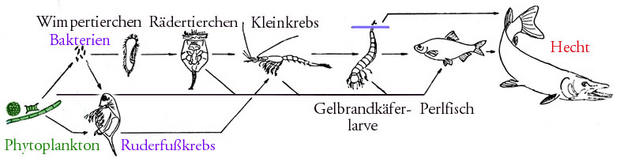
Food chain
A food chain is a model for the linear energetic and material relationships between different types of living beings , each type of food being a different type, with the exception of the type at the end of the food chain. One also speaks of trophic relationships (derived from the Greek trophein "to feed"). At the beginning of the food chain there are representatives of the producers (especially plants ), followed by consumers ( herbivores and carnivores ). At the end of such a chain there is often a top predator . Food chains are examined in ecology and are also of importance in ecotoxicology .
Food chains are simplified models. Most plants and animals are several other species for food and most consumers feed on several other species. Omnivores feed on both plants and animals. After their death, top predators are the food source for scavengers or destructors - as are many organisms of the other levels. The trophic relationships in real ecosystems are therefore much more complex and are known as the food web.
Trophy levels
Food chains are often mentally structured according to trophic levels . A trophy level comprises all organisms (or species) with the same position in the food chain. The lowest trophy level are the producers who do not feed on other organisms, but can serve as food for them (usually green plants). All organisms that are not producers can be summarized as consumers who need other organisms for nutrition. Most consumers are divided into primary consumers, i.e. H. Herbivores (also called herbivores or phytophages) and secondary consumers, or "robber" (also carnivores, predators , Zoophagen or carnivores called). Predators that capture secondary consumers are called tertiary consumers. The top level of trophies are top predators (or top predators), which do not or only exceptionally serve as food for other organisms. The organisms lying “between” the phytophagous and the top predators feed on other organisms (they are consumers), but also serve as food for others. Depending on their complexity, ecosystems have a different number of trophic levels, but not an unlimited number.
While the position of the herbivores can still be defined relatively clearly, the role of the predators is more difficult to grasp because they usually feed on different organisms that can have different positions in the food chain. For example, hawks can prey on pigeons (pigeons are seed-eaters , i.e. phytophages). The food chain then has three links, with the hawk in third position. Titmice also belong to their range of prey ; these are insectivores , so they are predators. Here the hawk would be in the fourth position (plant> insect> tit> hawk), or even in fifth if the insect captured by the tit was also a predator. A species can therefore have more than one trophy level. The trophic level is usually determined according to the mean importance of the food relationships for the species in question. The omnivores ("omnivores") are also problematic for the determination of the trophic level , they feed on both plants and as predators. Cannibalistic species are also problematic , in which old animals eat young animals of the same species, for example. Usually parasites are also disregarded when defining the trophic levels.
Food webs
A food chain in an ecosystem is usually a conceptual abstraction because species can feed on several types of prey and can themselves fall victim to different types of predators. Food chains are therefore usually branched out, one speaks of the food web .
The study of food webs in real ecosystems is a difficult ecological problem because of the extreme complexity that can already occur in species-poor ecosystems. In addition, all populations in the ecosystem are subject to constant fluctuations which, via the dependencies in the food web, have a direct or indirect effect on the populations of several other species and influence their food choices. A mathematical description of the population dynamics , as is possible for individual predator-prey relationships in the Lotka-Volterra equations , can only be applied to a limited extent to food webs. In addition to simplifying assumptions, investigations in artificially simplified ecosystems, the so-called mesocosms , are also used.
A distinction is made between “ cumulative ” networks, which include all types of which trophic relationships are known, and networks at specific points in time. These are usually much smaller than cumulative networks, but more difficult to capture. Networks determined cumulatively tend to overestimate the strength of the interactions between the species involved (the “coupling density”).
Saprophage and live-eater subsystems
Much of the plant biomass in real ecosystems is not consumed by herbivores, but remains until the plant or its organs die. For example, in a forest ecosystem, most of the production occurs as fallen leaves or dead wood . This dead substance is also eaten by specialized users called saprophages . Ultimately, the dead biomass (apart from small, possibly permanently stored residues) is broken down again into inorganic components ( mineralized ) by bacteria and fungi , the destructors . Since the destructors are more energetic than the plant litter itself, they are usually of decisive importance for the nutrition of the saprophages. The saprophages and destructors also have their predators. As a result, there are usually two different food chains in real ecosystems, both of which originate from the producers, but do not otherwise have to be directly linked. Typical of the saprophage system are tight cycles or loops, which arise from the fact that the same biomass can be eaten several times in succession by different saprophages (this is possible because the biomass of the destructors contained therein is decisive in terms of energy). The food chains emanating from the herbivores and the saprophages can be linked by common predators. One observes z. B. in forests that insectivorous birds such as titmice are predominantly from insects such. B. feed mosquitoes that belong to the saprophage system (via their larvae). When butterfly caterpillars (herbivores) multiply on a large scale, however, they shift to these as main food.
When determining the length of a food chain, a strict distinction is usually made between the subsystems, although the dead biomass of herbivores and predators is ultimately also included in the saprophage chain (e.g. by scavengers ). The main reason for this is that herbivores and their predators can have a direct effect on green plants and thus on the production of the ecosystem. Saprophages, on the other hand, cannot influence the amount of dead biomass (such as plant litter). One speaks here of “donor-controlled” systems.
Energy and material turnover
The trophic relationships in food chains cause material and energy sales , which are ultimately based on the primary structure of organic matter ( biomass ) by the producers ( net primary production ). The part of the energy turnover of an ecosystem that takes place via the food chains is therefore limited by the production of the ecosystem. Since the consumers maintain their metabolism with the energy they consume (see respiration , "breathing"), the system loses part of the usable energy in the form of heat or low-energy waste products with every step in the turnover along the food chain .
From a chemical point of view , it should be noted that substances generally referred to as “high-energy” do not store energy per se . The ability of an organism to do work by consuming substances such as sugar or cellulose, i.e. H. Being able to drive their metabolism (see definition of energy ) depends equally on the availability of electron acceptors . Molecular oxygen O 2 plays this role in many land and water ecosystems . Only certain (archae) bacteria can use other electron acceptors in the absence of air and a lack of oxygen ( anaerobia ). One example is the carbonate respiration of methane generators , in which carbon dioxide (CO 2 ) acts as the ultimate electron acceptor.
Along the food chain, consumers also take in the vast majority of the nutrients they need to build up their own biomass. The carbon turnover is directly linked to the energy turnover. Nitrogen has to be included as part of proteins . Phosphorus is required for numerous vital biological functions, larger amounts e.g. B. for the synthesis of DNA and RNA . A balanced intake of these elements can be a problem for herbivores, for example. Wood , for example, contains a lot of fundamentally usable energy in the form of the reduced carbon of cellulose , but generally only little usable nitrogen or phosphorus.
For the pure function of the simplest conceivable ecosystem, producers (who build up biomass) and destructors (who mineralize them again) would be sufficient. The presence of consumers can strongly influence production and many other structural parameters of an ecosystem and, under certain circumstances, “ control ” them. The type and formation of food chains can thus play a key role in shaping the structure and function of an ecological system. The search for rules and regularities of such influencing is an essential field of ecological research. Depending on the research question, the energy and material turnover itself, or the relationships and the structure of the organisms and species involved in the food web, may be the focus of interest.
- In the simplest case, only the trophic relationship of the different species is considered (usually drawn graphically as a connecting line or arrow). In this case one can see which species feed on which other species. Nothing is known about the meaning and effects of the relationship. Food chains and food webs in their simplest form are called topological networks.
- If the focus is on the energy turnover, one tries to quantitatively determine the turnover rates associated with the various relationships. Result could e.g. For example, a plant may be eaten by 20 herbivores, but one species is responsible for most of the feeding. For such an energetic food web, the turnover rate of every single connection of the network has to be determined, in practice an almost impossible task. In most cases, the researchers limit themselves to determining the strength of a few connections after they have tried beforehand with preliminary investigations and plausibility considerations to estimate what are probably the most important connections in the network. In the representation of an energetic food web z. B. the conversion rate can be represented by different line thicknesses. Key types in an energetic network are those types through which a large part of the energy turnover occurs.
- If there is particular interest not in the total turnover but in the individual species themselves, an attempt is made to grasp the interaction of the species involved in the network by modeling their population sizes. So you can see the different roles of different species, the influence of biodiversity and the like. try to estimate. A food web modeled in this way is called an interaction web. It regularly turns out that relationships that appear negligible when viewed from an energetic point of view can be functionally very important. Loops and feedback are particularly important for interaction networks. So z. For example, a particular herbivore may only be of minor importance in a system because its density is severely limited by a predator. If the influence of the predator ceases to exist (e.g. through hunting), completely different material flows in the system can result.
Depending on the question, instead of the energy stored and passed on in the food chain, individual substances or substance flows (carbon, nitrogen, etc.) are also examined as part of energy flow measurements . Due to the destructors (fungi, bacteria, but also worms, articulated animals, etc., involved in mechanical decomposition), which take their food from all other trophic levels and thereby return the substances from the food chain, there is no chain for the substances, but a material cycle ( see e.g. nitrogen and carbon cycle ).
Energetic and allometric restrictions on food chains and food webs
The English zoologist Charles Sutherland Elton and the American limnologist Raymond Laurel Lindeman are fundamental for the energetic consideration of food chains . The concept of the food pyramid ( Elton's number pyramid) goes back to Elton , which, as an energy pyramid, characterizes the various trophic levels of an ecosystem. As a result of the inevitable losses of energy with every consumption process, less energy is available for each trophic level than for the one below. As a rule of thumb, a remaining portion of 10% is assumed (i.e. a loss of 90%), which is in the order of magnitude numerous studies has been confirmed. This exponential loss of energy limits the length of the possible food chains, because at some point there will not be enough energy left for another trophic level. Furthermore, the length of the chains should depend on the productivity of the ecosystem under consideration. In actually examined ecosystems, the typical length of the food chains (in the sense defined above!) Is between two and five, almost always either three or four. Relationships with productivity were broadly confirmed.
The aforementioned form of the energy pyramid only applies in this form to the actual production of an ecosystem or its individual trophic levels. If one looks at the (much more easily measurable) existing biomass instead of production, the relationships become more complex. The (probable) main reason was found to be the influence of the proportions of the organisms involved. The constraints that a growing organism is subject to for purely physical and physiological reasons are called allometrics . Empirically found correlations with increasing size (scale laws, best known is Kleiber's law ) have been integrated into ecological theory through recent research.
If a trophic level is composed of large organisms, the biomass increases at the same production rate, and not linearly, but exponentially (there is still no agreement on the actual size of the exponent). Since larger organisms convert (and have to convert) energy more slowly per unit of body mass, the (per area) productivity of the system as a whole hardly changes at all. The large organisms thus have more biomass, but their production is not higher. This connection has been confirmed over more than 12 orders of magnitude (e.g. planktonic algae to forest trees). Naturally, it applies equally to consumers (e.g. grazing ungulates versus caterpillars). The constant (and only limited by the resources and the available energy) productivity of ecosystems, regardless of the species that make up them, has already been established earlier (one example is the carefully examined ecosystems of the Solling project ), but was still possible at that time cannot be explained.
Because of these connections there are "inverted" pyramids
- for the number of individuals when the organisms of the higher trophic level are smaller than those of the lower;
- for the biomass, when the organisms of the higher trophic level are larger than those of the lower one.
Upside down productivity pyramids by themselves are impossible.
Since the influences of body size cancel each other out per area, the length of the food chains in an ecosystem does not depend on the size of the organisms involved.
Patterns and processes in real food webs
The great advances in understanding the function of numerous ecosystems have shown that the effect on systems mediated by food chains and food webs is extremely complex and very different between different systems.
Differences between aquatic and terrestrial food webs

( McMurdo Sound , Antarctica)
The dominant producers in the sea and in freshwater lakes are unicellular algae ( phytoplankton ). Terrestrial systems are usually based on vascular plants, most commonly trees (forests) or grasses (savannas, steppes, tundras, man-made pastures). This results in u. a. the following differences:
- Size structure: In aquatic food webs, body size increases with the position in the food web (e.g. diatoms - copepods - plankton-eating fish - shark). This rule does not apply in terrestrial systems.
- Ratio of production to biomass: Phytoplanktons have a much lower biomass than land plants with comparable productivity per area.
- Nutrient balances: Land plants largely consist of supporting tissues such as wood and fibers, which are poor in nutrients and are therefore difficult to use by phytophages.
- Repellants against phytophagi: Land plants have a rich repertoire of "secondary" plant substances to repel them (e.g. alkaloids and glycosides) or mechanical repellants such as thorns. This is much less common in aquatic systems. Most planktonic algae can be used equally by all planktivores (with the exception of size effects).
- Heterogeneity: Land habitats are usually more spatially structured and usually consist of numerous compartments (subsystems, each with their own living conditions).
Overall, these differences show that the influence of phytophages on producers in aquatic systems tends to be higher (there are numerous exceptions and counterexamples!). Presumably more aquatic systems are decisively shaped by the influence of the phytophages and the predators acting on them, and more terrestrial systems by the producers. In forest ecosystems in particular, the influence of herbivores turned out to be surprisingly small.
Trophic cascades
Trophic cascade is the name given to the indirect effect that predators can exert on primary producers by limiting the population density of herbivores (phytophages). If such a trophic cascade is effective in an ecosystem , the production of plant biomass is high and the phytophage density is relatively low. This is not due to the fact that the latter are unable to increase the growth rates of their populations, but is due to the fact that their population density is kept low by the predators. This becomes clear when the predators are removed (e.g. experimentally): the herbivore density increases sharply and primary production drops sharply. If a trophic cascade is effective, the actual structure of the ecosystem is determined via the food chain. Trophic cascades have been demonstrated in a number of ecosystems. Nevertheless, there is no consensus on their general meaning. The spectrum of opinion ranges from "core thesis of ecology" to "rare exception."
Key types
In a classic study, the American ecologist RTPaine observed on the rocky coast of the Pacific that, in otherwise identical places, completely different communities can arise, depending on whether a predatory starfish species is present or not. This predatory species is therefore a key species (keystone predator ) for the system. Since this study, such key species have been searched for in other systems and in some cases found. In numerous other ecosystems, however, the search for key species was unsuccessful.
Case studies
Wolves, elk and beavers in Yellowstone National Park
In the American Yellowstone National Park the wolves were persecuted in the first decades of the 20th century and exterminated until 1926 in order to secure the population of the bison. As a result, an extreme density of elk deer was observed in the area , which in some regions stood densely like cattle on pasture. After attempts to limit the density of the elk by shooting failed, the wolf was reintroduced and placed under protection in 1995. The following effects now occurred:
- Along the banks of the river, poplar thickets grew instead of grass in places. Closer investigations showed that the places are confusing. These were now evidently avoided by the elk, who had previously suppressed the poplars by eating the seedlings. The decisive factor here was obviously not so much the density limitation of the wapiti population by the predator wolf's feeding pressure (as expected from the textbook), but simply the wapitis’s fear of wolves, i.e. a change in behavior. Such indirect effects are highly significant in numerous ecosystems, but are often neglected due to the focus of ecosystem research on production and energy turnover.
- Lured by the poplar thickets as a food resource, the beaver, which became extinct in the national park, began to migrate back into the area and soon reached high densities. It was shown that the presence of the predator wolf via indirect effects (quasi: around two corners) is decisive for the occurrence of the beaver. Nobody could have predicted this.
- Beavers and elk taken together can eventually suppress the softwood forests again.
Ungulates in the Serengeti
The Serengeti National Park in East Africa is world famous for the large herds of ungulates, from small gazelles to buffalo and rhinoceros. The interactions between the large grazers and vegetation has been explored for decades. In the Serengeti, too, the balance between trees and grasses is shaped by the grazers. The trees went z. B. fell sharply after the reintroduction of the elephants (in 1951). Surprisingly, the decline in wildlife density caused by the foot-and-mouth disease (brought in with cattle) did not lead to an expansion, but to a decline in the tree population. The reason here was the influence of fire. Due to the declining grazing, a lot of dead biomass accumulated, which led to stronger fires. By excluding the grazers with fences on the trial plots, it was found that the productivity of the grasslands was higher with grazing animals than without them. This was due on the one hand to the accelerated nutrient cycles, in particular the faster supply of nitrogen through urine and faeces (60% increase in production), and on the other hand to the fact that without grazing animals, higher grass species with more supporting tissue and lower productivity increased. The overall productivity of the Serengeti is not limited by the grazing animals, but by the rainfall.
Food web in the Hubbard Brook Experimental Forest
One of the most extensive and careful long-term studies in an ecosystem worldwide has been running since 1963 in the Hubbard Brook Experimental Forest in the Hubbard Brook Valley ( White Mountains , New Hampshire, USA). It is one of the few systems in which an attempt was made to quantify all essential energy and material flows. The results show that in this deciduous forest around 0.8% of the irradiated solar energy is used photosynthetically to build up plant biomass . About half of the built up biomass was lost again directly through breathing of the trees, so that the actual increase (the net primary production ) was about 0.4% of the irradiation. Production is divided into above-ground (leaves and stems) and underground production (roots and substances released by the roots). Since it is a deciduous deciduous forest, all newly formed leaves die in the same year. Of the leaf biomass formed annually, an average of about 99% was shed as fall leaves and thus enter the saprobiont food chains. Approx. An average of 1% was used by phytophagi , of which caterpillars are the most important in this forest. Similar orders of magnitude have been observed in most of the forests studied so far (e.g.). The Hubbard Brook study showed, however, that the ratios in individual years can differ significantly from the average values. With a mass increase in leaf-eating butterfly caterpillars, the proportion of leaf biomass consumed could increase to 40%. The butterfly caterpillars actually take up about 14% of the eaten leaf biomass, regardless of the year, the rest is v. a. lost as feces . Of the 14% absorbed, around 60% are inhaled in the metabolism, while around 40% are used for growth.
Among the vertebrates, songbirds are the most important predators of the butterfly caterpillars. Overall, birds consume about 0.2% of the annual net primary production in this forest. Birds, as small, warm-blooded organisms, have a higher resting metabolism than caterpillars. They therefore use almost 98% of the food they eat for their metabolism. So only 2% are used to build up their own biomass. About 15 species of breeding birds live permanently in the Hubbard Brook Test Forest. Another 10–12 species occur irregularly. The vast majority of species are migratory birds. The density of breeding birds at the beginning of the regular investigations at the end of the 1960s was approx. 20 animals / ha. The density dropped to 7.5 to 9 animals / ha by the mid-2000s. The decrease is mainly due to the fact that 3 previously common species have disappeared. Like all important predators in this forest (e.g. mice, squirrels and chipmunks, shrews, salamanders, centipedes, spiders and beetles) the songbirds are opportunistic, unspecialized predators, which, depending on what is available, can prey on soil animals of the saprobiotic chain as well as herbivores. In addition, seeds also play a role, so the birds are partly phytophagous themselves .
In years with mass reproduction of butterfly caterpillars, the density of breeding birds increased very sharply. Experimental food shortages or additions could also show that the food supply was a major factor in the bird frequency. Another important factor was the predation of eggs and nestlings, to which 17 to 42% of the nests fell victim, depending on the year. The main nest predators were squirrels. Since the density of the squirrels depended on the seed supply of the forest trees (above all the beech) , the breeding success of the songbirds fell sharply in the fattening years , although the birds themselves are partially seed-eaters. The predation of adult birds occurs, but is meaningless from the perspective of the food web.
As a result of many years of research, one could say: In this system, herbivores are already of little importance in terms of material and energy flows, and predators are largely meaningless. Food chains with a length of more than three links are rarely found above ground or are insignificant. However, this judgment is only based on a quantitative-energetic consideration of the food web. The question of how to control the structure of the trophic relationships or the ecosystem under consideration is not answered with this.
Why is the length of the food chain limited?
The biologist Charles Elton already established in the 1920s that food chains are in most cases limited to a length of four to five links, i.e. usually only extend over four to five stations ( trophic levels ). There are two approaches to explain this limitation:
- The energy hypothesis according to the energy loss of trophic trophic level to the limiting factor. Since the low trophic efficiency, i.e. the energy content stored in biomass that is passed on to the next stage, is only 10%, the energy available to each level decreases exponentially. This limits the length of the chain. If there is 100 kg of plant mass in a small pasture area, this is only sufficient to produce 10 kg of herbivore biomass and 1 kg of carnivore biomass . If the primary production in a food chain increases, the chain could, according to this hypothesis, become longer, because the energy is sufficient to supply a further trophic level despite the loss.
- The other approach attributes the restrictive potential to the “dynamic stability” of the food chain. The longer a food chain is, the more unstable it is. Fluctuations in the lower levels intensify upwards and can ultimately lead to the extinction of the top predators. However, if a food chain is shorter, the higher consumers can more easily respond to fluctuations, e.g. B. Environmental disasters, which affect the food supply at all trophic levels. In a very variable food structure, the food chains are therefore shorter.
More recent research takes into account both approaches and tries to elucidate which factors limit the length of the food chains in a given ecosystem and how they interact. Instead of a monocausal explanation, there is a case-specific investigation of various influencing variables. This includes the size of the ecosystem, its composition and its previous development, the frequency and intensity of disruptive events (e.g. periods of drought or the invasion of new species) and the individual predator-prey relationships . For example, the length of the food chains also depends on whether the predators are able to flexibly adapt their eating behavior to the food supply.
Significance for ecotoxicology
The term came into public interest in connection with the observation of an accumulation of pollutants in successive links (food chain or trophic levels). The work most frequently cited in this context is that of Woodwell and co-workers from 1967. In fact, especially fat-soluble and not or only slowly degradable substances (e.g. persistent chlorinated hydrocarbons, heavy metal ions) can accumulate in successive food chain links under certain conditions . In this context one also speaks of biomagnification .
This effect was initially considered to be generally valid, but on the basis of a large number of studies it turned out to be particularly important for air-breathing and / or terrestrial organisms (birds, seals, etc.). Humans can also be subject to this form of pollutant accumulation (e.g. in breast milk). In primary aquatic animals (e.g. water snails, water fleas, fish) in purely aquatic food chains, the direct exchange of substances from the water phase via the epithelia of the organism (the process of so-called bioconcentration into the organism and elimination via gills or skin surfaces out of the organism) is more significant than the biomagnification caused by eating contaminated food, although there are strong differences between the different types of substances and the individual groups of organisms.
In individual cases, a more detailed analysis is only possible by measuring real material fluxes and using suitable compartment modeling . An important property of fat-soluble substances is z. B. the octanol-water partition coefficient ; An important characteristic of the organisms examined is the size of the surface and the permeability of their membranes or epithelia, which enable the exchange of substances with the environment.
Fossil lore
The oldest fossil evidence of a three-part vertebrate food chain comes from the 290 million year old early Permian deposits of the “Humberg Lake” from the Saar-Nahe basin . In the digestive tract of a fossil of the approximately 70 centimeter long Xenacanthiform Triodus , a "freshwater shark ", the larva of a temnospondylic amphibian that had captured a spiny shark was found. This means that three trophy levels are preserved at the same time.
Web links
Individual evidence
- ↑ Overview in: Stuart L. Pimm, John H. Lawton, Joel E. Cohen: Food web patterns and their consequences. In: Nature . 350, 1991, pp. 669-674.
- ↑ See Josef Hofbauer, Karl Sigmund: Evolutionary games and population dynamics. Cambridge University Press, 1998, ISBN 0-521-62570-X .
- ↑ C. Elton: Animal ecology. Sidgwick and Jackson, London 1927.
- ^ R. Lindeman: The trophic-dynamic aspect of ecology. In: Ecology. 23, 1942, pp. 399-418.
- ↑ Review in: James H. Brown: Toward a metabolic theory of ecology. In: Ecology. 85 (7), 2004, pp. 1771-1789.
- ↑ J. Cebrian: Patterns in the fate of production in plant communities. In: American Naturalist. 154, 1999, pp. 449-468.
- ↑ Jonathan B. Shurin, Daniel S. Gruner, Helmut Hillebrand: All wet or all dried up? Real differences between aquatic and terrestrial food webs. In: Proceedings of the Royal Society B. 273, 2006, pp. 1-9.
- ↑ Lennart Persson: Trophic Cascades: abiding heterogeneity and the trophic level concept at the end of the road. In: Oikos. 85, 1999, pp. 385-397. (Full text, PDF; 2.9 MB)
- ↑ Jarrett Byrnes, John J. Stachowicz, Kristin M. Hultgren, A. Randall Hughes, Suzanne V. Olyarnik, Carol S. Thornber: Predator diversity strengthens trophic cascades in kelp forests by modifying herbivore behavior. In: Ecology Letters. 9, 2006, pp. 61-71.
- ↑ Jonathan M. Chase: Are there real differences among aquatic and terrestrial food webs? In: Trends in Ecology and Evolution. 15 (10), 2000, pp. 408-412. doi: 10.1016 / S0169-5347 (00) 01942-X
- ↑ Stephen D. Frettwell: Food chain dynamics. The central theory of ecology. In: Oikos. 50 (3), 1987, pp. 291-301.
- ^ Gary A. Polis, Donald R. Strong: Food web complexity and community dynamics. In: American Naturalist. 147 (5), 1996, pp. 813-846.
- ^ RT Paine: Food web complexity and species diversity. In: American Naturalist. 100, 1965, pp. 65-75.
- ^ RT Paine: A note on trophic complexity and community stability. In: American Naturalist. 103, 1969, pp. 91-93.
- ↑ Barbara L. Peckarsky et al .: Revisiting the classics: Considering nonconsumptive effects in textbook examples of predator - prey interactions. In: Ecology. Volume 89, No. 9, 2008, pp. 2416-2425. doi: 10.1890 / 07-1131.1
- ^ William J. Ripple, Robert L. Beschta: Wolves and the Ecology of Fear: Can Predation Risk Structure Ecosystems? In: BioScience. Volume 54, No. 8, 2004, pp. 755-766, doi : 10.1641 / 0006-3568 (2004) 054 [0755: WATEOF] 2.0.CO; 2 .
- ↑ Bruce W. Baker et al .: Interaction of beaver and elk herbivory reduces standing crop of willow. In: Ecological Applications. Volume 15, No. 1, 2005, pp. 110-118. doi: 10.1890 / 03-5237
- ^ SJ McNaughton, RW Ruess, SW Seagle: Large mammals and process dynamics in African ecosystems. In: BioScience. 38 (11), 1988, pp. 794-800.
- ^ SJ McNaughton, M. Oesterheld, DA Frank, KJ Williams: Ecosystem-level patterns of primary productivity and herbivory in terrestrial habitats. In: Nature . Volume 341, 1989, pp. 142-144.
- ↑ JR Gosz, RT Holmes, GE Likens, FH Bormann: The flow of energy in a forest ecosystem. In: Scientific American. 238 (3), 1978, pp. 92-102.
- ^ TH Fahey et al .: The biogeochemistry of carbon at Hubbard Brook. In: Biogeochemistry. 75, 2005, pp. 109-176.
- ^ Matthias Schaefer: The soil fauna of a beech forest on limestone: trophic structure and energy budget. In: Oecologia. 82, No. 1, 1990, pp. 128-136. doi: 10.1007 / BF00318544
- ^ RT Holmes, FW Sturges: Bird community dynamics and energetics in a northern hardwoods ecosystem. In: Journal of Animal Ecology. 44, 1975, pp. 175-200.
- ^ Richard T. Holmes: Understanding population change in migratory songbirds: long-term and experimental studies of Neotropical migrants in breeding and wintering areas. In: Ibis. 149 (Suppl. 2), 2007, pp. 2-13.
- ^ Neil A. Campbell, Jane B. Reece: Biology. 6th edition. Spectrum academic publishing house, Berlin / Heidelberg 2003.
- ^ David M. Post: The long and short of food-chain length. (PDF) ( Memento from July 28, 2011 in the Internet Archive ). In: Trends in Ecology and Evolution. Volume 17, No. 6, 2002, pp. 269-277. doi: 10.1016 / S0169-5347 (02) 02455-2
- ↑ Michio Kondoh, Kunihiko Ninomiya: Food-chain length and adaptive foraging. In: Proceedings of the Royal Society B: Biological Sciences. Volume 276, No. 1670, 2009, pp. 3113-3121. doi: 10.1098 / rspb.2009.0482
- ↑ George M. Woodwell, Charles F. Wurster Jr., Peter A. Isaacson: DDT residues in an East coast estuary: A case of biological concentration of a persistent insecticide. In: Science . Volume 156, No. 3776, 1967, pp. 821-824. doi: 10.1126 / science.156.3776.821
- ↑ S. Winter, B. Streit: Organochlorine compounds in a three-step terrestrial food chain. In: Chemosphere. 24, 1992, pp. 1765-1774.
- ^ B. Streit: Uptake, accumulation and release of organic pesticides by benthic invertebrates. 3. Distribution of 14 C-atrazine and 14 C-lindane in an experimental 3-step food chain microcosm. In: Arch. Hydrobiol. Suppl. 55, 1979, pp. 374-400.
- ↑ B. dispute: Bioaccumulation of contaminants in fish. In: T. Braunbeck, DE Hinton, B. Streit: Fish Ecotoxicology . Birkhäuser Publ., Basel et al. 1998, pp. 353–387.
- ↑ Volker Storch , Ulrich Welsch , Michael Wink : Evolutionary Biology. 2nd Edition. Springer-Verlag, Berlin / Heidelberg 2007.
- ^ J. Kriwet, F. Witzmann, S. Klug, HJ Heidtke: First direct evidence of a three-level trophic chain in the fossil record. In: Proceedings of the Royal Society B: Biological Sciences. 275, 2008, pp. 181-186. PMC 2596183 (free full text).
- ↑ Eat and be eaten in a primeval lake - unique insights into a 290 million year old food chain. Innovations-report.de, as of November 6, 2007.