Generation change
In biology, a generation change is a form of reproduction in which the two variants of reproduction - the sexual and the asexual - occur alternately in different generations . Generational change occurs in many protists , in red algae , mosses , ferns and seed plants , but also in cnidarians and tunicates . It combines the advantages of both modes of reproduction: the asexually reproducing generation can produce a large number of offspring without the need for a sexual partner. The sexually reproducing generation ensures that the gene pool is mixed .
Variations of the generation change
The generations can differ from one another both in their genetic makeup (the number of their chromosomes as a result of a nuclear phase change ) and in their external appearance. As a rule, generational changes are two-part, i.e. composed of two different generations, but there are also multi-part with more than two different generations:
Heterophasic generation change
In the heterophasic generation change, haploid and diploid generations alternate ( nuclear phase change ). The transition from the haploid to the diploid phase takes place through the union of two cell nuclei of different origins ( karyogamy ), which usually takes place after the union of two gametes (sex cells ). This process is known as fertilization . The transition from the diploid to the haploid phase is the meiosis or reduction division. Living beings with a heterophasic generation change are called diplohaplons (for details see there).
Examples:
- Ferns (often on the underside of the fronds) produce billions of spores in an asexual manner , which grow into a prothallium only a few millimeters in size . Only this generation, which the layman hardly notices, reproduces sexually and in turn produces the well-known "normal fern plant". Ferns therefore have a heteromorphic and heterophasic generation change.
- See also sporophyte and gametophyte and (for red algae ) tetrasporophyte .
- In contrast to ferns, mosses are characterized by the fact that the “normal moss plant” known to everyone does not produce spores, but gametes (sex cells). The generation that produces spores is usually just a low, hair-shaped stand with a spore capsule that grows on the "moss plant". The mosses also have a heteromorphic and heterophasic generation change.
- Plasmodia , the causative agent of malaria , cause the disease-typical episodes of fever due to their ability to reproduce (divide) asexually, but they also have a sexually active generation in alternation with this generation.
Homophasic generation change
There is no change in the degree of ploidy . This variant occurs with multiple generation changes.
A distinction can be made between a haplohomophasic (all forms haploid, except the zygotes) and a diplohomophasic (all forms diploid, except the gametes) generation change.
Metagenesis
Metagenetic generation change is a homophasic generation change that occurs exclusively in different groups of Metazoa (multicellular animals). A generation with bisexual reproduction is always followed alternately by a generation with asexual reproduction , which in turn is followed by a generation with sexual reproduction.
Examples:
- Jellyfish produce sex cells, after which the zygote becomes a larva ( planular larva ). This larva attaches itself to the ground and grows into a polyp . Various jellyfish tie themselves off from him again. This generation change is heteromorphic and homophasic diploid .
- Tapeworms are mostly protandric hermaphrodites (some with self-fertilization) in the ultimate host, but the Finns can rarely reproduce by budding in the intermediate host.
- Trematoda (flukes) are all parasitic metazoa and show an obligatory change of host and generation. Examples include: small liver fluke ( Dicrocoelium dendriticum ) and pair of fluke ( Schistosoma mansoni , the pathogen of schistosomiasis ).
Anisomorphic or heteromorphic generation change
The generations differ in their external appearance.
Examples:
Isomorphic generation change
The generations are outwardly indistinguishable.
Example:
- Ulva lactuca , the sea lettuce
Heterogony
Heterogeneous generation change: One or more generations that reproduce parthenogenetically follow a generation with sexual reproduction.
Examples:
- If the weather is favorable, aphids can multiply extremely quickly in spring, because in most species this happens over several generations by means of parthenogenesis . Only when the environmental conditions are less favorable does a winged, sexually reproducing generation form again, which at the same time represents the form of distribution and is able to fly over long distances to new host plants.
- Human dwarf threadworms have a generation change between parthenogenic female (as a parasite) and a free-living bisexual generation.
- Other examples are water fleas , gall wasps and gall flies .
The generation change in the phylogenetic history of plants
The tribal history of the plants - the transition from fresh water - green algae to mosses and ferns and later to the ginkgo plants , the coniferous plants and the flowering plants - becomes particularly easy to understand if it is viewed in the light of the consequences that a certain form of generation change has .
All eukaryotes that reproduce sexually master a haploid and a diploid phase in the course of their life, caused by meiosis (haploidy) and fertilization (diploidy). For the plants living on land, the reconstruction of their tribal history showed that their life cycle developed from an original dominance of the free- living haploid gametophyte to a dominance of the free- living diploid sporophyte . This evolutionary trend in particular meant that the land plants were able to generate a greater degree of genetic diversity than the more original eukaryotes by means of meiosis and the associated inter- and intrachromosomal recombination as well as subsequent fertilization .
The chandelier algae (Charophyceae or Charales) are currently the closest living relatives of the ancestors of all land plants . There is no generation change in them, because with the exception of the diploid zygote, candelabrum algae are haploid ( haplonts ). However, the land settlement is alternatively traced back to early species of ornamental algae from the group of Streptophyta . From the life cycle of the recent liverworts, in turn, it can be deduced why the dominant diploid generation developed in land plants.
Origins
From the observation that all prokaryotes are haploid, it is deduced that the earliest living things were haploid and that haploidy was the only form of life on earth for more than a billion years. Haploidy is advantageous in that mutations that are harmful to reproduction immediately disappear from the gene pool , since they cannot be masked by an allele . Conversely, diploidy is considered advantageous precisely because recessive alleles - which could be advantageous in the future under changed environmental conditions - are masked and retained in the gene pool. In addition, in multicellular organisms in which numerous mitotic divisions take place, the likelihood of potentially harmful mutations occurring in individual cells or cell lines increases, but this can be compensated for on the basis of the existing alleles - an assumption that also applies in particular to land plants because they are exposed to a significantly higher level of ultraviolet radiation compared to aquatic plants .
Eukaryotes have existed for around two billion years and have not only developed a variety of reproductive variants during this period, but also a large variety of physiological and morphological variants. Among the so-called protists there are, for example, species such as the green alga Chlamydomonas reinhardtii , whose two phases are single-celled and free- living . The haploid phase of the candy algae Chara vulgaris , on the other hand, is multicellular and can be several dozen centimeters long, while the diploid phase consists of a unicellular zygote. In Selaginella denticulata from the genus of moss ferns , both phases are multicellular. Finally, there are numerous derived eukaryotes in which the diploid phase is multicellular and the haploid phase only exists in the form of unicellular gametes (for example in all mammals ).
Although green algae of the genus Coleochaete were used as a model for the development of land plants as early as the second half of the 19th century , it was not until the 1970s and 1980s that precise comparisons of morphological characteristics of algae and land plants confirmed the close ancestral relationship of the under the name Charophyta summarized genera, especially those of the Streptophyta . In addition to the candelabrum algae, u. a. also the recent genera Mesostigma , Chlorokybus and Klebsormidium . In addition, genetic analyzes confirmed the assumption, also formulated in the 19th century, that moss , bear moss plants , ferns and seed plants can be traced back to a common ancestor . From one of these early relatives of today's candelabrum algae, the line of development, according to today's knowledge, went through early relatives of today's deciduous moss to primeval vascular spore plants and finally to seed plants.
The generation change among the mosses
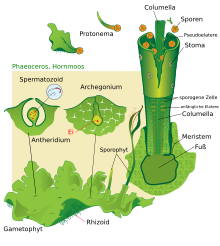
The mosses living today produce flagellated sex cells ( spermatozoids ) in their male sexual organs (the antheridia ) - like the green algae . These have to move in drip liquid water to the egg cells, which are stuck in the female sexual organs (the archegonia ). In contrast to the candelabrum algae, fertilization is not immediately followed by meiosis and thus the return to the haploid phase, but rather a diploid sporogon is formed which ultimately produces spores . This diploid generation, which sits on the mother plant, is therefore called the sporophyte , the haploid mother plant the gametophyte . The sporophyte is usually much smaller than the gametophyte. Mosses are specialists for damp locations because the gametophyte (like algae), unlike the sporophyte, has no protection against evaporation in the form of a cuticle . According to Walter Zimmermann, the development towards a dominant gametophyte has so far cut off any path for the mosses to really large-growing species, since their spermatozoids can only make their way from one plant to the neighboring plant close to the ground.
It is believed that before the emergence of vascular plants , moss was the predominant form of vegetation on land for around 100 million years.
The generation change in ferns

Ferns also produce flagellated sex cells ( spermatozoids ), which have to make their way to the female sexual organs in a liquid. In contrast to the mosses, however, in the ferns the sporophyte (the generation that produces spores) has become predominant in its size. This gave the ferns every opportunity to grow in size, up to 30 meter tall tree ferns . The gametophyte (the generation that produces sex cells), on the other hand, remained connected to the earth in the ferns - similar to the mosses - and indeed resembles liverworts in many fern species (for details see under Prothallium ).
The sexual act becomes independent of free water
In 1896 a Japanese researcher discovered that Ginkgo trees develop flagellated sex cells ( spermatozoids ); a little later, the same phenomenon was also found in the Japanese cycad ( Cycas revoluta ). Both plants can therefore be classified as living fossils and as evidence of biological evolution , of the transition from sperm fertilization to semen formation.
ginkgo
The ginkgo is a dioecious plant, so there are male and female individuals. In the still existing generation change, the ginkgo tree corresponds to the sporophyte of ferns and mosses. However, the spores of the female plants are no longer released into the air, they have rather settled in the sporangium and are relatively large (called megaspores; megaspores are also found in various algae). In addition, the megaspores receive so much nourishment from the ginkgo plants that they form that they can later develop sex cells without independent nutrition . The spores of male plants (called microspores or pollen ) experience their first mitotic divisions in the “microsporangium” (the anther ) and are thus spread to the microgametophyte and as such via the wind. The ginkgo tree (i.e. the sporophyte) excretes the water necessary for the movement of the male sex cells at the micropyle of the ovule itself ( pollination droplets ). The pollen tube that only grows after arriving at the female plant (with ginkgo it mainly serves as an adhesive organ) is nothing more than the rudiment of a gametophyte that once grew independently. The gender generation (the gametophyte ) is extremely reduced in Ginkgo (and in all other seed plants).
In phylogenetic terms, one can say: Inside the megaspore (embryo sac mother cell) an extremely reduced megagametophyte (embryo sac capable of fertilization) develops, which generates a megagamete ( egg cell ) via its megagametangium ( synergids ) . The microspore is blown as a strongly reduced microgametophyte (multicellular pollen grain ) by the wind to the megagametophyte , where it germinates, releases microgametes ( spermatozoids ) and thus fertilizes the megagamet .
More covered
The steady reduction in size of the gametophyte and finally the incorporation of the female gametophyte into its "mother sporophyte" ultimately led to the complete independence of the sexual act from drip-liquid water and perfected the adaptation of the plants to rural life: this can be seen as the most important reason for this that seed plants are the predominant form of life among land plants today.
With some naked samers ( yew and cypress trees ) there are still pollination droplets, but with them the pollen tube grows into the inside of the megaspore. In this way, a male cell nucleus (a sperm that has been reduced to its essentials) can penetrate directly to the egg cell. In the angiosperms the ovules are finally in a carpel included Enclosures which (ie the juvenile sporophytes) also optimizes the diet of the germinating seeds such as its spread by animals. The pollen lands here on a special structure of the carpel, the stigma .
literature
- Walter Zimmermann : History of Plants. An overview. 2nd edition, Munich 1969, dtv Volume 4043. (The sections on mosses, ferns, ginkgo and bedecktsagern essentially follow the argumentation of this source.)
- Klaus Ulrich Leistikow and Franziska Kockel: On the development history of plants - a didactic model. In: Palmarum Hortus Francofortensis (Scientific reports of the Frankfurter Palmengarten), Issue 2, Frankfurt am Main, June 1990 ( table of contents ( memento from July 19, 2011 in the Internet Archive ))
Web links
- Lexicon of Biology : Generational Change . Spectrum, Heidelberg 1999.
- Sexuality in Ginkgo (English)
Individual evidence
- ↑ metagenesis . In: Lexicon of Biology , online edition, 1999.
- ↑ metagenesis . In: Karlheinz Uhlenbrock: Fit for Abitur. Biology high school knowledge. Georg Westermann Verlag, online edition, 2001.
- ↑ Yin ‐ Long Qiu, Alexander B. Taylor and Hilary A. McManus: Evolution of the life cycle in land plants. Review in: Journal of Systematics and Evolution. Volume 50, No. 3, 2012, pp. 171–194, doi: 10.1111 / j.1759-6831.2012.00188.x , full text (PDF)
-
↑ Tomoaki Nishiyama, Hidetoshi Sakayama et al .: The Chara Genome: Secondary Complexity and Implications for Plant Terrestrialization. In: Cell . Volume 174, No. 2, 2018, pp. 448–464.e24, doi: 10.1016 / j.cell.2018.06.033
Solid cell wall prepared for shore leave . On: idw-online.de from July 12, 2018 - ↑ Jan de Vries, Bruce A. Curtis, Sven B. Gould and John M. Archibald: Embryophyte stress signaling evolved in the algal progenitors of land plants. In: PNAS . Volume 115, No. 15, E3471 – E3480, 2018, doi: 10.1073 / pnas.1719230115
- ↑ a b Yin ‐ Long Qiu et al., Evolution of the life cycle in land plants , p. 173
- ↑ Yin ‐ Long Qiu et al., Evolution of the life cycle in land plants , p. 174
- ↑ AH Knoll, EJ Javaux, D. Hewitt and P. Cohen: Eukaryotic organisms in Proterozoic oceans. In: Philosophical Transactions of the Royal Society of London, Series B: Biological Sciences. Volume 361, 2006, pp. 1023-1038, doi: 10.1098 / rstb.2006.1843
- ↑ Yin ‐ Long Qiu et al., Evolution of the life cycle in land plants , p. 177
- ↑ Walter Zimmermann : History of plants. An overview. 2nd edition, Munich 1969, dtv volume 4043
