Fentanyl
| Structural formula | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
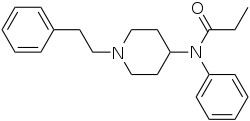
|
|||||||||||||
| General | |||||||||||||
| Non-proprietary name | Fentanyl | ||||||||||||
| other names | |||||||||||||
| Molecular formula | C 22 H 28 N 2 O | ||||||||||||
| Brief description |
White to almost white, polymorphic powder |
||||||||||||
| External identifiers / databases | |||||||||||||
|
|||||||||||||
| Drug information | |||||||||||||
| ATC code | |||||||||||||
| Drug class | |||||||||||||
| properties | |||||||||||||
| Molar mass | 336.47 g · mol -1 | ||||||||||||
| Melting point | |||||||||||||
| solubility | |||||||||||||
| safety instructions | |||||||||||||
|
|||||||||||||
| Toxicological data | |||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||
Fentanyl is a synthetic opioid that is used as a pain reliever in anesthesia (during anesthesia) and for the treatment of acute and chronic pain that can only be adequately treated with opioid analgesics. Fentanyl acts as an agonist on the μ- opioid receptor . Fentanyl falls under the German and Swiss Narcotics Act as well as the Austrian Narcotics Act .
development
In 1960, Paul Janssen developed Fentanyl as the first anilinopiperidine (4-anilinopiperidine-4-carboxamide). Since then, a number of more controllable derivatives have been developed from fentanyl by modifying the molecular structure.
application
Fentanyl is commonly used as fentanyl dihydrogen citrate. There are four forms of application:
- as intravenous administration (e.g. in anesthesia or emergency medicine )
- as a transdermal therapeutic system (i.e. as a membrane plaster stuck to the skin , which then releases the active ingredient (at either 12.5, 25, 37.5, 50, 75 or 100 micrograms per hour) in a controlled manner for absorption through the skin; e.g. . Durogesic SMAT , generics )
- as a fast-releasing formulation such as the oral-transmucosal therapeutic system ( Actiq from Elan Pharma ), which is used for breakthrough pain as a lozenge containing 200 to 1600 µg active ingredient with an integrated applicator on the oral mucosa, as a buccal tablet (trade name Effentora ) with 100 to 800 µg active ingredient or as a sublingual tablet (trade name Abstral ) with 50 to 800 µg fentanyl.
- the fentanyl nasal spray ( PecFent from Archimedes Pharma or Instanyl from Takeda )
These rapid-release formulations are indicated for the treatment of breakthrough pain (very intense, minute-long pain, typical of advanced cancer) in adults who are already receiving basic opioid therapy for their tumor pain.
effect
Fentanyl has a strong pain-relieving ( analgesic ) and sedating effect , as well as respiratory depressive , antitussive , pulse-slowing, blood pressure lowering, constipating and euphoric. It is about 70 to 100 or 120 times as potent as morphine (measured by mass, only a hundred twentieth the amount of fentanyl is necessary to achieve the same effect), has a higher effectiveness (the maximum effect is higher), while its duration of action is usually significantly shorter. Fentanyl works after five to twenty seconds when given intravenously . The maximum effect occurs after four to five minutes. The half-life is three to twelve hours. The effective dose ( ED 50 ) for treatment is 0.002 to 0.02 mg / kg body weight with constant breathing monitoring.
The analgesic duration of action of fentanyl depends on the drug form used: 48 to 72 hours for a transdermal patch, for transmucosal application 0.5 to 2 hours, for buccal tablets and sublingual tablets 4 to 6 hours.
Fentanyl is lipophilic , i. H. easily soluble in fat and therefore spreads quickly in fatty tissue. Fentanyl is mainly metabolized in the liver and less than ten percent of the time it is excreted unchanged by the kidneys .
The main area of application of the intravenous form is the administration as pain reliever during operations in connection with a sleeping aid and optionally a muscle relaxant ( muscle relaxant ). Depending on the choice of sleeping aid, one speaks of “balanced anesthesia” or “total intravenous anesthesia” ( TIVA ). Because of the risk of respiratory depression, constant monitoring and the availability of a ventilation option is necessary. Due to its lipophilicity , fentanyl is sometimes stored and released again in adipose tissue, which is difficult to control. That is why the related substances alfentanil , remifentanil and sufentanil are often used today instead of fentanyl .
In the case of fentanyl-containing active substance patches, continuous monitoring of the vital functions is not necessary after a setting phase due to the uniform effect and the usually significantly lower dose compared to anesthesia.
Interactions
The sedative effects of fentanyl can be increased by sedatives and alcohol. Serious circulatory and respiratory disorders can occur in connection with monoamine oxidase inhibitors . There should be at least 14 days between the use of MAOIs and fentanyl. Due to the plasma protein binding of 90%, there may be interactions with preparations such as furosemide , glibenclamide or omeprazole when used in pain plasters . The pathway of fentanyl via the cytochrome oxidase 450 , so that with simultaneous administration of CYP 3A4 - inducers (eg. St. John's wort preparations; accelerate the degradation of fentanyl) or CYP 3A4 inhibitors (eg. Ketoconazole , erythromycin , nefazodone , Diltiazem , grapefruit juice ; slow down the breakdown of fentanyl and can lead to increased plasma levels ) Interactions may occur.
Calcium antagonists: The combination of fentanyl (and other opioids) with calcium antagonists can lead to a potentiation of the analgesic effect in addition to reducing the development of tolerance. In 1985 the potentiating effect of nimodipine compared to the analgesic effect of fentanyl was described. The combined administration of 0.1 mg fentanyl together with nimodipine (1 µg / kg / min infusion) is said to have the same effectiveness as 2.45 mg fentanyl, which corresponds to a potentiation of 24.5 times. Further human experiments showed that, unlike nimodipine , nifedipine did not potentiate the fentanyl effect, but the effect of nimodipine in this study was also significantly weaker (3-fold potentiation) than in the first study.
The simultaneous administration of fentanyl-containing drugs with serotonergic drugs (SSRI / SNRI) poses a risk of developing a dangerous serotonin syndrome with symptoms such as agitation , coma , hallucinations , blood pressure crises , neuromuscular changes and palpitations , as was warned in March 2013.
Side effects
Like other opioids, fentanyl overdoses provokes a disorder of the central nervous system with impaired consciousness, somnolence and respiratory depression . The acute picture essentially shows pronounced sedation , miosis (constriction of the pupil) and respiratory depression, with this up to and including respiratory arrest. Fentanyl can be antagonized with naloxone .
Since 2005, the FDA has repeatedly reported serious side effects and deaths associated with fentanyl-containing transdermal therapeutic systems , 397 deaths in 2009 alone. In 2012, the Drugs Commission of the German Medical Association (AkdÄ) also pointed out that the indication for transdermal, fentanyl-containing patches must be strict. They should only be prescribed for chronic pain that can only be adequately alleviated with opioids and requires constant, continuous treatment. While the FDA only allows its use in patients who have taken 60 mg morphine per day or more for at least a week (or another opioid, respectively), the German specialist information advises that patients who have not previously taken opioids should initially give low-dose, non-delayed opioids. until the dose of the dose of 12.5 μg / h fentanyl (= 45 mg morphine / day ). and only then switch to a plaster.
In 2014, a Rote-Hand-Brief warned against "accidental transfer of the fentanyl patch to the skin of another person (e.g. while sharing a bed or during closer physical contact)". "To protect against accidental ingestion by children, the application site should be carefully selected and regularly monitored to ensure that the patch is still there."
Analytics
For the reliable qualitative and quantitative determination of fentanyl in blood / blood plasma samples and urine , the coupling of gas chromatography or HPLC with mass spectrometry is used after appropriate sample preparation . These procedures are also used for forensic examinations. For doping control , these methods are also used. Also immunoassays used.
Substance Abuse, Overdose, and Potential Hazards
Since 2006 at the latest, the abuse of fentanyl has been increasing in the USA. In a report, the CDC spoke of 1,000 deaths between 2005 and 2007; most of the cases were recorded in Chicago , Philadelphia, and Detroit . In 2014 there were 4,200 dead. Preliminary data shows that fentanyl was responsible for a large proportion of the estimated 59,000 to 65,000 drug deaths in the US in 2016 (see opioid epidemic in the US ). In the district of Montgomery County drug deaths fentanyl or an analog was detected in January and February 2017 99 of the 100th According to the New York Times , at least 220 people died from fentanyl abuse in 2016 in Long Island alone .
One reason for the high death rate with fentanyl applications is the extremely high efficacy or narrow therapeutic range . While a therapeutic dose above 0.02 mg / kg body weight is only possible when using the heart-lung machine , the lethal dose for (rhesus) monkeys is already 0.03 mg / kg body weight. Fentanyl can be fatal in an amount as low as 2 mg for an adult of normal weight.
Fentanyl in powder form also poses a particular risk, as it is acquired as a pure substance before processing into finished medicinal products, but is also traded on the black market in order to then e.g. B. to be taken as an injection, or by nasal consumption. There are known incidents in which the powder was involuntarily swirled in the air - for example when a container with the substance was dropped. Fentanyl aerosol can unintentionally get into the nasal and / or oral mucosa. Unintentional absorption through the skin is also possible here.
Fentanyl is also used to stretch heroin. If the fentanyl is not evenly distributed, each dose contains different amounts of fentanyl and there can be deadly fentanyl hot spots in the stretched drug mass. Fentanyl, which is used in this way or sold directly on the black market, comes partly from legal pharmaceutical and partly from illegal production.
By introducing various chemical structural elements into the fentanyl molecule , a number of designer drugs have been synthesized, such as methylfentanyl and benzylfentanyl.
Warfare agent
In addition to their use in medicine, fentanyl derivatives were also examined for their usefulness as chemical warfare agents . Suspicions have been made as to whether a particularly potent fentanyl derivative, carfentanyl , which has not been approved in human medicine , was used in aerosol form in the rescue of the hostages in Moscow's Dubrowka Theater in October 2002 and was one of the causes of 127 deaths from respiratory failure .
A derivative of fentanyl was used by an Israeli Mossad commando in the 1997 assassination attempt on Chalid Maschal in Jordan. Much more potent than fentanyl, it was absorbed through the skin like this (sprayed on the head) and should kill with the appearance of natural death in a few days. The agents were immediately arrested and, under diplomatic pressure, Israel gave the Jordanians the type of active ingredient and the effective antidote ( naloxone ) with instructions on treatment.
Use of fentanyl in executions
Fentanyl was first a drug mixture for as part of the August 14, 2018 execution of double murderer Carey Dean Moore in the state prison of Nebraska used. The reason for the use in lethal injections in the USA is the increasingly difficult procurement of the previously used toxins (including thiopental ) due to a delivery or production stop on the part of the manufacturer. The US state of Nevada was also planning to use fentanyl for executions. After the manufacturer filed a lawsuit against what it believed to be the illegal use of its preparation in July 2018, the execution of Scott Dozier was initially suspended.
Trade names
Abstral (D), Actiq (D, A, CH), Durogesic (D, A, CH), Effentora (D, A), Ernsdolor (A), Fentadolon (D), Fentamed (A), Fentaplast (A), Fentarichtex (A), Fentoron (A), Gelitanyl (A), Ionsys (A), Instanyl (D), Matrifen (D, A), Sintenyl (CH), numerous generics (D, A, CH)
literature
- Eberhard Klaschik : Pain therapy and symptom control in palliative medicine. In: Stein Husebø , Eberhard Klaschik (ed.): Palliative medicine. 5th edition, Springer, Heidelberg 2009, ISBN 3-642-01548-4 , pp. 207-313, here: pp. 233 f., 251 f. and 466 f.
Web links
- Fentanyl . In: Erowid . (English)
- Public Assessment Report (EPAR) of the European Medicines Agency (EMA) for: Fentanyl
Individual evidence
- ↑ a b European Pharmacopoeia Commission (Ed.): European Pharmacopoeia 5th Edition . tape 5.0-5.7 , 2006.
- ↑ a b c d e Entry on fentanyl in the ChemIDplus database of the United States National Library of Medicine (NLM)
- ^ Royal Pharmaceutical Society (Ed.): Clarke's Analysis of Drugs and Poisons FOURTH EDITION . Pharmaceutical Press, London / Chicago 2011, ISBN 978-0-85369-711-4 .
- ↑ a b data sheet fentanyl citrate salt from Sigma-Aldrich , accessed on March 31, 2011 ( PDF ).
- ↑ Entry on 4-anilinopiperidine-4-carboxamide at ChemicalBook , accessed on January 20, 2014.
- ↑ Summary of the product characteristics Instanyl ® , as of: July 20, 2009 on the website of the European Medicines Agency EMEA , (PDF, 381 kB). Accessed October 1, 2009.
- ↑ Holger Thiel, Norbert Roewer: Anästhesiologische Pharmakotherapie. General and special pharmacology in anesthesia, intensive care medicine, emergency medicine and pain therapy. Georg Thieme, Stuttgart / New York 2004 ISBN 3-13-138261-9 , pp. 173-176.
- ↑ a b Eberhard Klaschik : Pain therapy and symptom control in palliative medicine. 2009, p. 233 f.
- ↑ B. Bormann, J. Boldt, G. Sturm, D. Kling, B. Weidler, E. Lohmann, G. Hempelmann: Calcium antagonists in anesthesia. Additive analgesia using nimodipine in heart surgery. In: Anaesthesiologist. 34, 1985, p. 429. PMID 4083464 .
- ↑ J. Boldt, B. von Bormann, D. Kling, W. Russ, K. Ratthey, G. Hempelmann: Low-dose fentanyl analgesia modified by calcium channel blockers in cardiac surgery. In: Eur. J. Anesth. 4 (6), 1987, p. 387. PMID 3328680
- ↑ Rote-Hand-Letters on Fentanyl®-Janssen and Durogesic SMAT (12, 25, 50, 75, 100 µg / h), transdermal patch (active ingredient: fentanyl): New warning , notification from the higher federal authority BfArM of March 11, 2013, accessed on September 22, 2015.
- ↑ Fentanyl Transdermal System (marketed as Duragesic) information. In: FDA ALERT 7/15/2005; Update 12/21/2007. September 30, 2009, accessed August 26, 2010 .
- ↑ Reported Patient Deaths Increased by 14% in 2009. (68 kB; PDF) (No longer available online.) In: QuarterWatch: 2009 Quarter 4th Institute for Safe Medication Practices, June 16, 2010, p. 2 , archived from the original on December 4, 2010 ; Retrieved on August 26, 2010 (English, US 501c (3) nonprofit organization): "In 2009 fentanyl products accounted for 397 reported patient deaths, ranked 4th among all drugs" Info: The archive link was inserted automatically and has not yet been checked. Please check the original and archive link according to the instructions and then remove this notice. .
- ↑ Deutsches Ärzteblatt. Vol. 109, issue 14, April 6, 2012 (PDF; 259 kB) retrieved from the website of the Drugs Commission of the German Medical Association (AkdÄ).
- ↑ Rote-Hand-Brief from June 12, 2014 (link checked on June 20, 2014)
- ^ BA Goldberger, CW Chronister, ML Merves: Quantitation of fentanyl in blood and urine using gas chromatography-mass spectrometry (GC-MS). In: Methods Mol Biol. 603, 2010, pp. 245-252. PMID 20077075
- ↑ SQ Cheng, DS Ouyang, YJ Tang, K. Wu, J. Xiao: A rapid, simple and sensitive method for the determination and bioequivalence study of transdermal fentanyl in human plasma using liquid chromatography-electrospray ionization tandem mass spectrometry. In: Int J Clin Pharmacol Ther. 54 (2), Feb 2016, pp. 144–156. PMID 26754304
- ↑ RL Shaner, P. Kaplan, EI Hamelin, WA Bragg, RC Johnson: Comparison of two automated solid phase extractions for the detection of ten fentanyl analogs and metabolites in human urine using liquid chromatography tandem mass spectrometry. In: J Chromatogr B Analyt Technol Biomed Life Sci. 962, Jul 1, 2014, pp. 52-58. PMID 24893271
- ↑ J. Poklis, A. Poklis, C. Wolf, M. Mainland, L. Hair, K. Devers, L. Chrostowski, E. Arbefeville, M. Merves, J. Pearson: Postmortem tissue distribution of acetyl fentanyl, fentanyl and their respective nor-metabolites analyzed by ultrahigh performance liquid chromatography with tandem mass spectrometry. In: Forensic Sci Int. 257, Dec 2015, pp. 435-441. PMID 26583960
- ↑ R. Verplaetse, J. Tytgat: Development and validation of a sensitive ultra performance liquid chromatography tandem mass spectrometry method for the analysis of fentanyl and its major metabolite norfentanyl in urine and whole blood in forensic context. In: J Chromatogr B Analyt Technol Biomed Life Sci. 878 (22), Jul 15, 2010, pp. 1987-1996. PMID 20542478
- ↑ M. Thevis, H. Geyer, D. Bahr, W. Schänzer: Identification of fentanyl, alfentanil, sufentanil, remifentanil and their major metabolites in human urine by liquid chromatography / tandem mass spectrometry for doping control purposes. In: Eur J Mass Spectrom. (Chichester). 11 (4), 2005, pp. 419-427. PMID 16204809
- ↑ SM Thomasy, KR Mama, SD Stanley: Comparison of liquid chromatography-mass spectrometry and radioimmunoassay for measurement of fentanyl and determination of pharmacokinetics in equine plasma. In: J Anal Toxicol. 32 (9), Nov-Dec 2008, pp. 754-759. PMID 19021930
- ↑ T. Tobin, HH Tai, CL Tai, PK Houtz, MR Dai, WE Woods, JM Yang, TJ Weckman, SL Chang, JW Blake and others: Immunoassay detection of drugs in racing horses. IV. Detection of fentanyl and its congeners in equine blood and urine by a one step ELISA assay. In: Res Commun Chem Pathol Pharmacol. 60 (1), Apr 1988, pp. 97-115. PMID 2967991
- ↑ Nonpharmaceutical Fentanyl-Related Deaths - Multiple States, April 2005 – March 2007 . CDC, MMWR, July 25, 2008/57 (29), pp. 793-796.
- ↑ Tom Porter, What is Fentanyl, the Drug Linked to Thousands of Deaths in North America? In: Newsweek.com. April 28, 2017. Retrieved August 11, 2017 .
- ↑ Josh Katz: Drug Deaths in America Are Rising Faster Than Ever. In: New York Times. 5th June 2017.
- ↑ Kevin Deutsch: Toll of Fentanyl, more potent than heroin, is rising . In: New York Times . December 29, 2016.
- ↑ Akorn Inc .: Fentanyl Citrate Injection, USP ,. Retrieved July 15, 2020 .
- ↑ Lethal vs. Medicinal Doses of Fentanyl. In: oxfordtreatment.com. Retrieved September 25, 2018 .
- ↑ Fentanyl drug profile. European Monitoring Center for Drugs and Drug Addiction (EMCDDA) from January 8, 2015, accessed on September 25, 2018.
- ↑ Warning message from Interpol on risks associated with handling fentanyl, fentanyl derivatives and other synthetic opioids. Retrieved on August 15, 2020 (German).
- ↑ Rapid test for the super drug fentanyl - limit to poison in the nano range. Retrieved on August 15, 2020 (German).
- ↑ How lethal fentanyl 'hot spots' end up getting cut into street drugs. In: cbc.ca. October 17, 2016. Retrieved August 11, 2017 .
- ^ Fentanyl crisis: Is China a major source? In: bbc.com. September 24, 2018, accessed September 25, 2018 .
- ^ Enno Freye: Opioids in medicine . Springer, 2008, ISBN 3-540-46570-7 , pp. 371 ( limited preview in Google Book search).
- ^ Medical ethics and non-lethal weapons. In: Am J Bioeth. 4 (4), 2004, pp. W1-W2. PMID 16192174 .
- ↑ Fentanyl and its analogues in clinical and forensic toxicology. In: Przegl Lek. 62 (6), 2005, pp. 581-584. PMID 16225129 .
- ↑ Unexpected "gas" casualties in Moscow: a medical toxicology perspective. In: Ann Emerg Med . 41 (5), May 2003, pp. 700-705. PMID 12712038 .
- ↑ Paul McGeough, Kill Khalid, Allen and Unwin 2009, pp. 2, 183f. McGeough speaks of levofentanyl, which was a research by-product of the Janssen Pharmaceutica company in New Jersey that had not come onto the market and was also unknown in pharmaceutical databases at the time . Lofentanil is also sometimes mentioned .
- ↑ However, the Jordanian doctors already had clues from the antidotes found on the Israeli agents. See Mcgeough, pp. 183f; see also chemistry.stackexchange.com .
- ^ Telegraph Reporters: Judge clears way for America's first execution using fentanyl . In: The Telegraph . August 11, 2018 ( telegraph.co.uk [accessed August 11, 2018]).
- ↑ Nebraska uses fentanyl in execution. In: orf.at. August 2018, accessed August 14, 2018 .
- ↑ Chris Mc Greal: Nevada to become first state to execute inmate with fentanyl. In: theguardian.com. July 10, 2018, accessed September 25, 2018 .
- ^ Richard A. Oppel Jr .: Nevada Execution Is Blocked After Drugmaker Sues. In: The New York Times . July 11, 2018, accessed September 25, 2018 .