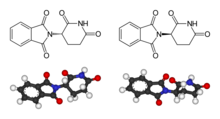
Thalidomide
| Structural formula | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|

|
|||||||||||||||||||
| Simplified structural formula without stereochemistry | |||||||||||||||||||
| General | |||||||||||||||||||
| Non-proprietary name | Thalidomide | ||||||||||||||||||
| other names |
|
||||||||||||||||||
| Molecular formula | C 13 H 10 N 2 O 4 | ||||||||||||||||||
| Brief description |
colorless solid |
||||||||||||||||||
| External identifiers / databases | |||||||||||||||||||
|
|||||||||||||||||||
| Drug information | |||||||||||||||||||
| ATC code | |||||||||||||||||||
| Drug class | |||||||||||||||||||
| characteristics | |||||||||||||||||||
| Molar mass | 258.23 g · mol -1 | ||||||||||||||||||
| Physical state |
firmly |
||||||||||||||||||
| Melting point |
270 ° C |
||||||||||||||||||
| solubility |
practically insoluble in water (45–60 mg · l −1 at 25 ° C) |
||||||||||||||||||
| safety instructions | |||||||||||||||||||
|
|||||||||||||||||||
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |||||||||||||||||||
Thalidomide (α-phthalimidoglutarimide) is a drug developed in 1954 that was sold as a sleeping and sedative under the brand names Contergan and Softenon and caused numerous serious damage to unborn life ( dysmelia, etc.) in the late 1950s / early 1960s led to the thalidomide scandal . Chemically speaking, like glutethimide , methyprylon and pyrithyldione , it belongs to the group of piperidinediones , a structural oneModification of the barbiturates . In addition to the sleep-promoting effect, the active ingredient also has anti-tumor, anti-inflammatory and anti-blood vessel formation properties. Since 1998, thalidomide has been used in the USA for the treatment of particularly severe forms of leprosy and since 2009 in Germany for the treatment of multiple myeloma, subject to strict safety requirements; it is offered as a generic by Celgene Company .
story
Sleep and sedative medication
In March 1954, in the research department of the Stolberg pharmaceutical company Grünenthal - looking for an inexpensive method to obtain antibiotics from peptides and as part of the development of novel sleeping pills such as pyrithyldione ( persedon ) and glutethimide - by heating the phthaloylisoglutamine from Heinrich Mückter, Wilhelm Kunz (born 1920) and Herbert Keller (born 1925) synthesized phthalimidoglutarimide. It was given the name Thalidomid by the head of the research department, Mückter, and in December 1960 accounted for almost half of Grünenthal's domestic sales.
Grünenthal's research results did not produce any pathogenic reactions for thalidomide or any other positive results that could be used in medicine in (non-pregnant) mice and rats. The active ingredient thus appeared to be harmless to Grünenthal. Also, high doses showed no lethal effect in rats, mice, guinea pigs, rabbits, cats and dogs, and no other side effects were found in Grünenthal. Thalidomide was therefore classified as “non-toxic”, which is very attractive for commercial evaluation. The patented compound has begun to be used. In animal experiments, thalidomide was initially not shown to be sedative . As a non-lethal sedative would have great market potential, the company decided to intensify research in the direction of a human sleep aid.
Initially, it was prescribed for seizure prophylaxis in epilepsy . Although it had no anticonvulsant effect, the patients reported the good sleep-inducing and relaxing properties of the drug.
Thalidomide was not allowed to be sold in Germany until its effectiveness was tested in animal experiments. Since thalidomide had no sedative effect in animal experiments, it was tested on mice in shaker cages. It was investigated to what extent the movements of the mice in the verum group (group that received the active ingredient) and the placebo group differed.
Before the drug came on the market, a Grünenthal employee took a sample home and gave it to his pregnant wife to help her sleep better. The child was born without ears and thus became the first Contergan victim.
Grünenthal marketed the substance from October 1, 1957 to November 1961 under the name Contergan as the "first bromine-free sleeping and sedative drug without major side effects ". Since thalidomide among other things, against the typical morning morning sickness helped in the early stages of pregnancy, it was specifically the end of the 1950s than the recommended sedatives and sleeping pills for pregnant women and advertised. With regard to side effects, it was considered to be particularly safe, because as a bromine-free sleep aid, side effects such as confusion, delirium and stuttering should be avoided . It was also significantly less dangerous than the barbituratesoften used in suicides. Thalidomide was also contained in the medicines Algosediv , Grippex and, as a component supplied by Grünenthal, in the cough medicine Peracon from Kali Chemie .
Contergan scandal
At the end of the 1950s, there was an initially inexplicable increase in malformations in newborns. In the public and in the course of the time the newly founded German Federal Ministry of Health was suspected of possible damage from nuclear energy and in particular the nuclear weapons tests that were carried out above ground at the time . The rate of malformations in children was examined and an increase was statistically denied, but with data that only reached up to the time of marketing approval of thalidomide. On the basis of these statistics, it was only believed that the newly occurring damage would accumulate by chance, which significantly delayed the investigation of the cause.
The teratogenic effects of the drug thalidomide were discovered independently in Germany and Australia. The Australian doctor William McBride published on December 16, 1961 a letter to the editor on malformations in newborns in connection with the administration of thalidomide in The Lancet . He had previously turned to the licensee in Australia, who forwarded the warning to the German manufacturer Grünenthal in November 1961. Shortly before, on November 15, 1961, the human geneticist Widukind had Lenznotified the company of his suspicions. On November 18, he reported on his suspicions at a specialist conference. The Grünenthal company initially reacted wait-and-see. Although in 1961 the Stolberg-based manufacturing company received 1,600 warnings about observed malformations in newborns, Contergan was still sold. Only after a newspaper article published Lenz's suspicion in Die Welt am Sonntag on November 26, 1961, Grünenthal finally withdrew Contergan from the trade the following day .
According to the Federal Association of Thalidomide Victims , around 5000 children with thalidomide disease were born. Other sources speak of 10,000 cases worldwide, of which 4,000 occurred in the Federal Republic of Germany , half of which had already died (as of 2007). In addition, there is an unknown number of miscarriages. In the GDR , the Central Advisory Committee for Pharmaceuticals had rejected the manufacture and sale of sleeping pills based on thalidomide, as this was classified as insufficiently checked. A few thalidomide packs came to the GDR from the Federal Republic. A total of 8 cases of malformations caused by Contergan are documented in the GDR.
In Austria and Switzerland the remedy was called Softenon and was only available on prescription . Officially, there are 13 thalidomide victims in Austria, as the prescription requirement was very restrictive there at the time. 9 Contergan-damaged children were born in Switzerland. Softenon was taken off the market in 1962.
There are also recognized cases of damage from thalidomide in Brazil . Contergan was also delivered in large quantities to Eastern Europe and the Middle East.
In the USA , Contergan was denied approval, but it was distributed in large quantities "for test purposes" after the American manufacturer Richardson-Merrell first applied for approval in September 1960. Frances Oldham Kelsey , the US Food and Drug Administration pharmacologist in charge, did not rely on the Richardson-Merrell Company, which did not include test results. Instead, only general statements from Grünenthal and the marketing department of Richardson-Merrell were given, and business people and politicians put pressure on Kelsey. She asked Richardson-Merrell to run tests and report the results. The company refused and asked for permission to be granted six times, which were refused each time. In 1962, Richardson-Merrell withdrew the application for approval. Nevertheless, a total of 17 children were born with thalidomide-related deformities.
The concentrated media presence of the topic, due to the 50th anniversary of the market launch of Contergan and the two-part television show Contergan , which was broadcast shortly afterwardson December 7, 2007 - 46 years after the damaging effect became known - led to initial discussions between Grünenthal and the Federal Association of Contergan Victims. Other victims are also calling for a dialogue and have started an online signature campaign based on the quote from the producer of the two-part film. Affected associations also hope for financial and other improvements in their situation as a result. On July 1, 2008, the doubling of the monthly compensation payments was implemented. Politicians and associations describe this as the “first important step” to mitigate the “financial impact of late and consequential damage”. On May 8, 2008, Grünenthal announced that it would voluntarily pay 50 million euros into the Contergan Foundation, in order to permanently improve the living conditions of the Contergan victims. This was implemented on July 15, 2009.
Thalidomide damage
Thalidomide , if taken within the first three months of pregnancy , leads to severe malformations of the limbs ( dysmelia , such as radial clubhand ), especially to the absence of long bones ( phocomelia ), or even to the total absence ( aplasia ) of limbs and organs in children. Thalidomide blocks the growth factor VEGF (Vascular Endothelial Growth Factor), which leads to a lack of vascularization(Blood vessel formation) occurs in the extremities of the embryo. This leads to a shortened or missing contact of the arms and legs, mostly on both sides. This has to be distinguished from a mechanical constriction of limbs (see amniotic ligament syndrome ).
The protein cereblon , which forms a ubiquitin ligase complex with other proteins, was identified as a direct, teratogenic target . This complex also controls limb morphogenesis, and the inhibition of ligase activity by binding of thalidomide to cereblon is a major biological cause of malformations. This mechanism became known in 2010.
In 2018 it was published that thalidomide and the similar drugs lenalidomide and pomalidomide break down various transcription factors , including all three SALL4 ("Sal-like protein 4"), which is necessary for growth. While in Okihiro syndrome , in which very similar malformations can occur, the failure of SALL4 is based on its genetic mutation, these drugs do not lead to a mutation. Apart from humans, this failure affects only a few animal species (primates and rabbits); in others, such as mice, SALL4 is not degraded by Cereblon due to genetic differences, which is why the teratogenic effect was not recognized in the animal experiments customary at the time.
In the case of thalidomide embryopathy, it has been particularly well investigated at which point in time when the drug is exposed to what damage: For example, if the drug is taken between the 34th and 37th day after the last menstrual period, the auricle is missing, between the 35th . and 41st days to an underdevelopment of the thumb, between the 38th and 45th day arm malformations, between the 41st and 47th day leg malformations and between the 46th and 50th day thumb malformations.
In the period after 1961, after Contergan was withdrawn from the market, it continued to be tested on various animal species . The results were mostly negative or showed no comparable malformations in the offspring. Although rats and mice were sedated by the substance (the reason why the sedative effect of thalidomide was even discovered, was that in an experiment in which a completely different effect of the active substance was to be investigated, the rodents, thalidomide received, unexpectedly fell asleep), no clear teratogenic effects of the preparation could be demonstrated even in classic animal experiments with high concentrations. It was not until 1962 that white New Zealanders were found- a breed of rabbit that was not a common animal model before it was launched on the market - a teratogenic effect. This species-specificity of a teratogenic effect, which has been proven in this way, was previously unknown and represented a lack of transferability of animal test results to humans.
The fear that thalidomide could also damage subsequent generations by changing the genetic make-up has not come true. The children of thalidomide-damaged people are not affected by thalidomide syndrome.
A polyneuropathy can develop in the consuming patient himself , which is the most important dose-limiting factor in the new indications . It can regress quickly and completely, but it can also persist for years. In addition, an increased risk of malignant degeneration is being investigated more closely; the manufacturer had already published a red-hand letter in 2013 .
More than 2000 research reports have written down the results of studies that have sought to understand the molecular mechanism of fetal damage since 1961, and more than thirty hypotheses to explain the damage have been put forward.
Different effects of ( S ) - and ( R ) -enantiomer

Thalidomide is a chiral compound that was sold as a racemate . The sedative effect is attributed to the (+) - ( R ) - enantiomer , the teratogenic effect is attributed to the (-) - ( S ) -enantiomer. By deuteration of the stereocenter thalidomide analogs could be stabilized successfully stereochemically.
Pharmacokinetics
Thalidomide is poorly soluble in the gastrointestinal tract and so absorption is slow and extensive in humans after oral intake. At a dose of 200 mg, a peak serum concentration of 1–2 mg / L is reached after 3 to 4 hours. The elimination half-life is about 6 hours, which, since the elimination is rapid, roughly reflects the absorption ratios. Thalidomide is hardly metabolized by the liver. However, its amide bonds are cleaved by hydrolysis . The hydroxylation of the molecular rings of thalidomide is species-dependent and of little importance in humans.
The two enantiomers convert into each other in the human body with a half-life of 2 to 6 hours - they racemize. Since the ( S ) -enantiomer is eliminated much faster than its ( R ) -configured counterpart, an equilibrium excess remains in the blood in the latter.
synthesis
Grünenthal's first synthesis variant is based on phthalic anhydride , which in the first step is converted into pyridine with L - glutamic acid. The resulting dicarboxylic acid intermediate is then cyclized to the anhydride in the presence of acetic anhydride. The target compound then results from a reaction with urea . This synthesis sequence results in a racemate .
New indications
In clinical studies, thalidomide has shown therapeutic effects in various diseases due to its anti-inflammatory (anti-inflammatory), antineoplastic (tumor-inhibiting) and antiangiogenic (reduction of new blood vessel formation) effectiveness. These include skin diseases ( erythema nodosum leprosum , lupus erythematosus ), multiple myeloma and autoimmune diseases . When used correctly, the active ingredient was therefore rated as a helpful therapy for patients for whose diseases no other adequate treatment exists. It was also used successfully for the treatment of aphthae in HIV-Patients used.
In prostate cancer patients who underwent intermittent hormone blockade , the administration of thalidomide resulted in an increase in the time it took for the PSA to rise again (as a sign that the cancer was reactivating).
leprosy
In 1964, the Israeli dermatologist Jacob Sheskin, who administered Contergan from residual stocks to a leprosy patient, found that her ulcers had receded significantly the next day. As a result of this discovery, thalidomide was tested for this indication, primarily in South American countries such as Colombia and Brazil .
However, the drug was also given to pregnant women, and malformations again occurred in newborns. Since the illiteracy rate in Brazil is high in some sections of the population, many women misunderstood the label with a crossed out pregnant woman on the packaging as a birth control pill . This resulted in a new generation of seriously malformed children in Brazil. As a result, Brazil initially banned the release of thalidomide to people of reproductive age. In Brazil there are a total of 480 recognized cases of thalidomide damage. Today, thalidomide has become the standard prescription medication for leprosy in Brazil; it is considered to be the most effective remedy for the disease.
In 1998, the US Food and Drug Administration approved a positive application for approval for a drug containing thalidomide for the treatment of erythema nodosum leprosum (ENL), a particularly severe form of leprosy. Marketing authorization holder Celgene has to ensure strict safety precautions during therapy. A patient who takes part must accept and adhere to strict requirements.
Multiple myeloma
The indication has now been expanded to include the treatment of multiple myeloma . After thalidomide had already been designated as a so-called "drug for rare diseases" ( orphan drug ) in 2001 and entered as such in the corresponding EU community register, the EU Commission granted approval for an oral dosage form (50 mg hard capsules) in April 2008 Combination therapy with melphalan and prednisone in patients with previously untreated multiple myelomas aged 65 and over or in patients for whom high-dose chemotherapy cannot be considered (marketing authorization holder: Pharmion Ltd.).
In 2019, a Cochrane review was published on the benefits and harms of some drug combination therapies for the primary treatment of multiple myeloma. This form of therapy is used in patients who are not eligible for a stem cell transplant . A drug combination with thalidomide was part of the review article. This combination of thalidomide, melphalan and prednisolonelikely to cause a prolongation of overall survival compared to therapy with melphalan and prednisolone alone. Thalidomide plus melphalan and prednisolone may cause a marked increase in progression-free survival. The administration of thalidomide plus melphalan and prednisolone probably lead to a marked increase in polyneuropathies. Thalidomide plus melphalan and prednisone caused a marked increase in patient discontinuation due to adverse events. Further research results are needed to assess the safety, effectiveness and impact on the quality of life of this form of therapy.
Myelodysplastic Syndrome
The administration of thalidomide for the treatment of myelodysplastic syndrome is being examined in multicenter studies. A precise mechanism of action is not yet known, but it may be the inhibition of tumor necrosis factor (TNF-α) and the stimulation of cytotoxic T cells . Preliminary data show that thalidomide can improve peripheral blood cell counts in many patients. Some patients achieve complete blood count normalization.
Crohn's disease
Thanks to its strong immunomodulatory effect, thalidomide was also able to show effects in the treatment of children and adolescents with Crohn's disease who have not responded to previous drugs. As an immunosuppressant , the active ingredient was able to achieve long-lasting remission in a number of patients .
distribution
Apart from the USA (since 1998), thalidomide is currently in Australia (since October 2003), New Zealand (since December 2003), Turkey (since June 2004), Israel (since September 2004) and in the member states of the EU (since April 2008 ) authorized. In the USA, China, Japan, Korea and Taiwan, thalidomide is marketed by Celgene. This also produces lenalidomide, which is similar to thalidomide . The British company Pharmion had acquired the marketing rights from Celgene for all other countries . At the beginning of 2008, however, Celgene took over Pharmion with its entire product portfolio. Formerly out-licensed products such as B. Thalidomide will now be managed exclusively by Celgene again. The application takes place under strict security guidelines.
In Germany, the dispensing of medicinal products containing thalidomide is regulated by Section 3a of the Medicinal Prescription Ordinance and is subject to official monitoring. The doctor must therefore use the so-called T-prescription for the prescription . Only one preparation may be prescribed per T-prescription. The doctor must ensure that the safety measures in accordance with the current product information are observed, in particular that a pregnancy prevention program is carried out if necessary. He must also note on the prescription whether the treatment is inside or outside ( off-label use) of the respective approved areas of application. Medicines containing thalidomide or lenalidomide may not be placed on the market by mail order (Section 17, Paragraph 2b of the Pharmacy Business Regulations ).
The company Chemie Grünenthal GmbH , which once produced Contergan, gave away remaining stocks of the product to scientists until the end of 2003. The company, which is now based in Aachen , no longer sells thalidomide.
See also
- Contergan (film) , cinematic implementation from 2006
- Contergan foundation for disabled people
literature
- K. Roth: An unending chemical history , in: Chemistry in our time , year 2005, No. 39, pp. 212-217, Wiley-VCH , Weinheim, doi: 10.1002 / ciuz.200590038 .
- Catia Monser: Contergan, Thalidomide. Misfortunes never come singly. Eggcup, Düsseldorf 1993, ISBN 3-930004-00-3 , book of a person affected.
- Beate Kirk: The Contergan case. An inevitable drug disaster? On the history of the drug thalidomide. Wissenschaftliche Verlagsgesellschaft, Stuttgart 1999, ISBN 3-8047-1681-4 (= Greifswald writings on the history of pharmacy and social pharmacy , Volume 1, also dissertation at the University of Greifswald , 1998).
- Niklas Lenhard-Schramm: The state of North Rhine-Westphalia and the Contergan scandal. Health Oversight and Criminal Justice in the "Long Sixties" . Vandenhoeck & Ruprecht, Göttingen 2016, ISBN 978-3-525-30178-4 .
- Ludwig Zichner , MA Rauschmann, KD Thomann et al. (Ed.): The Contergankatastrophe . A balance sheet after 40 years. In: Yearbook of the German Orthopedic History and Research Museum, Frankfurt am Main . Volume 6. Steinkopff, Darmstadt 2005, ISBN 978-3-7985-1479-9 .
- Henning Sjöstrom, Robert Nilsson: Thalidomide and the Power of the Drug Companies. Penguin Books, 1972; German translation: Contergan or the power of the pharmaceutical companies. VEB Verlag Volk und Gesundheit, Berlin (East) 1975.
- Takumi Ito et al .: Identification of a Primary Target of Thalidomide Teratogenicity. In: Science 327 (2010) no. 5971, pp. 1345-1350, doi: 10.1126 / science.1177319 .
- Klaus-Michael Wilsmann, Kai Zwingenberger: The contrasting history of thalidomids. Thalidomide and its fields of application. In: the scales. Magazine of Grünenthal GmbH, Aachen. Volume 35, 1996, No. 2, p. 66 f.
- Klaus-Dieter Thomann: The Contergan catastrophe: The deceptive security of “hard” data . In: Deutsches Ärzteblatt . tape 104 , no. 41 . Deutscher Ärzte-Verlag , October 12, 2007, p. A-2778 / B-2454 / C-2382 .
- A. Preuschhof, Introduction of T-Prescription - Drug Law Regulations for Dispensing Medicinal Products containing thalidomide or lenalidomide , Pharmazeutische Zeitung 2009, 406.
Web links
- Contergan - Thalidomid : extensive annotated link collection
- Representation from the perspective of the Contergan manufacturer Grünenthal
- Entries in the NIH study registry
- Discovery and development of thalidomide and its analogs
- Peter-Philipp Schmitt: "Only one tablet on the bus" . In: FAZ of May 13, 2008
- Contergannetzwerk Deutschland e. V. (CND)
Individual evidence
- ↑ a b c d e Entry on thalidomide in the GESTIS substance database of the IFA , accessed on January 9, 2019(JavaScript required) .
- ↑ G. Mall, E. Vosseler: Clinical experiences with "Persedon". In: DMW - German Medical Weekly. 75, 1950, p. 1084, doi: 10.1055 / s-0028-1117619 .
- ↑ a b Wolf-Dieter Müller Jahnke, Christoph Friedrich, Ulrich Meyer: Arzneimittelgeschichte, p. 146 . 2. revised and exp. Edition. Wissenschaftliche Verlagsgesellschaft mbH, Stuttgart 2005, ISBN 978-3-8047-2113-5 .
- ^ A b c d e William Silverman, MD: The Schizophrenic Career of a "Monster Drug" . In: Pediatrics . 110, No. 2, April 22, 2002, pp. 404-406. doi : 10.1542 / peds.110.2.404 .
- ↑ a b T. Stephens and R. Brynner: Dark Remedy - The Impact of Thalidomide and Its Revival as a Vital Medicine . Cambridge, MA; Perseus Publishing; 2001, ISBN 978-0-7382-0590-8 .
- ^ Bangen, Hans: History of the drug therapy of schizophrenia. Berlin 1992, ISBN 3-927408-82-4 , p. 78.
- ^ Dietrich Böhm: The compensation of the Contergan children. P. 159, Vorländer Verlag Siegen, 1973, stated in: German Society for Sociology: Child-Body-Identities: Theoretical and Empirical Approaches to Cultural Practice and Social Change, p. 168 . Ed .: Heinz Hengst. Beltz Juventa Verlag, Weinheim 2003, ISBN 978-3-7799-0225-6 . Online: limited preview in Google Book search.
- ^ A b Klaus-Dieter Thomann: The Contergan catastrophe: The deceptive security of "hard" data . In: Deutsches Ärzteblatt . tape 104 , no. 41 . Deutscher Ärzte-Verlag , October 12, 2007, p. A-2778 / B-2454 / C-2382 .
- ↑ W. von Lenz, K. Knapp: Die Thalidomid-Embryopathie. In: German Medical Weekly . Stuttgart, 1962, 87 (24): 1232-1242.
- ↑ See Klaus Huhn: According to the report, superfluous. Why there was no thalidomide in the GDR.
- ↑ See also in the GDR there were children with deformities. Bundesverband: Those affected do not want any public.
- ↑ Cf. Angelika Overath: Das Janusgesicht. Contergan. In: NZZ Folio . 04/01, topic: pills .
- ↑ See Stefanie Hallberg: From horror to bearer of hope. Comeback of the thalidomide active ingredient.
- ↑ See Contergan politically not an issue. The deadline has expired.
- ↑ See Linda Bren: Frances Oldham Kelsey: FDA Medical Reviewer Leaves Her Mark on History. In: FDA Consumer Magazine. March / April 2001.
- ↑ Press release of the Federal Association ( Memento from February 24, 2016 in the Internet Archive ).
- ↑ Grünenthal boss meets thalidomide victims for the first time , aachener-zeitung.de, December 10, 2007
- ↑ Signature campaign .
- ^ Requirements catalog of the federal association ( Memento of November 13, 2010 in the Internet Archive ), in addition to other associations exist.
- ↑ Press release from the Ministry of Family Affairs on the doubling ( memento of April 15, 2009 in the Internet Archive ).
- ↑ Federal Association for Doubling ( Memento from January 1, 2015 in the Internet Archive )
- ↑ Grünenthal press release: "Grünenthal offers Contergan sufferers 50 million euros - the solution should improve the living situation of those affected" Press release from Grünenthal GmbH (PDF).
- ↑ Press release RP-Online: "Grünenthal pays again 50 million euros for Contergan victims" ( Memento from July 16, 2009 in the Internet Archive ) RP Online.
- ↑ Ito T, Ando H, Suzuki T, et al. : Identification of a primary target of thalidomide teratogenicity . In: Science . 327, No. 5971, March 2010, pp. 1345-50. doi : 10.1126 / science.1177319 . PMID 20223979 .
- ↑ a b Ito T, Ando H, Handa H: Teratogenic effects of thalidomide: molecular mechanisms . In: Cell. Mol. Life Sci. . 68, No. 9, 2011, pp. 1569-79. doi : 10.1007 / s00018-010-0619-9 . PMID 21207098 .
- ^ Contergan: cause of malformations found. Ärzteblatt, August 3, 2018, accessed March 1, 2010 .
- ↑ Katherine A Donovan et al .: Thalidomide promotes degradation of SALL4, a transcription factor implicated in Duane Radial Ray Syndrome . In: Biochemistry and Chemical Biology . August 1, 2018, doi : 10.7554 / eLife.38430.001 .
- ↑ Miller, MT, Strömland, K .: Teratogen Update: Thalidomide: A Review, With a Focus on Ocular Findings and New Potential Uses . In: Teratology . 60, 1999, pp. 306-321. doi : 10.1002 / (SICI) 1096-9926 (199911) 60: 5 <306 :: AID-TERA11> 3.0.CO; 2-Y . PMID 10525208 .
- ^ Vaughan Monamy: Animal Experimentation. Cambridge University Press, 2009, ISBN 978-0-521-87879-1 , p. 84.
- ↑ Recommendations for the use of thalidomide in patients with multiple myeloma, consensus group of the German Society for Hematology and Oncology and the Austrian Society for Hematology and Oncology (PDF; 313 kB)
- ↑ Rote-Hand-Brief Thalidomid ( Memento from October 25, 2013 in the Internet Archive ) (PDF; 379 kB).
- ↑ As of 2000. Stephens TD, Bunde CJ, Fillmore BJ: Mechanism of action in thalidomide teratogenesis . In: Biochemical Pharmacology . 59, No. 12, 2000, pp. 1489-1499. doi : 10.1016 / s0006-2952 (99) 00388-3 . PMID 10799645 .
- ↑ T. Eriksson, S. Björkman, B. Roth, P. Höglund: Intravenous formulations of the enantiomers of thalidomide: pharmacokinetic and initial pharmacodynamic characterization in man . In: Journal of Pharmacy and Pharmacology . 52, No. 7, 2000, pp. 807-817. doi : 10.1211 / 0022357001774660 . PMID 10933131 .
- ↑ a b Jacques V, Czarnik AW, Judge TM, Van der Ploeg LH, DeWitt SH: Differentiation of antiinflammatory and antitumorigenic properties of stabilized enantiomers of thalidomide analogs . In: Proceedings of the National Academy of Sciences USA . 112, No. 12, 2015, pp. E1471–9. doi : 10.1073 / pnas.1417832112 . PMID 25775521 . PMC 4378388 (free full text).
- ↑ Teo SK, Colburn WA, Tracewell WG, Kook KA, Stirling DI, Jaworsky MS, Scheffler MA, Thomas SD, Laskin OL: Clinical Pharmacokinetics of thalidomide . In: Clinical Pharmacokinetics . 43, No. 5, 2004, pp. 311-27. doi : 10.2165 / 00003088-200443050-00004 . PMID 15080764 .
- ^ Pischek G, Kaiser E, Koch H: The thin layer chromatography of thalidomide and its hydrolysis products . In: Mikrochimica Acta . No. 3, 1970, pp. 530-5. doi : 10.1007 / BF01224156 . PMID 5489943 .
- ↑ Eriksson T, Björkman S, Roth B, Björk H, Höglund P: Hydroxylated metabolites of thalidomide: formation in-vitro and in-vivo in man . In: Journal of Pharmacy and Pharmacology . 50, No. 12, 1998, pp. 1409-16. PMID 10052858 .
- ↑ M. Reist, PA Carrupt, E. Francotte, B. Testa: Chiral inversion and hydrolysis of thalidomide: mechanisms and catalysis by bases and serum albumin, and chiral stability of teratogenic metabolites . In: Chemical Research in Toxicology . 11, No. 12, 1998, pp. 1521-8. doi : 10.1021 / tx9801817 . PMID 9860497 .
- ↑ T. Eriksson, S. Björkman, B. Roth, A. Fyge, P. Höglund: Stereospecific determination, chiral inversion in vitro and pharmacokinetics in humans of the enantiomers of thalidomide . In: Chirality . 7, No. 1, 1995, pp. 44-52. doi : 10.1002 / chir.530070109 . PMID 7702998 .
- ^ Patent GB 768 821 (Chemie Grünenthal; February 20, 1957).
- ↑ Kunz, W. et al .: Pharmaceutical Research 6 (1956) 426-430.
- ^ A b Silverman WA: The schizophrenic career of a "monster drug" . In pediatrics . 2002 Aug; 110 (2 Pt 1), pp. 404-406, PMID 12165600 .
- ↑ L. Calabrese, AB Fleischer: Thalidomide: current and potential clinical applications. In: The American journal of medicine. Volume 108, Number 6, April 2000, pp. 487-495, PMID 10781782 , (Review).
- ↑ Jeffrey M. Jacobson et al .: Thalidomide for the treatment of oral aphthous ulcers in patients with human immunodeficiency virus infection. In: New England Journal of Medicine , Volume 336, No. 21, 1997, pp. 1487-1493, doi: 10.1056 / NEJM199705223362103 .
- ↑ WD Figg, MH Hussain et al. a .: A double-blind randomized crossover study of oral thalidomide versus placebo for androgen dependent prostate cancer treated with intermittent androgen ablation. In: The Journal of Urology . 181, 2009, pp. 1104-1113. doi: 10.1016 / j.juro.2008.11.026 . PMID 19167733 . PMC 2838198 (free full text).
- ↑ See S. Hallberg: From horror to hope: Comeback of the thalidomide thalidomide.
- ↑ S. Singhal, J. Mehta et al. a .: Antitumor activity of thalidomide in refractory multiple myeloma. In: The New England Journal of Medicine. Volume 341, Number 21, November 1999, pp. 1565-1571, doi: 10.1056 / NEJM199911183412102 . PMID 10564685 .
- ↑ T. Hideshima, D. Chauhan et al. a .: Thalidomide and its analogs overcome drug resistance of human multiple myeloma cells to conventional therapy. In: Blood . Volume 96, Number 9, November 2000, pp. 2943-2950, PMID 11049970 .
- ↑ Appendix: Thalidomide Celgene 50 mg hard capsules May 6, 2008 (PDF, English; 516 kB).
- ↑ Vanessa Piechotta, Tina Jakob, Peter Langer, Ina Monsef, Christof Scheid: Multiple drug combinations of bortezomib, lenalidomide, and thalidomide for first-line treatment in adults with transplant-ineligible multiple myeloma: a network meta-analysis . In: Cochrane Database of Systematic Reviews . November 25, 2019, doi : 10.1002 / 14651858.CD013487 .
- ↑ Marzia Lazzerini, Stefano Martelossi u. a .: Effect of Thalidomide on Clinical Remission in Children and Adolescents With Refractory Crohn Disease. In: Journal of the American Medical Association . 310, 2013, p. 2164, doi: 10.1001 / jama.2013.280777 .
- ↑ Press release from Celgene Inc. ( Memento from July 11, 2012 in the web archive archive.today ).
- ↑ Federal Institute for Drugs and Medical Devices: Announcement on the Medicinal Prescription Ordinance of June 17, 2011 , Federal Gazette p. 99 of July 6, 2011, p. 2415.
- ↑ Medicinal Prescription Ordinance (AMVV) § 3a .
- ↑ Hubert Giess: DRUGS: Comeback for thalidomide. In: Focus Online . May 2, 1994, accessed October 10, 2015 .