka Ehlers-Danlos syndrome Type III and Ehlers-Danlos syndrome hypermobility type): Clinical description and natural history (pages 48-69). In: Ehlers-Danlos Society. American Journal of Medical Genetics, 2017, accessed September 23, 2019 .
- ↑ Chopra et al .: Pain management in the Ehlers – Danlos syndromes (pages 212-219). In: Ehlers-Danlos Society. American Journal of Medical Genetics, 2017, accessed September 23, 2019 .
- ↑ a b c d e f g h i j k Brady et al .: Ehlers-Danlos Syndomes, rarer types. In: Ehlers-Danlos Society. American Journal of Medical Genetics, 2017, accessed September 23, 2019 .
- ↑ Bowen et al .: Ehlers-Danlos syndrome, classical type. In: Ehlers-Danlos Society. American Journal of Medical Genetics, 2017, accessed September 23, 2019 .
- ^ Byers et al .: Diagnosis, natural history and management in vascular Ehlers-Danlos Syndrome. In: Ehlers-Danlos Society. Americal Journal of Medical Genetics, accessed September 23, 2019 .
- ^ The Types of EDS. In: The Ehlers Danlos Society. Retrieved September 23, 2019 (American English).
- ^ Fransiska Malfait, MD, PhD, Richard Wenstrup, MD, and Anne De Paepe, MD, PhD: Ehlers-Danlos Syndrome, Classic Type. In: ncbi.nlm.nih.gov. May 29, 2007, accessed May 12, 2015 .
- ↑ a b c d e f g Ehlers-Danlos syndrome in: Peter Altmeyer : Encyclopedia of Dermatology, Allergology, Environmental Medicine
- ↑ Martin Röcken: 19 Genetic Diseases of Connective Tissue. Thieme, 2010, p. 200.
- ^ Ehlers-Danlos syndrome. In: Online Mendelian Inheritance in Man . (English)
- ↑ COL5A3 collagen, type V, alpha 3 [Homo sapiens (human)] - genes - NCBI. In: ncbi.nlm.nih.gov. November 12, 2014, accessed December 28, 2014 .
- ↑ Harold Chen: Atlas of Genetic Diagnosis and Counseling. Humana Press, 2006, ISBN 1-60327-161-9 , p. 342. Restricted preview in Google book search
- ^ Howard P Levy: Ehlers-Danlos Syndrome, Hypermobility Type. In: ncbi.nlm.nih.gov. October 22, 2004, accessed December 28, 2014 .
- ↑ S. Premalatha, KN Sarveswari, K. Lahiri: Reverse Namaskar: a new sign in Ehlers-Danlos syndrome: a family pedigree study of four generations. In: Indian journal of dermatology. Volume 55, number 1, 2010, pp. 86-91, doi: 10.4103 / 0019-5154.60360 , PMID 20418985 , PMC 2856381 (free full text).
- ↑ Ehlers-Danlos syndrome
- ↑ COL3A1 Symbol Report - HUGO Gene Nomenclature Committee. In: genenames.org. Retrieved December 28, 2014 .
- ↑ Andreas Luttkus: The Ehlers-Danlos Syndrome: An Interdisciplinary Challenge , 2017, ISBN 3-11-047396-8 .
- ^ DP Germain: Ehlers-Danlos syndrome type IV. In: Orphanet Journal of Rare Diseases. Volume 2, 2007, p. 32, doi: 10.1186 / 1750-1172-2-32 , PMID 17640391 , PMC 1971255 (free full text) (review).
- ^ Nevo syndrome. In: Orphanet (Rare Disease Database).
- ↑ Phenotypic variability of the kyphoscoliotic type of Ehlers-Danlos syndrome (EDS VIA): clinical, molecular and biochemical delineation In: Orphanet Journal of Rare Diseases. , Volume 6, 2011, p. 46, ISSN 1750-1172 . doi: 10.1186 / 1750-1172-2-32 . PMID 17640391 . PMC 3135503 (free full text). (Review).
- Jump up ↑ A. Kariminejad, B. Bozorgmehr, A. Khatami, MH Kariminejad, C. Giunta, B. Steinmann: Ehlers-Danlos Syndrome Type VI in a 17-Year-Old Iranian Boy with Severe Muscular Weakness - A Diagnostic Challenge? In: Iranian journal of pediatrics. Volume 20, Number 3, September 2010, pp. 358-362, PMID 23056730 , PMC 3446046 (free full text).
- ↑ M. Nakajima (Ed.): Mutations in B3GALT6, which Encodes a Glycosaminoglycan Linker Region Enzyme, Cause a Spectrum of Skeletal and Connective Tissue Disorders . Laboratory for Bone and Joint Diseases, Center for Integrative Medical Sciences, RIKEN, Tokyo 108-8639, The American Society of Human Genetics.
- ↑ About rare diseases . Eurodis - Rare Diseases Europe, January 9, 2012.
- ^ Fransiska Malfait: Ehlers-Danlos Syndrome, Classic Type. In: ncbi.nlm.nih.gov. May 29, 2007, accessed December 28, 2014 .
- ^ Howard P Levy: Ehlers-Danlos Syndrome, Hypermobility Type. In: ncbi.nlm.nih.gov. October 22, 2004, accessed December 28, 2014 .
- ↑ Karin Mayer: Ehlers-Danlos Syndrome (EDS) Type IV (Q79.6) ( Memento from February 26, 2014 in the Internet Archive ). Center for Human Genetics and Laboratory Medicine, Martinsried, January 9, 2012.
- ^ The Types of EDS. In: The Ehlers Danlos Society. Retrieved September 24, 2019 (American English).
- ↑ Julia Sander, Werner Plötz: Barmherzige Brüder Hospital in Munich cares for patients with the rare Ehlers-Danlos syndrome. In: Order magazine Misericordia. Edition 11/08
- ↑ Malfait et al .: The 2017 International Classification of the Ehlers – Danlos Syndromes. In: Ehlers-Danlos Society. American Journal of Human Genetics, 2017, accessed September 27, 2019 .
- ↑ EurordisCare2: Study on diagnosis times using the example of eight rare diseases, Report on the European Conference on Rare Diseases 2005, p. 26.
- ↑ Britta Berglund, Mattiasson Anne-Cathrine, Ingrid Randers: Dignity not fully upheld when seeking health care: experiences expressed by individuals suffering from Ehlers-Danlos syndrome . In: Disability and Rehabilitation . tape 32 , no. 1 , 2010, ISSN 0963-8288 , p. 1-7 , doi : 10.3109 / 09638280903178407 , PMID 19925271 .
- ↑ So You Think You Might Have EDS? ( Memento of August 27, 2013 in the Internet Archive ) (PDF; 241 kB) Ehlers-Danlos National Foundation
- ↑ Ehlers-Danlos-Self-Help e. V .: Statistical questionnaire - symptom catalog 1st edition. September 2011.
- ↑ a b theilcfoundation.org (PDF)
- ↑ A. Castriota-Scanderbeg, B. Dallapiccola: Ehlers-Danlos Syndromes (PDF) In: Abnormal Skeletal Phenotypes - From Simple Signs to Complex Diagnoses. Springer, p. 695. PDF file, p. 2.
- ↑ Robert Ploier: Differential Diagnoses in Child and Adolescent Medicine. P. 290 Limited preview in Google Book search
- ↑ Mitakides et al .: Oral and Mandibular Manifestations in the Ehlers-Danlos Syndromes. In: Ehlers-Danlos Society. American Journal of Medical Genetics, 2017, accessed September 27, 2019 .
- ↑ a b c Fikree et al .: Gastrointestinal Involvement in the Ehlers-Danlos Syndromes. In: Ehlers-Danlos Society. American Journal of Medical Genetics, 2017, accessed September 27, 2019 .
- ↑ Hakim et al .: Chronic Fatigue in Ehlers-Danlos Syndrome - Hypermobile Type. In: Ehlers-Danlos Society. American Journal of Medical Genetics, 2017, accessed September 27, 2019 .
- ^ Chiari malformation. In: dr-virella.com. Archived from the original on December 4, 2014 ; accessed on December 28, 2014 .
- ↑ Tethered Spinal Cord in Patients with Vascular Ehlers-Danlos Syndrome. In: ashg.org. Retrieved December 28, 2014 .
- ^ Dysautonomia International - Underlying Causes of Dysautonomia. In: dysautonomiainternational.org. Retrieved December 28, 2014 .
- ^ I. Robertson: Keratoconus and the Ehlers-Danlos syndrome: a new aspect of keratoconus. In: The Medical journal of Australia. Volume 1, Number 18, May 1975, pp. 571-573, ISSN 0025-729X . PMID 1143149 .
- ↑ RK MISHRA, IB GOEL: DISCIFORM MACULAR DEGENERATION ASSOCIATED WITH EHLERS-DANLOS SYNDROME. (CUTIS HYPERELESTICA). In: Journal of the All-India Ophthalmological Society. Volume 11, December 1963, pp. 87-95, ISSN 0044-7307 . PMID 14098999 .
- ↑ NC Voermans, H. Knoop u. a .: Pain in Ehlers-Danlos syndrome is common, severe, and associated with functional impairment. In: Journal of pain and symptom management. Volume 40, Number 3, September 2010, pp. 370-378, ISSN 1873-6513 . doi: 10.1016 / j.jpainsymman.2009.12.026 . PMID 20579833 .
- ↑ NC Voermans, H. Knoop u. a .: Pain in Ehlers-Danlos syndrome is common, severe, and associated with functional impairment. In: Journal of pain and symptom management. Volume 40, Number 3, September 2010, pp. 370-378, ISSN 1873-6513 . doi: 10.1016 / j.jpainsymman.2009.12.026 . PMID 20579833 .
- ↑ a b c Linda Stapleford Bluestein: Pain Management in Patients With Hypermobility Disorders: Frequently Missed Causes of Chronic Pain. In: Topics in Pain Management. 2017, accessed on June 10, 2019 .
- ↑ Ehlers – Danlos syndrome ( Memento from April 15, 2015 in the Internet Archive ) OrphanAnesthesia - a project of the German Society for Anesthesiology and Intensive Care Medicine eV (PDF, German and English)
- ↑ T. Wiesmann, M. Castori et al. a .: Recommendations for anesthesia and perioperative management in patients with Ehlers-Danlos syndrome (s). In: Orphanet Journal of Rare Diseases. Volume 9, 2014, p. 109, ISSN 1750-1172 . doi: 10.1186 / s13023-014-0109-5 . PMID 25053156 . PMC 4223622 (free full text).
- ↑ Babak Abai, Dena Thayer, Paul M. Glat: Two Cases of Traumatic Wounds in Patients With Ehlers-Danlos Syndrome Successfully Treated With a Bioengineered Skin Equivalent. In: Wounds. Volume 15, Number 6, 2003.
- ↑ A. De Paepe, F. Malfait: Bleeding and bruising in patients with Ehlers-Danlos syndrome and other collagen vascular disorders. In: British Journal of Hematology . Volume 127, Number 5, December 2004, pp. 491-500, ISSN 0007-1048 . doi: 10.1111 / j.1365-2141.2004.05220.x . PMID 15566352 . (Review).
- ↑ Elizabeth Russell: The Ehlers-Danlos Syndromes: An Overview. In: FACP The Pain Practitioner. Volume 16, No. 2, pp. 27-39.
- ^ Pradeep Chopra, Brad Tinkle, Claude Hamonet, Isabelle Brock, Anne Gompel: Pain management in the Ehlers – Danlos syndromes . In: American Journal of Medical Genetics Part C: Seminars in Medical Genetics . tape 175 , no. 1 , 2017, ISSN 1552-4876 , p. 212–219 , doi : 10.1002 / ajmg.c.31554 ( wiley.com [accessed October 5, 2019]).
- ↑ L. Remvig, DV Jensen, RC Ward: Are diagnostic criteria for general joint hypermobility and benign joint hypermobility syndrome based on reproducible and valid tests? A review of the literature. In: The Journal of Rheumatology . Volume 34, 2007, pp. 798-803, ISSN 0315-162X . PMID 17295436 . (Review).
- ^ R. Keer, R. Grahame: Hypermobility Syndrom - Recognition and Management for Physiotherapists. ISBN 978-0-7506-5390-9 , p. 20.
- ↑ Irman Forghani: Updates in Clinical Genetics and Aspects of hypermobile Ehlers Danlos Syndrome . In: Balkan Medical Journal . tape 36 , no. 1 , January 2019, ISSN 2146-3123 , p. 12–16 , doi : 10.4274 / balkanmedj.2018.1113 , PMID 30063214 , PMC 6335943 (free full text).
- ^ I. Bjelland, GS Tell u. a .: Folate, vitamin B12, homocysteine, and the MTHFR 677C-> T polymorphism in anxiety and depression: the Hordaland Homocysteine Study. In: Archives of general psychiatry. Volume 60, Number 6, June 2003, pp. 618-626, ISSN 0003-990X . doi: 10.1001 / archpsyc.60.6.618 . PMID 12796225 .
- ↑ L. Yan, L. Zhao et al. a .: Association of the maternal MTHFR C677T polymorphism with susceptibility to neural tube defects in offsprings: evidence from 25 case-control studies. In: PloS one. Volume 7, number 10, 2012, p. E41689, ISSN 1932-6203 . doi: 10.1371 / journal.pone.0041689 . PMID 23056169 . PMC 3463537 (free full text).
- ↑ Homocystinuria due to methylene tetrahydrofolate reductase deficiency. In: Orphanet (Rare Disease Database).
- ↑ ZH Shagirova, LN Ushenkova u. a .: [Investigation of detoxification polymorphisms genes, methylene tetrahydrofolate reductase (MTHFR) and P53 in the radiosensitive human cells]. In: Radiatsionnaia biologiia, radioecologiia. Volume 50, Number 2, 2010 Mar-Apr, pp. 128-133, ISSN 0869-8031 . PMID 20464958 .
- ↑ Talking with MTHFR Expert Dr. Ben Lynch - Show Timeline. In: gapsdietjourney.com. June 11, 2012, accessed December 28, 2014 .
- ↑ mthfrheds.com. February 12, 2010, accessed December 28, 2014 .
- ↑ Ch. Noli and F. Scarampella: Ehlers-Danlos syndrome . In: Practical Dermatology in Dogs and Cats . Schlütersche Verlagsanstalt, 2nd edition 2005, pp. 331–332. ISBN 3-87706-713-1
- ↑ Falling Slowly. Retrieved July 1, 2020 .
- ↑ myFanbase team: Dr. House: # 7.18 Truths spilled. Retrieved July 1, 2020 .
- Jump up ↑ Atlanta Medical - Season 3 Episode 3: Holy Sinner. June 24, 2020, accessed on July 1, 2020 (Austrian German).
- ↑ Yvie Oddly wins RuPaul's Drag Race 11 and the hearts of the EDS community. In: The Ehlers Danlos Society. May 30, 2019, Retrieved July 27, 2020 (American English).