Flavonoids
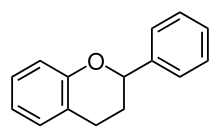
The flavonoids are a group of natural substances to which a large part of the flower pigments belong. Formally, they are derived from the basic body Flavan (2-phenylchroman): two aromatic rings that are connected by a tetrahydropyran ring. There are around 8000 compounds in nature, the diversity of which is created by various oxidation states in the oxygen-containing ring, different substitutions on the aromatic rings and the addition of sugars ( glycoside formation). Biosynthesis takes place via the shikimic acid pathway .
Flavonoids are universally present in plants as secondary plant substances, thus also in human food. They are said to have particularly antioxidant properties. Several plants containing flavonoid are used medicinally.
The flavonoids were discovered in the 1930s by Nobel Prize winner Albert von Szent-Györgyi Nagyrápolt and initially referred to as vitamin P. The "P" in vitamin P stands for " permeability factor ".
Surname
Some plants such as the dyer's oak ( Quercus tinctoria ), the dyer'sweed ( Reseda luteola ) or the dyer's mulberry tree ( Maclura tinctoria ) were used for yellowing in the past. After identifying their ingredients, this group of colorants was called flavones, from the Latin word flavus for yellow. When it was recognized that many of the ingredients have the same structure, but are of a different color or colorless, the group of substances was called flavonoids.
Occurrence
Flavonoids are universal in the plant kingdom and occur in seed plants as well as in mosses and ferns . The formation of flavonoids is known only from a few microorganisms, such as the watering can mold Aspergillus candidus . Animals cannot produce flavonoids. The occurrence in some animal species, for example in the wings of some butterflies , is due to the ingestion of vegetable flavonoids with food and their storage in the body. According to other sources, the flavonoids are limited to the plants.
Structure, diversity and subgroups
The basic structure of the flavonoids consists of two aromatic rings, which are connected by a C 3 bridge. Ring A mostly shows the substitution pattern of phloroglucin , which points to its acetogenin origin. Ring B and the C 3 bridge originate from the shikimic acid pathway , ring B is mostly hydroxylated at 4 ', often also at 3' or 3 'and 5'. In the vast majority of flavonoids (with the exception of chalconies ), the C 3 bridge is closed to form an O-heterocyclic ring (ring C). The basic structure of the flavonoids is therefore the flavan (2-phenylchroman). Ring B is rarely shifted to position 3 (isoflavane, derived from isoflavones ) or 4 (neoflavane).
A total of over 8000 different flavonoids have already been described (as of 2006). The design, especially the degree of oxidation at the C 3 bridge, serves to subdivide the flavonoids into the various subgroups.
Six major subgroups are found in most higher plants: Chalcones , Flavones , Flavonols , Flavandiols , Anthocyanidins, and Condensed Tannins . The aurons are very common, but not ubiquitous. Isoflavones (especially in Fabaceae ) and 3-deoxy-anthocyanidins, which are formed as precursors of phlobaphenes by, for example, Vitis vinifera , Arachis hypogaea and Pinus sylvestris , are restricted to a few groups .
The structural diversity of flavonoids is due to the large number of substitution patterns on rings A and B, as well as to the fact that the flavonoids are usually not free, but rather as glycosides . Over 80 different sugars have been identified. 179 different glycosides have been described for quercetin .
| Subgroup | Basic structure | Examples |
|---|---|---|
| Flavanols |

|
Catechin , gallocatechin , epicatechin , epigallocatechin gallate |
| Flavanols |

|
Taxifolin |
| Chalconies |

|
Isoliquiritigenin , xanthohumol (R2 = OH) |
| Anthocyanidins ( Anthocyanins ) ("Flavenols") |

|
Cyanidin , delphinidin , malvidin , pelargonidin , peonidin , petunidin (R3 = OH) |
| Flavonols |

|
Morin , quercetin (glycoside rutin and methyl ether isorhamnetin ), kaempferol , myricetin , fisetin |
| Aurone |

|
Aureusidin |
| Flavones |

|
Luteolin , apigenin |
| Flavanones |

|
Hesperetin , naringenin , eriodictyol |
| Isoflavones |

|
Genistein , Daidzein , Licoricidin |
biosynthesis
The starting point for the biosynthesis of the flavonoids is the aromatic amino acid phenylalanine , which is formed via the shikimic acid pathway . Phenylalanine is converted into trans - cinnamic acid by the phenylalanine ammonia lyase (PAL) . This is in turn hydroxylated to p - coumaric acid by cinnamic acid 4- hydroxylase. This path is common to all phenylpropanoids . The p -cumaric acid is activated to form cumaryl-coenzyme A.
In the next step of the second aromatic ring is formed: the enzyme chalcone synthase (CHS) is formed from the cinnamoyl-CoA and three molecules of malonyl-coenzyme A , consisting of the fatty acid synthesis originate the chalcone . Chalcone is in equilibrium with flavanone through the action of chalcone isomerase (CHI). This closes the third ring.
The three key enzymes (PAL, CHS and CHI) as well as some of the enzymes of the further synthesis steps are present as enzyme complexes . The complex is probably on the cytosolic side of the endoplasmic reticulum .
The various paths lead from flavanone to flavones, flavonols, isoflavones and anthocyanidins.
The biosynthesis of the flavonoids is induced by light; the storage takes place mainly in the vacuole .
Most of the enzymes for flavonoid biosynthesis come from three classes of enzymes that are found in all organisms: oxoglutarate-dependent dioxygenases, NADPH-dependent reductases and cytochrome P450 hydroxylases. The two key enzymes CHS and CHI belong to different families. CHI is likely to be unique to plants in terms of both sequence and three-dimensional structure. CHS, in turn, belongs to the superfamily of vegetable polyketide synthases.
meaning
The various flavonoids fulfill a multitude of functions in plants.

Flavonoids form the most important group among the flower pigments and serve to attract pollinators . The anthocyanidins provide a variety of colors ranging from orange to red to blue. All anthocyanidin-containing flowers also contain flavones and / or flavonols, which serve to stabilize the anthocyanidins, but in higher concentrations also cause the flower color to shift into the blue area. Yellow flower color is less often caused by flavonoids. Flavonols such as gossypetin and quercetagetin are responsible for the yellow flower color in Gossypium hirsutum , Primula vulgaris and some sunflower plants such as Chrysanthemum segetum . Chalcones and aurones are responsible for the yellow flower color in some other daisy plants such as Coreopsis and Dahlia and in nine other plant families. Yellow flavonoids are often found in the daisy family together with the yellow carotenoids . 95% of the white flower color is caused by flavonoids: flavones such as luteolin and apigenin and flavonols such as kaempferol and quercetin , with flavonols absorbing somewhat further in the long-wave range.
The condensed tannins interact with the glycoproteins in the saliva of herbivores and have an astringent effect . They reduce the digestibility of the plants and thus deter many potential herbivores.
Other flavonoids act as protection against herbivores ( repellants ). For specialized insects, such flavonoids are in turn feeding stimulants. Flavone and flavonol glycosides, for example based on rutin , quercitrin and isoquercitrin , are particularly toxic to insects, while they are non-toxic to higher animals. The growth of various caterpillars is reduced dramatically in the presence of, for example, isoquercitrin in the diet, to 10% of the control groups. These flavonoids are mainly found in herbaceous plants and should replace the condensed tannins of the wood plants.
Flavones and flavonols in particular act as protection against UV radiation and short-wave light. They are deposited in the free form of plants in extreme locations such as in arid or alpine areas on the leaf surface, often in the form of flour-like coatings. In this way you prevent the photooxidative destruction of membranes and photosynthetic pigments . Due to their lipophilicity , they also reduce the colonization of the leaf surface with microorganisms . The flavonoids also have direct antiviral , antibacterial and antifungal effects.
Certain plant flavonoids play a role in the regulation of gene expression of the nodule bacterium Rhizobium .
Heavily methoxylated flavonoids are often found in exudates from buds and other lipophilic secretions. They have a fungicidal effect , as does the nobiletin in citrus leaves.
Flavonoids act as a structural theme in the development of selective GABA A receptor - ligand .
Flavonoids in food and medicine
food
Humans take in larger quantities of flavonoids with their food. Around two thirds of the approximately one gram phenolic substances that humans consume every day are flavonoids. The main group are flavanols (especially in countries with tea consumption). For a long time it was assumed that the potential effect of flavonoids was based on their antioxidant effects, but this is increasingly being questioned and other modes of action are more likely.
Epidemiological studies showed a lower risk of various diseases with higher flavonoid intake, including mortality from cardiovascular diseases . Flavonoids act on the arachidonic acid metabolism and thus on blood clotting. Various studies (as of 2016) show that flavonoids are also directly effective against cancer or its development.
Some compounds have been shown to have mutagenic or genotoxic effects in in vitro tests . Green tea extracts are suspected of damaging the liver , and one of the side effects of a drug based on (+) - catechin that has since been withdrawn from the market was hemolytic anemia . Flavonoids are also suspected of leading to premature closure of the ductus arteriosus .
Certain flavonoids lead to a strong inhibition of the cytochrome P450 -dependent monooxygenases (phase I enzymes), others in turn lead to an activation. A dose-dependent activation of phase II enzymes can also occur. All of this can lead to drug interactions, such as grapefruit .
Pharmaceutical importance
Several flavonoid-containing medicinal drugs are used therapeutically, as well as some pure substances. They are used as venous agents due to their vascular protective, edema-protective effect, as cardiovascular agents due to their positive inotropic , antihypertensive effect, as diuretics , as spasmolytics for gastrointestinal complaints and as liver therapeutics. Its effect is mainly attributed to its antioxidant properties and the inhibition of enzymes .
Epidemiological as well as most in-vivo studies indicate that flavonoids have a positive influence on various cardiovascular diseases . Traditionally, these effects have only been attributed to their antioxidant activities. However, in addition to the direct binding of reactive oxygen species (ROS), there are a number of other effects that, in pharmacologically achievable concentrations, also have a positive effect on the cardiovascular influence of the flavonoids such as e.g. B. Taxifolin may be responsible. These include in particular the inhibition of ROS-forming enzymes, inhibition of platelet function , inhibition of leukocyte activation and vasodilating properties.
Among the numerous effects of flavonoids that have been demonstrated in in vitro and in vivo tests, the most important are:
- anti-allergic and anti-inflammatory effect
- antiviral and antimicrobial effects
- antioxidant effect
- antiproliferative and anti- carcinogenic effect
Flavonoids work through several mechanisms of action. The focus is on the interaction with DNA and enzymes, the activation of cells, their property as radical scavengers and the influencing of various signal transduction pathways in the cells ( NF-κB , MAPK ). Flavonoids inhibit over thirty enzymes in the human body. They activate a wide variety of cell types in the immune system . The last two properties are responsible for the anti-inflammatory effects of flavonoids.
The following flavonoids are used as pure substances as venous agents:
Among the medicinal drugs predominate those that contain flavonol glycosides and glycosyl flavones. Important medicinal drugs that contain large amounts of flavonoids are:
- Arnica flowers ( arnica )
- Birch leaves ( silver birch , bog birch )
- Buckwheat herb ( Fagopyrum esculentum )
- Ginkgo leaves ( Ginkgo )
- Goldenrod (from Solidago virgaurea , Solidago gigantea and Solidago canadensis )
- Elderflower ( black elderberry )
- Hop cones ( real hops )
- Chamomile flowers ( chamomile )
- Cat's Paw Flowers ( Common Cat's Paw )
- Larch extract ( Taxifolin )
- Meadowsweet herb and flowers ( meadowsweet )
- Milk thistle fruits ( milk thistle )
- Passion flower herb (from Passiflora incarnata )
- Bitter orange peel ( bitter orange )
- Marigold flowers ( marigold )
- Roman chamomile
- Red vine leaves ( Vitis vinifera )
- Safflower flowers ( safflower )
- Pansy herb ( Viola arvensis and Viola tricolor )
- Licorice root ( liquorice )
- Hawthorn leaves with flowers (several species of hawthorn )
literature
- O. M. Andersen, KR Markham: Flavonoids: Chemistry, Biochemistry and Applications . CRC Press, Taylor and Francis, Boca Raton 2006, ISBN 978-0-8493-2021-7 .
Individual evidence
- ↑ St. Rusznyak, Albert Szent-Györgyi: Vitamin P: flavonols as vitamins. In: Nature. Volume 138, 1936, p. 27.
- ↑ a b c d e f g h i j k Rudolf Hänsel, Otto Sticher (Ed.): Pharmakognosie. Phytopharmacy . 9th edition. Springer Medizin Verlag, Heidelberg 2009, ISBN 978-3-642-00962-4 , pp. 1098-1152.
- ↑ a b c d e f Martin Luckner: Secondary Metabolism in Microorganisms, Plants and Animals . 3rd edition, VEB Gustav Fischer Verlag, Jena 1990, ISBN 3-334-00322-1 , pp. 406-415.
- ↑ a b c Brenda Winkel-Shirley: Flavonoid Biosynthesis. A Colorful Model for Genetics, Biochemistry, Cell Biology, and Biotechnology . In: Plant Physiology. Volume 126, 2001, pp. 485-493, doi: 10.1104 / pp.126.2.485 .
- ↑ a b c d Bernhard Watzl, Gerhard Rechkemmer: Basic knowledge updated: Flavonoide. In: Nutrition review. Volume 48, No. 12, 2001 ( ernaehrungs-umschau.de ).
- ↑ a b c Hans-Walter Heldt : Plant biochemistry . Spektrum Akademischer Verlag, Heidelberg 1996, ISBN 3-8274-0103-8 , pp. 423-437.
- ^ Dieter Schlee: Ecological Biochemistry . 2nd Edition. Gustav Fischer Verlag, Jena 1992, ISBN 3-334-60393-8 , p. 67 f.
- ↑ a b J. B. Harborne: Introduction to Ecological Biochemistry . Third edition. Academic Press, London 1988, ISBN 0-12-324684-9 , pp. 47-53.
- ^ JB Harborne: Introduction to Ecological Biochemistry . Third edition. Academic Press, London 1988, ISBN 0-12-324684-9 , pp. 95, 175 f.
- ↑ Joseph Naghski, Michael J. Copley, James F. Couch: The antibacterial action of flavonols. In: Journal of Bacteriology. Vol. 54, 34 (1947).
- ^ Dieter Schlee: Ecological Biochemistry . 2nd Edition. Gustav Fischer Verlag, Jena 1992, ISBN 3-334-60393-8 , p. 271 f.
- ↑ JR Hanrahan, M. Chebib, GA Johnston: Flavonoid modulation of GABA (A) receptors . In: Br J Pharmacol . 163, No. 2, 2011, pp. 234–245. doi : 10.1111 / j.1476-5381.2011.01228.x . PMID 21244373 . PMC 3087128 (free full text).
- ↑ a b c Anna Vogiatzoglou, Angela A. Mulligan, Marleen AH Lentjes, Robert N. Luben, Jeremy PE Spencer: Flavonoid Intake in European Adults (18 to 64 Years) . In: PLOS ONE . tape 10 , no. 5 , May 26, 2015, p. e0128132 , doi : 10.1371 / journal.pone.0128132 ( plos.org [accessed August 15, 2017]).
- ↑ Robert J. Williams, Jeremy PE Spencer, Catherine Rice-Evans: Flavonoids: antioxidants or signaling molecules? In: Free Radical Biology and Medicine . tape 36 , no. 7 , p. 838–849 , doi : 10.1016 / j.freeradbiomed.2004.01.001 ( elsevier.com [accessed August 15, 2017]).
- ↑ Anja Klappan: Influence of the flavonoid quercetin on the mTOR signaling pathway of cervical and breast cancer cells . Munich 2014, DNB 1048361322 , urn : nbn: de: bvb: 19-165609 (dissertation, LMU Munich).
- ↑ See also José Rueff et al. a .: Genetic toxicology of flavonoids: the role of metabolic conditions in the induction of reverse mutation, SOS functions and sister chromatid exchanges. In: Mutagenesis. Volume 1, No. 3, 1986, pp. 179-183.
- ↑ Stephanie E. Martinez, Neal M. Davies, Jonathan K. Reynolds: Toxicology and Safety of Flavonoids . In: FLAVONOID PHARMACOKINETICS . John Wiley & Sons, Inc., 2012, ISBN 978-1-118-46852-4 , pp. 249-280 , doi : 10.1002 / 9781118468524.ch6 .
- ↑ D. Shinkov, I. Urumov, N. Doĭchinova, V. Manolova, R. Ananieva: [Immune hemolytic anemia caused by catergen] . In: Vutreshni Bolesti . tape 28 , no. 5 , 1989, pp. 84-87 , PMID 2618015 .
- ↑ Paulo Zielinsky, Stefano Busato: Prenatal effects of maternal consumption of polyphenol-rich foods in late pregnancy upon fetal ductus arteriosus . In: Birth Defects Research Part C: Embryo Today: Reviews . tape 99 , no. 4 , December 1, 2013, p. 256-274 , doi : 10.1002 / bdrc.21051 .
- ↑ P. Mladenka, L. Zatloukalová, T. Filipský, R. Hrdina: Cardiovascular effects of flavonoids are not only Caused by direct antioxidant activity . In: Free Radic Biol Med . 2010, 49, pp. 963-975, PMID 20542108 .