Properties of water
| Space-filling model of the water molecule | |
|---|---|

|
|
| General | |
| Surname | water |
| other names |
|
| Molecular formula | H 2 O |
| CAS number | 7732-18-5 |
| PubChem | 962 |
| Brief description | transparent and almost colorless liquid, also tasteless and odorless if pure substance |
| central material data | |
| Molar mass | 18.015268 g mol −1 |
| density | 3.98303 ° C: 0.999975 g · cm −3 20 ° C: 0.9982067 g · cm −3 |
| Melting point | 101.325 kPa: 0.002519 ° C |
| boiling point | 101.325 kPa: 99.974 ° C |
| Triple point | 0.01 ° C / 6.11655 hPa |
| critical point | 373.946 ° C / 22.064 MPa / 322 kg / m³ |
| Isentropic exponent (adiabatic exponent) | 1.3367 (100 ° C, 1 bar) 1.3180 (200 ° C, 1 bar) |
| Saturation vapor pressure | 31.6874 hPa (25 ° C) |
| specific heat capacity | 4.184 kJ / (kgK) (14.5 ° C) |
| Thermal conductivity | 0.597 W / (m K) (20 ° C) |
| Self diffusion coefficient | 2.299 · 10 −9 m² · s −1 (25 ° C) |
| Heat of evaporation | 2257 kJ / kg and 40.8 kJ / mol, respectively |
| Heat of fusion | 333.5 kJ / kg |
| Standard enthalpy of formation | −285.8 kJ / mol (l) −241.8 kJ / mol (g) |
| Refractive index | 1.33-1.35 (25 ° C, visible light) / 1.310 (ice) |
| Dynamic viscosity | 1.0 mPa s (20 ° C) |
| relative permittivity | 80.35 (20 ° C) |
| pH value (22 ° C) | 7.0 |
| solubility | Good in polar , bad in non-polar solvents |
| As far as possible and customary, SI units are used. Unless otherwise noted, the data given apply to standard conditions . | |
The properties of water have fundamental meanings for life on earth . These physical , chemical , electrical and optical properties are based on the structure of the water molecule and the resulting linkages and interactions between the water molecules via hydrogen bonds , electrical dipole forces and other forces such as Van der Waals forces .
In nature, water does not occur as a pure substance ; it practically always contains dissolved substances (mainly ions of salts ), even if in concentrations that can hardly be measured . Such dissolved substances change the properties of the water. Very pure water is produced in the laboratory by distillation and is called distilled water . Demineralized water is often used in technical applications .
Physical Properties
The molar mass of the mean water , calculated from the measurements of the atomic masses determined in 2012, is 18.015268 g / mol .
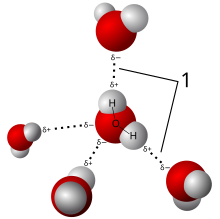
The properties of water are particularly determined by the three-dimensional chaining of the water molecules via hydrogen bonds , without which a substance with such a low molar mass as water would have completely different properties. This is especially true for the high melting and boiling point as well as for the density, which is highest under normal pressure at around 3.98 ° C at 0.999975 kg / dm 3 (see density and density anomaly ).
From 1901 to 1964 the unit liter was defined as the volume of 1 kg of water at the temperature of its highest density under normal pressure.
The physical properties of water are also heavily dependent on temperature and pressure . So take surface tension and viscosity with increasing temperature. The compressibility is also temperature dependent.
Physical states
As can be seen in the phase diagram , water is a liquid under normal conditions . It is the only known substance that exists on the earth's surface (see hydrosphere ) in significant quantities in all three classical states of aggregation . The phase diagram shows to what extent the physical state of the water depends on temperature and pressure. The critical point of the water is at 373.946 ° C and 2.2064 · 10 7 Pa (322 kg / m³), the triple point at 0.01 ° C and 611.657 ± 0.010 Pa.
For the properties and peculiarities of the gaseous and solid aggregate states of water, see the articles water vapor and ice . In the case of supercritical water , i.e. above the critical point, it is not possible to differentiate whether a substance is liquid or gaseous.
Melting and boiling point
Compared to chemically analogous compounds such as hydrogen sulfide (−61 ° C), hydrogen selenide (−41 ° C) and hydrogen telluride (−2 ° C), water has a relatively high boiling point. The increase in boiling points is due to the likewise increasing molar mass and the resulting higher amount of energy that has to be applied in order to convert the respective substance into the gaseous state of aggregation. Methane , for example, has a molar mass that is very similar to water and boils at −162 ° C under normal pressure. If you take the molar mass of water as the only characteristic, then it should boil at −80 ° C and would therefore be gaseous at room temperature. However, the boiling point is 100 ° C, which is 180 K higher.
The same picture emerges with the melting point; it is −86 ° C for hydrogen sulfide, −66 ° C for hydrogen selenide and −49 ° C for hydrogen telluride. For water it should be around −100 ° C according to the molar mass, but in fact it is 0 ° C. If one compares the area in which water occurs as a liquid, the result is a range of 20 K for the case that one considers the molar mass alone. In reality, however, this range is considerably larger at 100 K.
All these peculiarities result from the structure of the water molecule and its tendency to form linked clusters via hydrogen bonds as shown in the picture. These additional bonds, which do not occur with the other substances, have to be overcome and taken into account with each phase transition.
So under normal conditions water boils at 100 ° C and ice melts at 0 ° C. Accordingly, water solidifies at 0 ° C; however, it can still be present as a liquid under normal conditions below 0 ° C. It is then supercooled water . Theoretically, ultrapure water can remain liquid up to −48 ° C (see #New findings ). At pressures between 1000 and 2000 bar solidifies water below -138 ° C in the amorphous state . Conversely, ice can remain solid for a short time above 0 ° C as long as this temperature is not reached on the surface. The boiling point of water is strongly dependent on the saturation vapor pressure . The boiling temperature decreases as the triple point is approached together with the boiling pressure and both reach their minimum at this point. In addition, water can also be heated slightly above its boiling point, which is known as delayed boiling .
Substances dissolved in water also change the boiling and melting points. For example, water has a molar decrease in melting point of 1.853 K kg / mol and a molar increase in boiling point of 0.513 K kg / mol.
In the past, the Celsius temperature scale was defined by the melting and boiling points of water. Due to the current definition of the Celsius scale using the Kelvin scale, the melting and boiling points of water are no longer exactly 0 ° C and 100 ° C, but are at 0.002519 ° C and 99.9839 ° C (99.9743 ° C C according to ITS-90).
A special feature of the solidification of water is the Mpemba effect , named after its discoverer , according to which hot water freezes faster than cold water under special conditions.
Heating water
If water is heated in a saucepan on a stove, the water on the bottom heats up faster than that on the surface. This creates an unstable temperature gradient which, however, soon largely disappears due to convection . When the water on the bottom reaches boiling temperature, water vapor bubbles form there. When they rise, they cool down again and collapse. They produce the typical crackling noise that can be heard shortly before the boil. With further heat supply, only the small bubbles collapse, the large ones rise. The boiling noise becomes quieter and disappears completely when the water is completely boiled.

The vapor bubbles do not rise in the water under weightlessness . Instead, they stay near the bottom of the pot and conglomerate into larger bubbles and eventually into a single large bubble. The lack of convection and the reduced heat conduction through the steam bubbles make it difficult to boil water quickly in a spaceship.
Sublimation and resublimation
In the temperature range from about 0 K to 273.16 K (−273.15 ° C to +0.01 ° C) and a pressure range from high vacuum to about 0.006 bar, in the range below the triple point, water does not exist in liquid form, but only gaseous and solid. In this area, i.e. at the point of sublimation , ice changes directly into the gaseous state without any change in the state of aggregation into a liquid . This process is known as sublimation or, in the opposite direction, as resublimation . In a vacuum, sublimation takes place down to almost 0 Kelvin (−273.15 ° C). The upper limit, however, is given by the triple point.
Specific heat capacity
Liquid water has a very high specific heat capacity of around 4.2 kJ / (kg · K) (under normal pressure in the temperature range from zero to one hundred degrees Celsius between 4.219 and 4.178 kJ / (kg · K)). So you need 4.2 kilojoules of thermal energy to heat one kilogram by one Kelvin . This means that water can absorb quite a lot of energy compared to other liquids and solids. The comparatively high specific heat capacity of water is used, for example, in heat storage systems for heating systems.
If you heat 1 kg of water from 15 ° C to 100 ° C, then you need 4.2 kJ / (kg · K) · 85 K · 1 kg = 357 kJ. One kilowatt hour (kWh) is 3.6 MJ. To heat one liter of water from pipe temperature to 100 ° C under normal pressure, you need about 0.1 kWh of energy. To allow the water to evaporate, six times the amount of energy is also required ( see below ).
Water vapor (at 100 ° C) has a specific heat capacity of 1.870 kJ / (kg · K) and ice (at 0 ° C) 2.060 kJ / (kg · K). Solid substances usually have a significantly lower specific heat capacity. For example, lead has a heat capacity of 0.129 kJ / (kg · K), copper one of 0.380 kJ / (kg · K).
Heat of fusion and evaporation
For thawing, i.e. the conversion of 0 ° C cold ice into 0 ° C cold water, an energy of 333.5 kJ / kg must be applied. With the same amount of energy, the same amount of water can be heated from 0 ° C to 80 ° C. To convert 100 ° C warm water into 100 ° C warm steam , 2257 kJ / kg are required. To convert 0 ° C cold water into 100 ° C warm steam, you need 100 K · 4.19 kJ / (kg · K) + 2257 kJ / kg = 2676 kJ / kg.
The specific heat of vaporization of water is much higher than that of other liquids. Methanol has a heat of vaporization of only 845 kJ / kg and mercury even of only 285 kJ / kg. However, if one compares the molar evaporation heats, then mercury with 57.2 kJ / mol has a higher value than water with 40.6 kJ / mol.
In meteorology, the heat of fusion and evaporation are of great importance in the context of latent heat .
Thermal conductivity
Compared to other liquids, water has a high thermal conductivity , but very low compared to metals. The thermal conductivity of liquid water increases with increasing temperature, but ice conducts heat much better than liquid water.
At 20 ° C, water has a thermal conductivity of 0.60 W / (m · K). For comparison: copper 394 W / (m · K) and silver 429 W / (m · K). Even the worst heat conductor among all metals, bismuth comes to 7.87 W / (m · K).
The thermal conductivity of water in the form of ice at −20 ° C is at least 2.33 W / (m · K).
Density and density anomaly
Water has a density of around one kilogram per liter (one liter corresponds to one cubic decimeter ). This round relationship is no coincidence: it goes back to the unit Grave , which is one of the historical roots of today's international system of units (SI) . A grave was defined as the mass of one liter of water at 4 ° C.
At normal pressure, water has its greatest density at 3.98 ° C and thus shows a density anomaly . This consists in the fact that water below 3.98 ° C expands again when the temperature drops further, even when it changes to the solid state of aggregation, which is only known from a few substances.
In addition to the temperature, substances dissolved in the water also influence its density, which can be measured with a hydrometer . Since the dissolved particles are distributed between the water molecules and the increase in volume is small, the density increases as a result. The increase in density roughly corresponds to the mass of dissolved substance per volume and plays an important role in large-scale water movements, for example in the context of thermohaline circulation or the dynamics of freshwater lenses .
Smell and taste
In its pure state, water is tasteless and odorless .
Optical properties
Refraction and reflective properties
In the visible light range, water has an index of refraction of approximately 1.33. If light hits the interface between air (refractive index ≈ 1) and water, it is therefore refracted towards the perpendicular . The refractive index is lower in comparison to many other materials, so the refraction through water is less pronounced than, for example, when air passes into most types of glass or even into diamond . But there are also materials like methanol that have a lower refractive index. The refraction of light leads to optical illusions , so that one sees an object underwater in a different place than where it actually is. The same applies to a view from the water into the airspace. Animals that specialize in fishing, such as herons, or fish hunting for insects over the water can take this image shift into account and therefore usually hit their prey without any problems.
According to Fresnel's formulas, the reflectivity of the surface air-water is about 2% at normal incidence. As with all materials, this value increases with a flatter angle of incidence and is approximately 100% with grazing incidence. The reflection behavior, however, depends on the polarization of the light. Parallel polarized light generally has a lower degree of reflection than perpendicularly polarized light, which means that light is polarized when it hits the interface between air and water. Due to the relatively low refractive index of water, this effect is less pronounced than with many other (transparent) materials with a higher refractive index. The polarization effect is generally stronger the flatter the light hits the water surface. This is used, for example, in photography, where a polarization filter is used to filter out a certain polarization, which can reduce interfering reflections.
If light from the water hits the water-air interface, the direct consequence of the law of refraction is total reflection from a critical angle of 49 ° . This means that light rays hitting the boundary surface more flatly do not exit the water, but are reflected.
Some optical effects in the atmosphere are linked to the refractive properties of water. For example, a rainbow is caused by water droplets or a halo phenomenon is caused by ice crystals, in which the light is refracted and split into spectral colors. The darkening of the earth by clouds is also based on the refraction of light and total reflections in or on water droplets.
Absorption behavior and color
Water absorbs light in the visible spectral range only very weakly, that is, the imaginary part of the complex refractive index ( extinction coefficient ) is approximately 0. Water is therefore generally regarded as transparent and colorless. The high light permeability of the water enables the existence of algae and plants in the water that need light to live.
However, the low extinction coefficient in the visible spectral range also shows changes of several orders of magnitude (see figure). The extinction coefficient and therefore also the absorption (cf. Lambert-Beer law ) is lowest in the wavelength range of approx. 400–440 nm . This means that light of these wavelengths is only completely absorbed after several meters. The extinction coefficient increases slightly in the red visible and near infrared range. Long-wave (red) light is therefore more strongly absorbed than short-wave (blue) light. This gives water a faint bluish color. However, this is only noticeable with the naked eye in thick layers from a few meters. UV light is only absorbed to a significant extent at a wavelength below 240 nm.
Another decisive factor that influences the optical properties of water are substances dissolved in the water and particles floating in the water. Substances dissolved in water can lead to a significant change in these properties, which is described by the spectral absorption coefficient . Small particles with a diameter in the range of the wavelength, however, lead to the scattering of the light, the water then appears slightly cloudy (or colloquially milky ). The color and turbidity of the water, depending on the substances it contains, play an important role as indicators of water quality and also as an examination method in water analysis.
Electromagnetic excitation and resonance
The lowest resonance frequency of the free water molecule is 22.23508 GHz. The integer multiples of this frequency in turn result in resonance.
On the other hand, only around a ninth of this is the usual frequency of 2.455 GHz of the microwave oven - in the household as well as in the chemistry laboratory. Only this lower frequency (in an ISM band ) allows several centimeters of penetration into the water-containing medium and thus the heating "from the inside".
Specific resistance and electrical conductivity
Chemically pure water is only slightly dissociated into the electrical charge carriers H 3 O + and OH - at a pH value of 7 . It therefore has a high specific resistance of 18.2 MΩ · cm (= 1.82 · 10 11 Ω · mm² / m) at 25 ° C. This corresponds to a specific conductance of 54.9 nS · cm −1 . This conductance increases with the temperature by approximately 1.5 to 2% per Kelvin . Dissolved salts and acids increase the charge carrier concentration. Depending on its mineral content, tap water already reaches up to 10,000 times the conductivity of an average of 500 µS · cm −1 , while sea water reaches values of 50 mS · cm −1 .
The Grotthuss mechanism increases the conductivity of protons and hydroxide ions in water, which are caused by the autodissociation of water.
viscosity
The dynamic viscosity (tenacity) of the water at 20 ° C is 1.0 mPa s. It has a higher viscosity than petroleum (0.65 mPa s at 20 ° C), but also a lower viscosity than , for example, mercury (1.5 mPa · s at 20 ° C). The viscosity of the water decreases due to the decreasing number of hydrogen bonds with increasing temperature and reaches 0.283 mPa s at the boiling point.
The viscosity is changed by dissolved substances. In addition to the concentration, the type of dissolved substance is decisive for the viscosity of the solution.
diffusion
The diffusion of water molecules within water or an aqueous solution is called self-diffusion and is described by the self-diffusion coefficient D; at 25 ° C this is D = 2.299 · 10 −9 m 2 · s −1 . The quantity D describes the translational mobility of the water molecules within the liquid water. In Newtonian liquids, this mobility is qualitatively linked to the viscous behavior, i.e. the viscosity of the water, which decreases with increasing temperature, is associated with an increasing self-diffusion coefficient.
The temperature dependence of the self-diffusion coefficient is measured very precisely and is often used as a set of reference values when studying diffusion in other liquids.
In addition to translational diffusion, there is rotational diffusion in water - as in other liquids too - namely the random change in the orientation of the symmetry axes of the water molecules through random movements ( random walk ) within the liquid. The correlation time characterizing this reorientation movement , i. H. Approximately the time during which a water molecule in the liquid has rotated once around itself by random small steps is at 25 ° C in the range of a few picoseconds , as measured by means of nuclear magnetic relaxation and dielectric relaxation . So it is about extremely fast, random reorientations of the water molecules and thus also about just as fast changes in the microstructure of the water.
When water of a different isotopic composition is present, e.g. B. heavy water D 2 O, then a so-called dynamic isotope effect occurs, which affects both the translational and the rotational diffusion. Since the relative change in weight due to isotope substitution is comparatively large with the low molar mass of water, the greatest isotope effects occur with water compared to other known liquids. At 25 ° CD 2 O has a 23% lower diffusion coefficient than H 2 O.
Dissolved substances, e.g. B. salts, can lower the self-diffusion coefficient and the rotational diffusion of the water ("structure-forming" salts with small ionic radii, such as lithium chloride ) as well as increase ("structure-breaking" salts with large ionic radii, such as cesium iodide ). Structure breaking salts, in which the anion causes the water structure breaking, are also often chaotropic salts. If non-polar or electrically uncharged species are dissolved in water, a hydrophobic effect occurs which, in addition to the rotation, also slows down the diffusion movement of the water molecules in the vicinity of these “ hydrophobic ” species and thus reduces the mean diffusion coefficient of the water in the solution.
Surface tension and wettability
Water has a comparatively high surface tension because the water molecules attract each other relatively strongly. The surface tension is about 73 mN / m at 20 ° C and decreases with increasing temperature. Because of the large surface tension, for example, can Sandpipers move on the water. Surface tension is a hindrance during washing processes, which is why detergents contain surface-active substances ( tensides ) that lower the surface tension. However, their occurrence in natural waters is low.
With a smooth surface, contact angles of a maximum of 120 ° can be achieved. In the case of roughened surfaces with a hydrophobic character, however, this angle can also be up to 160 °, which is referred to as superhydrophobicity. Many plants take advantage of this via the lotus effect.
Compression modulus and speed of sound
At a temperature of 4 ° C under normal pressure, water has a compression modulus of around 2.06 GPa - at 100 MPa (a thousand times normal pressure or water pressure at a depth of almost 10 km) it is therefore compressed by around five percent. Corresponding to the density of 1 kg / dm 3 , this results in a speed of propagation of sound in water of 1435 m / s.
Isotope fractionation
Water molecules can consist of different isotopes of hydrogen (e.g. Protium 1 H or Deuterium 2 H) and oxygen (e.g. 16 O or 17 O), each of which occurs in different concentrations. There are nine different stable configurations of the water molecule. So-called isotope effects occur. In certain processes such as the formation of precipitation and its phase transitions , isotope fractionation occurs , which means that the water changes its isotopic composition. Depending on the environmental conditions and the original composition, this results in specific isotope signals that can act as a kind of fingerprint for different processes and regions of origin. The corresponding methodology is mainly used in hydrogeology and paleoclimatology .
Water as a solvent
Because of its dipole, water is a good polar solvent for most substances. In general, the water solubility increases with the polarity of the substance. Water has a comparatively high dielectric constant of 80.35 (at 20 ° C).
The solubility in water is often strongly dependent on the temperature. Solids and gases behave differently. Gases dissolve in water proportionally to the partial pressure of the gas without a fixed limit on the amount that can be dissolved ( Henry's law ). The equilibrium concentration per pressure unit, referred to as “solubility”, decreases with increasing temperature. In contrast, solids usually dissolve better in water with increasing temperature, but there are also many exceptions, such as lithium sulfate .
Some substances such as acetone or ethanol can be mixed with water in any ratio, i.e. they are soluble in one another. In other cases there are reciprocal solutions with a miscibility gap , for example with phenol or chloroform.
Normally, the more polar groups there are, the better a molecular substance will dissolve in water. However, supercritical water shows similar solubility properties as non-polar organic solvents.
When ionic substances dissolve in water, the endothermic lattice breakdown and exothermic hydration take place, which enables heat mixtures (sulfuric acid in water) and cold mixtures (salts in water). The difference between the exothermic hydration and the endothermic degradation of the lattice determines whether heating or cooling occurs. In the case of salts, the ratio between the lattice energy and the hydration energy of the ions involved determines the solubility, which is defined here as the product of the molar ion concentrations at equilibrium with the crystalline substance ( solubility product ). As a rule of thumb for the solubility of ionic compounds, the following can apply: the higher the charge number of the ions involved , the less soluble the substance is in water.
In contrast to simple compounds such as sodium chloride, the ionic bonds of complexes are not broken. A distinction is made between two groups. On the one hand there are the strong complexes, such as the cyanide complexes of heavy metals, and on the other hand the weak complexes ( aqua complexes ) of the metal ions with sulfate, hydroxy or carbonate ions. The type and occurrence of the various metal species are important issues in chemical water analysis and water treatment .
In the case of molecules with different polarity, as in the case of many amphiphilic lipids, the water solubility or water affinity depends on its orientation. Almost all living things make use of this effect with their biomembranes . In this context, one also speaks of hydrophilicity or hydrophobicity .
Chemical properties
Water has a molar mass of 18.01528 g · mol −1 . In many reactions, water is a catalyst , that is, without the presence of water, a reaction would proceed much more slowly and only with a higher activation barrier. Many reactions are even enabled or accelerated by normal air humidity. This is practically not noticeable due to the actually always present traces of moisture in our environment, since it is the normal case on earth. This can only be proven when even the smallest residues of moisture are removed by special drying processes and the chemical tests are carried out in closed systems. In this environment, for example, carbon monoxide does not burn in oxygen and alkali metals do not react with sulfuric acid and chlorine.
Reactivity
Water is amphoteric , so it is a substance that - depending on the environment - can act as both an acid and a base .
Water reacts with anhydrides to form acids or bases . Examples:
- Phosphorus pentoxide (acid anhydride) reacts with water to form phosphoric acid (acid):
- Sodium oxide (base anhydride) reacts with water to form sodium hydroxide (base):
Water reacts with base metals under hydrogen formation to metal oxides, but these metal oxides are Basenanhydride and dissolve mostly right back into water to bases, as just described. An example:
- Magnesium reacts with water vapor to form magnesium oxide and hydrogen :
Leveling effect
In aqueous solutions, strong acids and strong bases dissociate completely to H 3 O + or OH - ions. So the different acid strengths of z. B. No longer differentiate hydrogen chloride and perchloric acid in water on the basis of the pH value. Here one speaks of the leveling effect (from French : niveler = equalize) of the water. In order to be able to distinguish even very strong acids with regard to acid strength, equilibrium constants are determined in non-aqueous solutions and these are approximately transferred to the solvent water.
PH value
Chemically pure water at 22 ° C has a theoretical pH value of 7. The equilibrium constant for the dissociation of water is then exactly 10 −14 . This value is defined as chemically neutral. This dissociation means that a proportion of the water of 10 −7 is separated into the ions H + and OH - . These in turn are hydrated and only remain in this state for a short time, then other water molecules dissociate. As a result of the movement of heat , these ions rarely find their previous partner again when they reunite, but take any other partner. This leads z. For example, when equal amounts of H 2 O and D 2 O are poured together, HDO is predominantly formed after a short time.
Chemically pure water does not have a buffer effect and therefore reacts to the smallest amounts of acidic or basic substances with a significant change in the pH value. In previously chemically pure water, a pH value between 4.5 and 5 immediately sets in when air is exposed as a result of the solution of CO 2 . On the other hand, water reacts with dissolved salts (e.g. hydrogen carbonates) much less sensitive to the addition of acidic or basic substances.
Ion product
The ion product of the water is the product of the concentrations of the H 3 O + and OH - ions in the water. In 1894 Friedrich Wilhelm Georg Kohlrausch and Ernst Heydweiller investigated the conductivity of distilled water by distilling water in the absence of air (see dissociation ). From these measurements and from knowledge of the equivalent conductivities of hydronium ions and hydroxide ions, the ion product of water could be calculated.
When measuring the conductivity of distilled water, a small amount of current flows. This is an indication of ions in the water, which can only have arisen through the autoprotolysis of the water, according to the following reaction:
The law of mass action can be applied to the protolysis equilibrium:
Since the concentration of the water molecules remains almost constant even if the equilibrium is shifted (55.5 mol / l), the value can be included in the constant.
and combine both into a new constant, the K w value, which is the product of the respective concentrations of the oxonium and hydroxide ions:
At 22 ° C, K w = 10 −14 ( mol / l ) ². So the equilibrium is very much on the water side. The concentrations of H 3 O + and OH - ions are each 10 −7 mol / L. So the pH is 7.
If the concentration of one of the two ions is increased, the ion product of 10 −14 is retained, i.e. that is, the concentration of the other ion decreases. The sum of pH and pOH must therefore always be 14.
The pK W of the water changes depending on the temperature.
| T in ° C | 0 | 20th | 22nd | 40 | 80 |
|---|---|---|---|---|---|
| pK W | 14.9 | 14.2 | 14.0 | 13.5 | 12.6 |
| c (H 3 O + ) in mol / l | 10 −7.45 | 10 −7.1 | 10 −7 | 10 −6.75 | 10 −6.3 |
(values determined experimentally by conductivity measurement)
With knowledge of the ionic product of water, the pH values of dissolved salts, acids, bases in water (e.g. sodium acetate, sodium carbonate, calcium oxide, hydrochloric acid, sulfuric acid, caustic soda) can be calculated.
Reaction order of the autoprotolysis of water
If the autoprotolysis of water is considered in the following form:
For the forward reaction, i.e. the dissociation, formally a reaction of 0th order results. For the reverse reaction, a second order reaction follows formally.
Water hardness
Water hardness describes the equivalent concentration of the ions of the alkaline earth metals dissolved in the water . The "hardness builders" essentially include calcium and magnesium ions and traces of strontium and barium ions . These cations have a great, positive physiological importance , but interfere with some uses of the water.
New insights
In 2011, the Americans Emily B. Moore and Valeria Molinero from the University of Utah determined in complex computer simulations that pure water - i.e. H 2 O without the presence of any crystallization nuclei - only freezes at −48.3 ° C. It does this through crystallization in a tetrahedral shape; in the center of the crystal is a water molecule, which is surrounded by four other molecules. At the temperature mentioned above, there are only these crystals and no more free water molecules.
literature
- Klaus Scheffler: Steam panels: thermodynam. Properties of water and steam up to 800 ° C u. 800 bar. Berlin 1981, ISBN 3-540-10930-7 .
- Leopold Lukschanderl: Water: the substance that looks ordinary, but has very unusual properties. Vienna 1991, ISBN 3-85128-062-8 .
- LA Guildner, DP Johnson, FE Jones: Vapor pressure of Water at Its Triple Point: Highly Accurate Value. In: Journal of Research of the National Bureau of Standard - A. Vol. 80A, No. 3, 1976, pp. 505-521; doi: 10.1126 / science.191.4233.1261 ; PDF .
- Felix Franks (Ed.): Water, a Comprehensive Treatise Vol.I - Vol.VIII, Plenum Press, New York London, 1972–1982.
- Philip Ball: H 2 O - Biography of Water Piper, 2001, ISBN 3-492-04156-6 .
Web links
- hydroskript.de: The physical properties of water. ( Memento from August 30, 2016 in the Internet Archive )
- Martin Chaplin: Water Structure and Science .
Individual evidence
- ^ R. Panico, WH Powell, J.-C. Richer (Eds.): A Guide to IUPAC Nomenclature of Organic Compounds . Ed .: IUPAC Commission on the Nomenclature of Organic Chemistry. Blackwell Scientific Publications, Oxford 1993, ISBN 0-632-03488-2 , pp. 37 .
- ↑ a b Lechner, Lühr, Zahnke (ed.): Pocket book of water management. Gabler Wissenschaftsverlage, 2001, ISBN 3-8263-8493-8 , p. 5 ( limited preview in the Google book search).
- ↑ a b Guideline on the Use of Fundamental Physical Constants and Basic Constants of Water. (PDF) IAPWS G5-01 (2016). International Association for the Properties of Water and Steam, September 2016, accessed on August 15, 2017 (English): "The relative molar mass used in calculations (with the exception, mentioned above, of cases in which the isotopic composition is known to be different) should be that of VSMOW. This is computed by combining the isotopic composition of VSMOW with the accepted values of the masses of each isotope. Performing this calculation with the 2012 atomic mass evaluation yields a relative molar mass of 18.015 268, with an uncertainty of no greater than two in the last digit. "
- ↑ a b c M Tanaka, G Girard, R Davis, A Peuto, N Bignell: Recommended table for the density of water between 0 ° C and 40 ° C based on recent experimental reports . In: Metrologia . 38, No. 4, 2001, pp. 301-309. doi : 10.1088 / 0026-1394 / 38/4/3 .
- ^ Rainer Feistel, Wolfgang Wagner: A New Equation of State for H 2 O Ice Ih . In: Journal of Physical and Chemical Reference Data . 35, No. 2, 2006, pp. 1021-1047. doi : 10.1063 / 1.2183324 .
- ^ A b c W. Wagner, A. Pruss: The IAPWS Formulation 1995 for the Thermodynamic Properties of Ordinary Water Substance for General and Scientific Use . In: Journal of Physical and Chemical Reference Data . 31, No. 2, 2002, pp. 387-535. doi : 10.1063 / 1.1461829 .
- ↑ Isobaric Properties for Water , on webbook.nist.gov
- ↑ CRC Handbook, pp. 5–13 ( Memento from April 26, 2015 in the Internet Archive ).
- ^ G. Audi, M. Wang, AH Wapstra, FG Kondev, M. MacCormick, X. Xu, B. Pfeiffer: The Ame2012 atomic mass evaluation . In: Chinese Physics C . 36, No. 12, 2012, pp. 1287-1602. doi : 10.1088 / 1674-1137 / 36/12/002 .
- ↑ Maurice L. Huggins: 50 Years Theory of Hydrogen Bonding . In: Angewandte Chemie . 83, No. 5, 1971, pp. 163-168. doi : 10.1002 / anie.19710830503 .
- ↑ International Office for Weights and Measures (Ed.): Declaration concernant la définition du liter (= Comptes rendus des séances de la… Conference Générale des Poids et Mesures / Bureau International des Poids et Mesures . Volume 3 ). Gauthier-Villars, Paris October 16, 1901, DNB 010970436 , LCCN sv91-007003 , OCLC 476955218 , Deuxième séance, p. 38 f . (French, 104 p., bipm.org [PDF; 12.1 MB ; accessed on August 15, 2017]).
- ^ Revised Release on Surface Tension of Ordinary Water Substance. (PDF) International Association for the Properties of Water and Steam, June 2014, p. 6 , accessed on August 16, 2017 .
- ^ Release on the IAPWS Formulation 2008 for the Viscosity of Ordinary Water Substance. (PDF) International Association for the Properties of Water and Steam, September 2008, p. 9 , accessed on August 16, 2017 (English).
- ^ Rana A. Fine, Frank J. Millero: Compressibility of water as a function of temperature and pressure . In: The Journal of Chemical Physics . 59, No. 10, 1973, pp. 5529-5536. doi : 10.1063 / 1.1679903 .
- ↑ a b Jan Oliver Löfken: The true freezing point of water - minus 48 degrees Celsius. In: pro-physik.de. November 24, 2011, accessed October 21, 2019 .
- ^ PH Handle, M. Seidl, T. Loerting: Relaxation Time of High-Density Amorphous Ice . In: Physical Review Letters . tape 108 , no. 22 , 2012, p. 225901–225904 , doi : 10.1103 / PhysRevLett.108.225901 .
- ↑ Hot ice. In: pro-physik.de. January 13, 2006, accessed May 3, 2016 .
- ↑ H. Iglev, M. Schmeisser, K. Simeonidis, A. Thaller and A. Laubereau: Ultrafast superheating and melting of bulk ice. In: Nature . 439, January 12, 2006, pp. 183-186, doi: 10.1038 / nature04415 .
- ↑ Martin Chaplin: Water Structure and Science. Water Properties (including isotopologues). In: London South Bank University . February 26, 2016, accessed May 3, 2016 .
- ↑ Isobaric Properties for Water , on webbook.nist.gov
- ↑ The photolysis of water protected the primordial soup and early life forms . Prof. Blum's educational server for chemistry, March 14, 2001.
- ↑ a b M. Holz , SR Heil, A. Sacco: Temperature-dependent self-diffusion coefficients of water and six selected molecular liquids for calibration in accurate 1H NMR PFG Measurements. In: Phys. Chem. Chem. Phys. 2, 2000, pp. 4740-4742; doi: 10.1039 / b005319h .
- ↑ HGHertz: Micro Dynamic behavior of liquids as of studied by NMR relaxation times. In: Progress in Nuclear Magnetic Resonance Spectroscopy. Vol. 3, Pergamon Press, 1967, p. 159.
- ^ Felix Franks: Water a matrix of life. Second Edition, RSC Paperbacks, Cambridge 2000, ISBN 0-85404-583-X , p. 28.
- ↑ Edme H. Hardy, Astrid Zygar, Manfred D. Zeidler, Manfred Holz, Frank D. Sacher: Isotope effect on the translational and rotational motion in liquid water and ammonia. In: J. Chem Phys. 114, 2001, pp. 3174-3181; doi: 10.1063 / 1.1340584 .
- ^ KJ Müller and HG Hertz : A-Parameter as an Indicator for Water-Water Association in Solutions of Strong Electrolytes. In: J. Phys. Chem. 100, 1996, pp. 1256-1265; doi: 10.1021 / jp951303w .
- ^ Roberto Fernandez-Prini, AH Harvey, DA Palmer: Aqueous Systems at Elevated Temperatures and Pressures Physical Chemistry in Water, Steam and Hydrothermal Solutions . Academic Press, 2004, ISBN 978-0-08-047199-0 , pp. 290 ( limited preview in Google Book Search).
- ^ Bernward Hölting, Wilhelm G. Coldewey: Hydrogeology Introduction to General and Applied Hydrogeology . Springer-Verlag, 2013, ISBN 978-3-8274-2354-2 , pp. 114 ( limited preview in Google Book search).
- ↑ a b Because of zero degrees. On: Wissenschaft.de from November 23, 2011.
- ^ Emily B. Moore, Valeria Molinero: Structural transformation in supercooled water controls the crystallization rate of ice . In: Nature . tape 479 , no. 7374 , November 23, 2011, p. 506–508 , doi : 10.1038 / nature10586 .