Stem cell
When stem cells are generally cells of the body , made up, in different cell types or tissues differentiate can. Depending on the type of stem cell and how it is influenced, they have the potential to develop into any tissue (embryonic stem cells) or into certain specified types of tissue (adult stem cells).
Stem cells are able to generate daughter cells , which in turn have stem cell properties, but also those with greater differentiation. A not yet fully understood mechanism of asymmetrical cell division enables them to do this . The biological milieu in which they are located determines the respective fate of the cells.
Stem cells are distinguished primarily by their ontogenetic age and their differentiation potential: the ontogenetically earliest stem cells are the pluripotent embryonic stem cells, from which the primitive germ stem cells as well as the somatic stem and progenitor cells (or progenitor cells) later emerge. Phylogenetically , the stem cells go back to the last common eukaryotic ancestor (LECA).
Even plants have stem cells. These are located at the tip of the shoot in the so-called apical meristem and at the root tips in the root meristem. In contrast to almost all animal and human cells, practically all cells in plants have the ability to regenerate a complete organism .
Embryonic stem cells
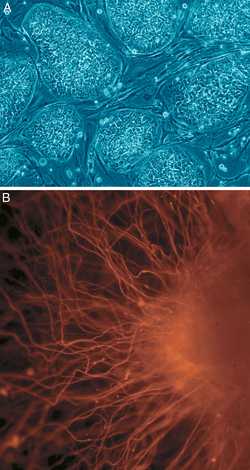
Embryonic stem cells (ES cells) in vivo and in vitro in a position, in cells of all three germ layers ( endoderm , ectoderm and mesoderm ), and in germ line cells to differentiate . They are therefore called pluripotent. ES cells are for experimental purposes - after fertilization of the egg cell in the embryo -Entwicklungsstadium the blastocyst from the inner cell mass - (ICM also embryoblast won called).
ES cells were first isolated in 1981 - from mouse blastocysts . They tend to differentiate spontaneously in vitro . This can be prevented by factors that promote the self-renewal of the cells. Several such substances have been identified since the late 1980s, mainly by the Austin Smith group in Edinburgh. In principle, ES cells can therefore be multiplied indefinitely, which is also due to the high activity of the enzyme telomerase . This distinguishes them from other (so-called primary) body cells, which usually stop their division activity in the culture dish after a short time ( replicative senescence ).
In the embryo, ES cells form the precursors for all body cells, but not for the embryonic parts of the placenta . In 2003 it was also possible for the first time to show in the mouse model that ES cells can also differentiate into germ cells (gametes, in the study mentioned above on egg cells).
A remarkable property of mouse ES cells is that they can be reintroduced into preimplantation embryos and, once transferred into pseudopregnant animals, are involved in building all fetal tissues. This can be used for the targeted deactivation of certain genes in mice. Knock-out mice , which can be produced significantly faster using ES cells than with conventional techniques, are of great value for the study of gene functions and are also used as human disease models.
Furthermore, ES cells can be differentiated more or less specifically into a wide variety of cell types in vitro , e.g. B. in nerve cells. This area was revitalized in particular from 1998 with the first establishment of human ES cells (hES cells) by James Thomson . hES cells are generally obtained from so-called surplus embryos, which were created by in vitro fertilization , are no longer needed for reproductive purposes and are therefore stored frozen. The main interest in research on hES cells is the differentiation into specialized cells in order to make them available for possible cell replacement therapies.
ES cells may one day be useful as substitute materials in medicine. The disease Parkinson's disease was differentiated using human embryonic stem cells are at least already treated in animal experiments. These and other findings - mainly obtained in animal models - have not yet been confirmed in larger mammals. Nevertheless, the US company Geron has already announced the first clinical studies using hES cells for the treatment of spinal cord injuries for 2008. In principle, however, the results so far still require a strict review, so that, in contrast to the adult stem cells (see below), a possible clinical application is still a long way off. For example, hES cells can form tumors after transplantation into test animals, so that before clinical use it must be ensured that the transplants no longer contain any undifferentiated hES cells. The immunologically induced rejection of corresponding transplants by the recipient could also be of great relevance, a problem that is well known from transplant medicine.
An ethically largely harmless source of embryonic stem cells could be unfertilized egg cells that are produced during fertility treatments and are not fertilized. These egg cells can be stimulated to divide by electrical or chemical stimuli ( parthenogenesis ), from which heart muscle cells, for example, can be grown.
Clone
In addition to obtaining ES cells from IVF blastocysts, it is also possible to obtain ES cells by cloning embryos. The basis for this possibility was the first successful cloning of a mammal in 1996, the sheep "Dolly" . Using this technology, an early embryo can be created from which ES cells can be created by transferring the cell nucleus from a body cell into an unfertilized egg cell that has been freed from the internal cell mass. If applied to humans, the method would have the advantage that genetically (and thus immunologically) identical ES cells would be available to the donor.
Research on embryonic stem cells also attracted the public's attention due to falsified results: In 2004, the research team led by the South Korean veterinarian Hwang Woo-suk had succeeded for the first time in cloning a human embryo and in this way obtaining stem cell lines ( Therapeutisches Cloning ). A publication followed in 2005, also in the respected journal Science , according to which the world's first custom-made embryonic stem cells had been established for seriously ill patients. Both publications turned out to be largely falsified.
A possible breakthrough in therapeutic cloning of primates are the results of a US research team headed by Shoukhrat Mitalipov , presented for the first time in June 2007. The team succeeded in cloning rhesus monkeys and obtaining two lines of embryonic stem cells from the embryos. The same procedure was used as with the “Dolly” sheep. These results were independently confirmed on November 14, 2007.
Ethical controversy
The way in which human embryonic stem cells (often abbreviated: hES cells; h = human) are obtained after in-vitro fertilization led to a heated ethical debate that continues to this day.
The use of human embryonic stem cells in research and medicine is rejected by a section of society because their extraction requires the destruction of early human embryos (“consuming” embryo research). Basically, the discussion in Germany is primarily about the question of whether the early embryo as a human being falls under the protection of dignity under the Basic Law and thus its life should not be subject to any considerations. The opponents of stem cell research often use the so-called SKIP arguments to convince them of their position. Proponents of research on embryonic stem cells, on the other hand, often argue that research with human embryonic stem cells may have a very high positive potential: The scientists hope, among other things, for a cure for serious diseases ( Parkinson's disease , diabetes mellitus , paraplegia ) and the possibility to regrow destroyed organs. At the moment, however, there are only concrete indications of such therapeutic successes from animal experiments with rodents.
The German Bishops' Conference (Catholic) is of the opinion that human life is present from the fertilization of the egg cell. Accordingly, from fertilization onwards, humans are entitled to a dignity that prohibits destruction of the fertilized egg cell. Likewise, the Congregation for the Doctrine of the Faith under Joseph Ratzinger expressed itself in 1987 in the instruction Donum Vitae . Pope John Paul II expressed himself a. a. on this in the encyclical Evangelium Vitae 1995 and took up the relevant aspects of the question from Donum Vitae. The Pontifical Academy for Life issued a corresponding declaration in 2000, which deals in particular with technical issues relating to stem cell research. One of the last Catholic statements in this regard was the Declaration Dignitas Personae of the Roman Congregation for the Doctrine of the Faith.
There are also theological beliefs that the early embryo is already animated. This embryo has a soul and is therefore under special protection. With reference to this, some proponents of stem cell research point out that following Thomas Aquinas in the Catholic Church up to modern times it was believed that the ensouling of the embryo takes place gradually (successive ensouling) and the highest form of the soul , the "anima intellectiva “, Only transmitted about three months after conception . It was only in the Bull Apostolicae Sedis (1869, under Pius IX ) that the Catholic Church finally gave up the doctrine of the full Incarnation on the 80th day.
The Protestant Church, on the other hand, would like to support the finding of dialogue in order to find a consensus on this question. The representatives of the Protestant Church agree that no embryos may be produced for the isolation of embryonic stem cells. It has not yet taken a position on the question of the general use of existing embryonic stem cells; she would like to "leave basic research on embryonic stem cells behind" as quickly as possible.
In the case of a dispute about the moral status of the embryo, the following points in time are summarized and supplemented:
- Moment of conception (nuclear fusion)
- Differentiation of the embryo and placenta
- Point in time from which multiple births are excluded
- Nidation
- First formation of brain cells
- birth
- First weeks after giving birth
Legal situation
European Court of Justice
According to the European Court of Justice (ECJ) on October 18, 2011, human embryonic stem cells, for the production of which embryos have to be destroyed , may not be patented, since fertilized egg cells are already human life. With this judgment, the ECJ decided a patent dispute between the neurobiologist Oliver Brüstle and Greenpeace .
Germany
According to the Embryo Protection Act , it is forbidden in Germany to produce, clone or destroy human embryos (including blastocysts, which serve as a source for embryonic stem cells) for research purposes . However, research on imported embryonic stem cells is possible under certain conditions and was initially regulated by the Stem Cell Act of July 2002. This law, and in particular the regulation contained therein, that only embryonic stem cells obtained before January 1, 2002 (key date regulation) could be imported into Germany, was controversial from the start. In the spring of 2008, the German Bundestag debated an amendment to the Stem Cell Act which, in addition to postponing the deadline, proposed the complete approval of imports and the ban on stem cell research with embryonic stem cells in group applications. On April 11, 2008, the German Bundestag resolved a new deadline so that stem cells obtained before May 1, 2007 can now be imported.
Austria
In Austria, research on imported pluripotent embryonic stem cells is permitted without restrictions under current law. This also applies to therapeutic cloning. However, according to Section 9 of the Reproductive Medicine Act, the extraction of embryonic stem cells would be prohibited if this took place in Austria. The use of totipotent stem cells is only allowed for reproductive purposes.
Poland
In Poland research on human embryos is punished if it leads to the destruction of the embryo in vitro. This is equated with abortion and can be punished with imprisonment of up to three years. There are no binding legal regulations for research with embryonic stem cells from abroad. The directive of January 13, 2004 is currently in force; it states that “research on embryonic stem cells should only be permitted if the aim is to save human life.” However, the definition of “human life” leaves a lot of room for interpretation.
Switzerland
In Switzerland, scientists are allowed to extract stem cells from surplus human embryos (i.e. those that are not used in reproductive medicine) and conduct research with the cells. The Swiss Federal Council passed a corresponding law in February 2005 after more than 66 percent of Swiss voters had voted in favor of this law in a referendum. The prerequisite according to Art. 5 ff. Stem Cell Research Act is that the donor couple has given their consent, that the donation is free of charge and that none of the persons for whose research project the cells are obtained was involved in the reproductive process. Cloning of human cells is prohibited.
Great Britain
The UK allows both the creation of human embryonic stem cells and the cloning of human embryos for research purposes.
United States
In the US , research on embryonic stem cells was only funded with federal government funds until the end of 2009 if the stem cell lines used existed before August 2001. In July 2006, the US Senate and the House of Representatives voted to lift this restriction; however, President Bush has vetoed this. However , this restriction did not apply to state research funding or privately funded research. Therefore California was able to decide in a referendum in 2004 to support embryonic stem cell research with three billion dollars. In March 2009, President Obama announced that he would again use state funds to support stem cell research. This announcement was implemented in December 2009 by the responsible authorities by allowing the use of an initial 13 lines of embryonic stem cells. In August 2010, this funding was blocked again by a US court because it violated a law that prohibits the destruction of human embryos.
Postembryonic stem cells
The group of postembryonic stem cells includes all those stem cells that occur in the organism of mammals after the embryonic development is complete. According to their ontogenetic age , they are further divided into fetal, neonatal, and adult stem cells.
According to current knowledge, the differentiation potential of postembryonic stem cells is limited to the maturation of genetically determined tissues such as the skin , the liver or the haematopoietic system . In contrast to ES cells, they are therefore no longer referred to as pluripotent but only as multipotent .
Adult stem cells
While embryonic stem cells only occur in the early embryo, adult stem cells (from Latin for adult , also called somatic ) are present in the organism after birth (postnatal stage). New specialized cells are formed from these cells throughout the life of the organism. Adult stem cells, which are found in organs (especially in the bone marrow , in the skin , but also in adipose tissue, in the umbilical cord and in the umbilical cord blood , in the menstrual blood , in the brain , the liver or the pancreas ) generally have a cell culture Significantly lower self-renewal capacity and a restricted differentiation potential than embryonic stem cells. Neural stem cells can develop into all cell types of the nerve tissue (neurons, glia, etc.), but not into liver or muscle cells. A differentiation potential of certain stem cell types (ability to transdifferentiate ) beyond the cotyledon has been observed in various studies, but is highly controversial.
Adult stem cells are available in every individual, so the prospect of replacement by the body's own, i.e. H. autologous cells are given and they are therefore suitable for the technology of tissue engineering . The tendency to malignant degeneration also seems to be lower with implantation of adult stem cells than with embryonic stem cells. A degeneration has not yet been observed in the clinical use of adult stem cells.
The extraction of adult stem cells and progenitor cells from the bone marrow takes place by puncturing the pelvic bone under general anesthesia or, more recently, by means of stem cell apheresis . The extraction of cord blood is carried by the umbilical cord of the child, the rest still in umbilical cord and from collection, placental blood contained. Multipotent stem cells are obtained from the skin by means of a small skin biopsy under local anesthesia in the outpatient area. The stem cells are then removed from the tissue and are available for further use or storage for many years as a preventative measure , as is already being offered by German companies today. As part of a normal autologous blood donation, circulating endothelial progenitor cells can be obtained. The potential of these autologous (endogenous) precursor cells for the therapy of cardiovascular diseases is currently being investigated in clinical studies. The advantage of using autologous progenitor cells lies in the lack of immunogenicity; H. the transplanted cells are not recognized as foreign by the immune system .
In 2009, the Dutch immunologist and molecular biologist Hans Clevers developed a process for the reproduction of adult stem cells with which he can grow rudimentary organs in miniature format (so-called organoids), for which he received the Körber European Science Prize, endowed with 750,000 euros, in September 2016 . The winner is particularly interested in the signals that stimulate stem cells to divide. Using a receptor he discovered (Lgr5), which is only found in stem cells, he was able to isolate them from removed intestinal tissue. Mini-organs can be created from tumor tissue on which drugs can be tested. In 2013 Clevers was able to use genome editing to rid the intestinal stem cells of patients suffering from the hereditary disease cystic fibrosis of this genetic defect.
Artificially reprogrammed stem cells
Kazutoshi Takahashi and Shinya Yamanaka from the University of Kyoto and researchers from the University of Wisconsin reported in Cell and Science in 2006 and 2007, respectively , that they had succeeded in converting body cells of adult humans into induced pluripotent stem cells (iPS). Four central, dormant developmental genes were activated in the cells so that they were returned to a kind of embryonic state. From the artificially reprogrammed stem cells, the researchers were able to put matured cells in the Petri dish, e.g. B. myocardium and nerve cells grow.
For the first reprogramming, the genes Oct-4 , Sox-2 , c-Myc and Klf-4 were channeled into the cells with retroviruses ( transduction ). In animal experiments, one fifth of the mice used developed tumors , presumably because two of the genes used can promote cancer (so-called proto-oncogenes ). In order to exclude a risk from introduced cancer-promoting genes in medical applications, alternative methods of reprogramming are being sought. Research is carried out u. a. on small molecules (e.g. peptides ) that activate the stem cell genes naturally occurring in the cell's genetic material. In order to avoid tumor formation, researchers are also trying to avoid the method of gene injection with retroviruses and the use of the proto-oncogenes c-Myc and Klf-4 by introducing non-integrating adenoviruses and alternative genes ( Nanog , lin-28 ).
In contrast to retroviruses, adenoviruses do not integrate the desired gene sequence into the genome of the host cell, which means that the integrity of the host genome is preserved.
In addition, it was possible to generate iPS cells by transfecting only one pluripotency gene from cells that naturally express the other three genes.
In December 2007, researchers led by Jacob Hanna from the Whitehead Institute for Biomedical Research in Cambridge , USA , reported that iPS cells had been used to cure mice that had suffered from sickle cell anemia . In the iPS cells obtained from the tail by means of reprogramming, the researchers replaced the modified gene that triggers sickle cell anemia with the healthy genetic make-up using homologous recombination . From the stem cells treated in this way, blood-forming progenitor cells were cultured, which can develop into various blood cells and cells of the immune system. The progenitor cells were transplanted into the diseased mice, where they evidently grew into healthy blood cells. As the team reports, the symptoms of the test animals almost completely disappeared as a result of the treatment. These and related publications should, however, be viewed with a certain degree of reservation. There are several unclear information about proteins, cellular biochemistry and cell processes, such as: B. to the homeobox protein NANOG or to c-Myc regarding cancer development.
Research in Germany a. a. Hans Schöler , Director at the Max Planck Institute for Molecular Biomedicine in Münster and Oliver Brüstle , Director of the Institute for Reconstructive Neurobiology at the University of Bonn on iPS cells. The two scientists jointly lead the stem cell research network NRW . Based on stem cell research, their expectations are based on the development of drugs that they see as particularly promising in the medium term.
Stem cell medicine
The blood-forming stem cells of the bone marrow have been used in the treatment of leukemia and lymphomas for over 40 years (see also stem cell transplantation ). During chemotherapy e.g. B. most rapidly growing cells are destroyed by cytotoxic components. This not only kills the cancer cells; the stem cells, which other body cells are supposed to repair, are also affected by the therapy. The blood-forming stem cells are particularly affected. Therefore, before chemotherapy, stem cells are obtained from the patient's bone marrow (by so-called autologous transplantation ) or from a suitable donor (so-called allogeneic transplantation ). After the chemotherapy treatment is complete, the blood-forming stem cells are injected. These stem cells then produce large amounts of red and white blood cells , which can keep the blood healthy and help fight off infections.
Non-hematopoietic adult stem cells have already been used successfully in individual studies for paralysis after spinal injuries and for Parkinson's disease . In successful clinical studies, stem cells from the bone marrow were able to help patients regenerate better after a heart attack or multiple sclerosis. In the meantime, multipotent stem cells have also been discovered in the skin , which potentially develop in all human organ tissues and can contribute to regeneration here.
An initial clinical test phase began in the USA in October 2010 to clarify whether successful experiments with paraplegic rats can be transferred to people who have recently had spinal cord injuries.
Stem cell research
Experiments on rats are currently successfully treating brain tumors by injecting adult stem cells. Scientists at Harvard University have genetically engineered the cells so that they convert another substance injected at the same time into a substance that kills cancer cells. The size of the tumors could be reduced by 80 percent.
Stem cells also appear to be able to regenerate cells that have been damaged by a heart attack . Columbia-Presbyterian University has succeeded in improving heart function by 33 percent after an infarction in mice by injecting bone marrow stem cells. The destroyed tissue regenerated 68 percent. However, it is now assumed that this is due to paracrine or other effects of the transplanted cells, but transdifferentiation of haematopoietic stem cells to cardio myocytes did not take place.
The use of autologous stem cells in heart damage is being investigated in clinical studies in various heart centers across Europe. The extent to which heart muscle cells are actually regenerated has not yet been clarified. In Germany u. a. Researched the benefits of stem cells for heart regeneration in a clinical study at the Frankfurt University Hospital . Adult stem cells are obtained here from blood by centrifugation, cultivated by subsequent application on fibronectin plates and selectively enriched on these; they adhere to the plates so that other cells can be washed away. After three days of cultivation, they can be detached from the plates and - with the help of suitable nutrient media - introduced into the heart. In a comparable way, adult stem cells can also be obtained from muscle tissue from the skin, but here the cultivation takes about 20 days, not three.
Another important area of application for adult stem cells is the regeneration of cartilage and bones. Various renowned research institutes in Israel, England and Slovenia have published relevant clinical data.
However, research with pluripotent stem cells suffered a massive setback in 2011, which in the worst case could lead to the end of the previous hope of regenerative medicine . Scientists from the University of California, the San Diego School of Medicine, and the Scripps Research Institute found severe genetic changes in pluripotent stem cell lines. According to this, human embryonic stem cells (hESC) and induced pluripotent stem cells (iPSC) show more genome aberrations than their normal cell counterparts. In the examined hESCs there were considerable duplications , while the iPSCs had considerable deletions . However, the impact of these genetic changes on potential clinical applications is still unclear.
On May 15, 2013, Cell reported that it was possible for the first time to obtain pluripotent human stem cells by means of cell nucleus transfer and to develop them into specialized cells of the pancreas as well as blood, heart, liver and nerve cells.
There are now new therapeutic options based on embryonic stem cells. As of April 2011, the only two US FDA (Food and Drug Administration) -approved patient trials have been running. Here elderly patients with dry macular degeneration (AMD) and younger patients with Stargardt Dystrophy (SMD) are treated with (RPE) cells, i. H. 50–200,000 retinal pigment epithelium (RPE) cells are injected into the retina of one eye. These cells are obtained from embryonic stem cells without destroying an embryo (patented blastomer technique, similar to PGD diagnostics). Over 40 patients have now been treated in four eye clinics in the USA and two in the UK. A peer-reviewed article was published in The Lancet in October 2014 . According to him, the majority of patients had significant improvements in vision. This has led the US FDA to approve a series of tests for younger patients as well. In principle, such experiments begin with older patients who already have an advanced disease of their visual performance. The primary concern is the safe use of the therapy.
Extraction of stem cells from the pulp of the milk teeth
The deciduous teeth are suitable as a source of stem cells. The cells in the pulp can be extracted, cultivated with a special growth agent and finally preserved for medical purposes. The stem cells can be used in dentistry for the regeneration of the dental pulp in adults. With the help of transplanting stem cells as part of tissue engineering , parts of the root canals can be renewed.
literature
Books
- Thomas Heinemann, Jens Kersten: stem cell research. Scientific, legal and ethical aspects. Status reports of the DRZE. Volume 4. Verlag Karl Alber, Freiburg 2007, ISBN 978-3-495-48196-7 .
- DRZE / Scientific department on behalf of the competence network stem cell research NRW (ed.): Dossier stem cell research. Central national and international legal guidelines and agreements as well as statements by national and international institutions.
- Elmar Brähler (Ed.): From the family tree to the stem cell. Reproductive medicine, prenatal diagnostics and human raw material. 2002.
- Achim Limbeck: Embryo Protection Act and research on human stem cells. A criminal investigation into research on human stem cells, especially against the background of the Embryo Protection Act , 2006, ISBN 3-9810745-9-9 . (Extensive reproduction of the state of research as well as the criminal law problems associated with the research).
- Eberhard J. Wormer : More knowledge about stem cells. With introductory contributions by Werner Kaufmann, Detlev Ganten, Gerd Kempermann. Lingen, Cologne 2003, ISBN 3-937490-00-0 .
- Mike S. Schäfer: Science in the media. The medialization of scientific topics . Wiesbaden: Publishing house for social sciences. 2007, ISBN 978-3-531-15592-0 .
- Anna M. Wobus u. a .: stem cell research and cell therapy. State of knowledge and general conditions in Germany. With contributions by Christine Hauskeller and Jochen Taupitz. Munich 2006, ISBN 3-8274-1790-2 .
- Gerd Kempermann : Humans need new cells. Stem cell research and the revolution in medicine. Piper Verlag, Munich 2008, ISBN 978-3-492-05179-8 .
Essays
- Michael Feld, Jürgen Hescheler: Stem cells: Potent cells. In: Spectrum of Science . May 2003, pp. 66-73.
- Michael Groß : The island of stem cell researchers. In: News from chemistry. 52 (12), 2004, pp. 1261-1263, ISSN 1439-9598 .
- Lars Grotewold: How do cells stay pluripotent? A quarter of a century of stem cell research. In: Naturwissenschaftliche Rundschau . 58 (8), 2005, pp. 413-419.
- Stem cells. In: Nature Volume 441, Issue 7097, from June 29, 2006, pp. 1059–1102 (a very detailed overview of the state of research, in English)
- Stephan Ernst : Man or Material? Theological-ethical remarks on the levels of discourse on stem cell research. In: Würzburger medical history reports 23, 2004, pp. 457-470.
- Davor Solter : From teratocarcinomas to embryonic stem cells and beyond: a history of embryonic stem cell research. In: Nature Reviews Genetics . (7), 2006, pp. 319-327.
- Peter Löser, Anna M. Wobus: Current developments in research with human embryonic stem cells. In: Naturwissenschaftliche Rundschau. 60 (5), 2007, pp. 229-237.
- Alexander A. Maximow: The lymphocyte as common stem cell of the different blood elements in the embryonic development and in the postfetal life of the mammals. Demonstration lecture given at the extraordinary meeting of the Berlin Hematological Society on June 1, 1909. In: Folia Haematologica. 8.1909, pp. 125-134 (now open access).
Web links
Historical
- Remarks to trace the scientific steps that eventually led to the recognition of the blood stem cell as an important entity of its own Notes from Theodor M. Fliedner in Stem Cells, Volume 16 (6), 1998, pp. 357-360.
Social
- "Stem cell research - a controversial topic for a demanding classroom"
- Competence network stem cell research NRW
- Focus on "Research with Human Embryonic Stem Cells" ( German Reference Center for Ethics in the Biosciences )
- Stemzell-Debatte.de: Information on the debate on embryonic and adult stem cells, therapeutic and reproductive cloning (Interests Community Critical Bioethics Germany)
- Article about the status of stem cells and the bioethical discussion in Germany in NOVO magazine .
- Andrew Siegel: Ethics of Stem Cell Research. In: Edward N. Zalta (Ed.): Stanford Encyclopedia of Philosophy .
Individual evidence
- ↑ Sean J. Morrison, Nirao M. Shah, David J. Anderson: Review: Regulatory mechanisms in stem cell biology. In: Cell . Volume 88, No. 3, 1997, pp. 287-298, doi: 10.1016 / S0092-8674 (00) 81867-X . PMID 9039255 . (Full text; PDF; 260 kB) (PDF)
- ^ Eugene V. Koonin: The origin and early evolution of eukaryotes in the light of phylogenomics. In: Genome Biology. 2010. 11: 209. doi: 10.1186 / gb-2010-11-5-209
- ^ Beddington, Robertson: An assessment of the developmental potential of embryonic stem cells in the midgestation mouse embryo. In: Development. Volume 105, 1989, pp. 733-737.
- ↑ K. Hubner et al. a .: Derivation of oocytes from mouse embryonic stem cells. In: Science . Volume 300, 2003, pp. 1251-1256.
- ↑ Passier, Mummery: Origin and use of embryonic and adult stem cells in differentiation and tissue repair. In: Cardivascular Research. Volume 58, 2003, pp. 324-335.
- ↑ Michael Didié u. a .: Parthenogenetic stem cells for tissue-engineered heart repair. In: Journal of Clinical Investigation. Volume 123, No. 3, 2013, pp. 1285-1298, doi: 10.1172 / JCI66854 .
- ↑ Monkey stem cells cloned . In: Nature . No. 447, June 20, 2007, p. 891.
- ↑ Kathrin Zinkant: Biomedicine: The clone test . In: Zeit Online . October 1, 2009 ( zeit.de [accessed on May 17, 2016] No. 46.): “An Australian research team genetically tested the primate stem cells and confirmed that they were definitely cloned ES cells from the experimental monkeys from Oregon. "
- ↑ G. Damschen, D. Schönecker: The moral status of human embryos. de Gruyter, Berlin 2002.
- ^ Comment by the DBK on the stem cell ruling of the European Court of Justice .
- ↑ The instruction Donum Vitae .
- ^ Encyclical Evangelium Vitae .
- ^ Declaration of the Pontifical Academy for Life .
- ^ Dignitas Personae .
- ↑ Statement on the postponement of the deadline for importing ES cells .
- ↑ Stephan Ernst: Man or Material? Theological-ethical remarks on the levels of discourse on stem cell research. In: Würzburger medical history reports 23, 2004, pp. 457-470; here: pp. 461–466.
- ↑ ECJ slows down stem cell researchers. on: www.orf.at October 18, 2011.
- ↑ www.bundestag.de ( Memento of March 10, 2009 in the Internet Archive ) Overview of the various draft laws for the Bundestag debate on February 14, 2008.
- ↑ Tagesschau : Bundestag loosens stem cell law ( memento of February 20, 2009 in the Internet Archive ) of April 11, 2008.
- ↑ austria.gv.at
- ↑ Witold Jacorzynski, Marcin Kozlowski: On the way (s) anywhere: The Polish stem cell research. In: Wolfgang Bender u. a .: Crossing borders: cultural, religious and political differences in the context of stem cell research worldwide = crossing borders. (= Darmstadt interdisciplinary contributions. 10). agenda-Verlag, Münster 2005, ISBN 3-89688-258-9 , p. 471.
- ↑ Switzerland allows the extraction of embryonic stem cells. on: www.3sat.de
- ↑ Denise Stevens: Embryonic stem cell research: will President Bush's limitation on federal funding put the United States at a disadvantage? A comparison between US and international law. In: Houston Journal of International Law. 25, No. 3, 2003, pp. 623-653. PMID 15199940 .
- ↑ Veto against stem cell research . In: Stern , July 20, 2006.
- ↑ Michael Minkenberg : Christian Right and American Politics from the First to the Second Bush Administration . In: Federal Center for Political Education. B 46/2003.
- ↑ http://www.answers.com/topic/stem-cell .
- ↑ California gives go-ahead to stem-cell research , MSNBC , Nov. 3, 2004.
- ↑ USA is researching stem cells again after eight years. on: orf.at , December 3, 2009: “Research with embryonic stem cells again in the USA”.
- ↑ US court blocks Obama's stem cell policy. on welt.de, August 24, 2010.
- ↑ Image of Science . Edition 2/2010.
- ^ Hans Clevers - 2016 Körber Prize Winner (video). Retrieved September 7, 2016 .
- ↑ Hans Clevers - 2016 Körber Prize Winner (press release). Retrieved November 29, 2018 .
- ↑ Toshiro Sato, Hans Clevers et al .: Single Lgr5 stem cells build cryptvillus structures in vitro without a mesenchymal niche . In: Nature . 459, No. 7244, 2009, pp. 262-265. bibcode : 2009Natur.459..262S . doi : 10.1038 / nature07935 . PMID 19329995 .
- ↑ Körber Prize for stem cell researchers: Replacement organs from the Petri dish (article in the ARD Tagesschau from 09/07/2016 1:31 p.m.). Retrieved September 8, 2016 .
- ↑ K. Takahashi, S. Yamanaka: Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors . In: Cell. 126, No. 4, August 25, 2006, pp. 663-676. doi: 10.1016 / j.cell.2006.07.024 . PMID 16904174 .
- ↑ Turning point in research: Artificial production of stem cells succeeded. In: Frankfurter Allgemeine Zeitung. November 20, 2007.
- ↑ Gretchen Vogel: Researchers Turn Skin Cells Into Stem Cells. In: ScienceNOW. November 20, 2007, sciencemag.org .
- ↑ Reprogrammed skin cells: The royal road to stem cells. In: Frankfurter Allgemeine Zeitung. June 7, 2007.
- ↑ M. Stadtfeld, M. Nagaya u. a .: Induced pluripotent stem cells generated without viral integration. In: Science. Volume 322, Number 5903, November 2008, pp. 945-949, ISSN 1095-9203 . doi: 10.1126 / science.1162494 . PMID 18818365 .
- ↑ M. Nakagawa, M. Koyanagi et al. a .: Generation of induced pluripotent stem cells without Myc from mouse and human fibroblasts. In: Nature biotechnology . Volume 26, Number 1, January 2008, pp. 101-106, ISSN 1546-1696 . doi: 10.1038 / nbt1374 . PMID 18059259 .
- ↑ JB Kim, V. Sebastiano u. a .: Oct4-induced pluripotency in adult neural stem cells. In: Cell. Volume 136, Number 3, February 2009, pp. 411-419, ISSN 1097-4172 . doi: 10.1016 / j.cell.2009.01.023 . PMID 19203577 .
- ↑ Gretchen Vogel: Reprogrammed Skin Cells Strut Their Stuff. In: ScienceNOW. December 6, 2007, sciencemag.org .
- ↑ Network Stem Cell Research NRW .
- ↑ Stem cell therapy for paraplegics. October 13, 2010.
- ↑ Murry, Soonpaa et al. a .: Haematopoietic stem cells do not transdifferentiate into cardiac myocytes in myocardial infarcts. In: Nature. 428, 2004, pp. 664-668.
- ↑ Vlad Georgescu: Stem cells: The unpredictable. In: DocCheck News. February 16, 2011, (online)
- ↑ Louise C. Laurent et al. a .: Dynamic Changes in the Copy Number of Pluripotency and Cell Proliferation Genes in Human ESCs and iPSCs during Reprogramming and Time in Culture. In: Cell Stem Cell. 8 2011, pp. 106–118, doi: 10.1016 / j.stem.2010.12.003 , online (PDF document; 1.81 MB).
- ↑ Masahito Tachibana et al. a .: Human Embryonic Stem Cells Derived by Somatic Cell Nuclear Transfer. In: Cell . Online publication from May 15, 2013, doi: 10.1016 / j.cell.2013.05.006 .
- ↑ ongoing-clinical-trials ( memorial from March 31, 2015 in the Internet Archive ). OCATA Therapeutics website. Retrieved March 28, 2015.
- ↑ Steven D. Schwartz et al. a .: Human embryonic stem cell-derived retinal pigment epithelium in patients with age-related macular degeneration and Stargardt's macular dystrophy: follow-up of two open-label phase 1/2 studies. In: The Lancet. Volume 385, No. 9967, 2015, pp. 509-516, doi: 10.1016 / S0140-6736 (14) 61376-3 .
- ↑ Suseela Keerti Popuri: Concerns of a Pediatric Dentist in Dental Stem Cells: An Overview . Ed .: The open dentistry journal. 2018, p. 596-604 .
- ↑ J. Jobst: Extraction of stem cells from milk teeth. In: Kigorosa. Roman Safreider, January 14, 2019, accessed on March 11, 2019 .
- ↑ G. Schmalz: On the way to the new pulp: can we regenerate the pulp? Ed .: Austrian Dental Congress 2012 and Symposium for Pediatric Dentistry. Volume 109.Springer Vienna, September 2012, p. 52-96 .