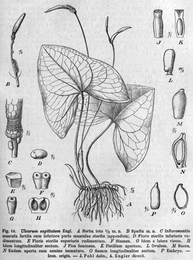
Arum family
| Arum family | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|

Inflorescences of Xanthosoma roseum |
||||||||||||
| Systematics | ||||||||||||
|
||||||||||||
| Scientific name | ||||||||||||
| Araceae | ||||||||||||
| Yuss. |
The arum family (Araceae) are a family of plants within the monocotyledons (monocotyledons). The family includes 115 to 129 genera with 3270, 3305 or 4025 species depending on the definition . They are distributed almost all over the world, but most species are found in the tropics .
description



The characteristics of the Lemnoideae often differ greatly from the other Araceae.
Illustration of different species of the subfamily Lemna (Lemnoideae)
Tribus Zomicarpeae: illustration of Ulearum sagittatum
Tribus Caladieae: Illustration by Hapaline brownii
Appearance and leaves
The species of the arum family are evergreen or seasonal green, mostly herbaceous plants , and can be from tiny (Lemnoideae) to meters high. There is a broad spectrum of life forms: in addition to many terrestrial species there are epiphytes , lithophytes or climbing plants , many species are geophytes ; the Lemnoideae are plants floating on or under the surface of the water. Some species of the Lemnoideae lack roots. Many species form aerial roots or rhizomes or tubers as underground perennial organs . Sometimes bulbils are formed , for example on the leaves, for vegetative reproduction. Many species form above-ground, independently upright, climbing to creeping stems , which are often green. The stems of some taxa are reinforced ( e.g. Montrichardia ). Often there are prophylls and / or cataphylls under every leaf .
The alternate, only basal or distributed on the stem have leaves in most species with a petiole with a leaf sheath at the base. The very differently shaped leaf blades can be undivided or divided in different ways. Often the base of the spade is heart-shaped or arrow-shaped; sometimes the leaf is shield-shaped (peltat).
Inflorescences and flowers
The membranous prophylls and / or cataphylls are below the inflorescences. Often there is a leafless inflorescence stem. Most Araceae have an inflorescence typical of this family with an unbranched piston, called a spadix , which usually contains many flowers, and a single bract , called a spathe , which more or less surrounds the cob. The spathe usually has a tubular base, the edges of which can be fused and have a durable to more or less rapidly perishable bract blade.
The flowers are often small and inconspicuous. The flowers of the original taxa are hermaphroditic. Often the flowers are unisexual, sometimes the flower parts are then greatly reduced. If the flowers are unisexual, then the species are mostly monoecious ( monoecious ), rarely dioecious ( dioecious ) separately sexed. In single-sex species there is a sterile section at the base of the spadix, the female flowers in the lower area and the male in the upper area; sometimes sterile flowers are also found.
The flowers are two or three-digit. In hermaphrodite flowers there are six, four or no mostly free bracts . Usually four to six, rarely up to 22 stamens and one ovary are present. Your stamens are free and the dust bags have two counters. Three fruit leaves are a mostly dreikammerigen or sometimes unilocular ovary grown.
Single-sex flowers are almost always missing bracts, only three African genera, including Zamioculcas , have them. Male flowers usually have two to four (one to six) free stamens or two to twelve (rarely up to 32) stamens that have grown together to form a "synandrium". They open at their tips with pores or straight to horseshoe-shaped slits. In most genera, the pollen grains have no apertures and no sporopollenin ; the exine has very different patterns. Staminodes are sometimes present in the female flowers . The female flowers fruit leaves are single-chamber to a mostly, sometimes three to vierkammerigen, mostly upper constant ovary grown. There are one to many ovules in each ovary chamber . The stylus, which is usually barely recognizable, ends in a scar that is moist during anthesis and is sometimes clearly lobed.
In the Lemnoideae, the inflorescence (which is rarely found) is reduced to one or two flowers and the unisexual flowers contain only one or two stamens or only one carpel with one to seven ovules.
Infructescence, fruits and seeds
They form one to many-seeded berries . The berries are usually found individually in the fruit stands or they rarely form a collective crop in the Cryptocoryne or Syngonium . The berries usually stay closed or in the Monstereae without Amydrium they burst. The berries stay green or when ripe they usually turn red, white or yellow, rarely blue. The berries are sometimes juicy. Endosperm is abundant or absent.

Flower ecological characteristics using examples of individual species
In spring you can spot the arrow leaves of the spotted arum ( Arum maculatum ) in the deciduous forest. The brownish bulb (spadix) gives off a musty, urine-like odor that some insects find irresistible. Trap hairs on the constriction in the lower third prevent the insects from escaping prematurely. Only after sufficient pollen has passed from the male to the female arum flowers does the prison open again. The lizard root ( Typhonium venosum ) has a similar pollination mechanism .
Other arum plants such as the genera Flamingo flowers ( Anthurium ), Spathiphyllum or Zantedeschia seem to be particularly attractive to male bees due to the color of their bracts and their scent.
Among the Philodendron the plant with the "largest inflorescence", actually the "largest one flower " (the largest inflorescence is that of Puya raimondii ) in the world, consisting of Sumatra originating Titan Arum ( Amorphophallus titanum ). This inflorescence smells unpleasantly of carrion, to which carrion beetles are strongly attracted.
As a special feature, the Araceae with their alternative respiratory chain have developed a way to produce heat in the spadix. The Arum maculatum species, for example, uses this heat development to better evaporate the scents of flowers (carrion odor) and thus attract insects . Other species such as Symplocarpus foetidus, on the other hand, protect their inflorescences from frost and cold damage through the heat generated in the mitochondria of the central flower stalk: the plant is able to keep its internal temperature constant at 20 ° C even at ambient temperatures below freezing point .



























Systematics and distribution
Botanical history and taxonomy
The Araceae family was established in Familles des Plantes in 1789 by Antoine Laurent de Jussieu . Heinrich Wilhelm Schott published in Genera Aroidearum in 1858 and in Prodromus Systematis Aroidearum in 1860 the first classification of the Araceae with a narrow genus concept based on flower characteristics. A clearly different classification of the Araceae was created by Adolf Engler from 1876 with revisions up to 1920 based on vegetative and anatomical features. These two systems competed with one another until the last decade of the 20th century molecular biological studies provided additional data.
Synonyms for Araceae Juss. are Arisaraceae Raf. , Caladiaceae Salisb. , Callaceae Rchb. ex Bartl. , Cryptocorynaceae J. Agardh , Dracontiaceae Salisb. , Lemnaceae Martinov , Orontiaceae Bartl. , Pistiaceae Rich. ex C. Agardh, Pothaceae and Wolffiaceae Bub.
Overview of the distribution
The Araceae family is found almost worldwide. Most species thrive in the tropics . Of the 3305 species accepted in the Araceae family in 2012, around 1889 are distributed in the Neotropic , around 1105 species occur in Asia , 152 species are found in Africa , 23 species are native to Australia , 78 species are distributed in the Mediterranean region, 20 species occur only on the Mascarene Mountains and in North America there are only 7 species. Only 2 species thrive in the boreal zone and 29 species have a circumtropical distribution. In Central Europe only the genera Arum ( Arum ), Dragon Arum ( Calla ), Dwarf Duckweed ( Wolffia ), Pond Lentils ( Spirodela ) and Duckweed ( Lemna ) occur naturally; the threefold pinellia ( Pinellia ternata ) has overgrown from botanical gardens.
External system
Traditionally, the Araceae family was placed in its own Arales order within the monocot family. Molecular biological findings (gene sequence analyzes) show that the Araceae family belongs to the order of the alismatales .
Internal system
The Araceae family includes around 115 genera with around 4025 species . The relationships and biogeography of the Araceae are now relatively well investigated using molecular sequence data. Of these, the duckweed family (Lemnaceae) were treated as an independent family until recently and are now assigned to the Araceae as the subfamily Lemnoideae, so that the Araceae are monophyletic . The Araceae family is divided into eight subfamilies .
The cladogram of the Araceae family with its eight subfamilies today:
| Araceae |
|
|||||||||||||||||||||||||||||||||||||||
|
|
The eight subfamilies are listed here with their branches and genera:
- Subfamily Aroideae Arn. : It contains 26 tribes with 70 genera and 2670 species:
- Tribus Aglaonemateae Engl .: It contains only two genera:
-
Aglaodorum Schott : It contains only one species:
- Aglaodorum griffithii (Schott) Schott (Syn .: Aglaonema griffithii Schott , Aglaonema palustre Teijsm. & Binn. ): It occurs in southern Vietnam , in Sumatra , on the Malay Peninsula and in Sarawak .
- Cob thread ( Aglaonema Schott ): The 21 to 23 species are distributed in tropical and subtropical Asia from northeast India to Papua New Guinea .
-
Aglaodorum Schott : It contains only one species:
- Tribe Ambrosineae Schott : It contains only one monotypical genus:
-
Ambrosina Bassi : It contains only one species:
- Ambrosina bassii L .: It iswidespreadin the Mediterranean area and occurs in Europe in Italy , Sardinia , Sicily and Corsica . In North Africa it occurs in Algeria and Tunisia.
-
Ambrosina Bassi : It contains only one species:
- Tribus Anubiadeae Engl .: It contains only one genus:
- Spear blades ( Anubias Schott ): The eight to nine species are distributed in tropical West Africa to tropical southern Africa .
- Tribus Areae R.Br. : It contains seven to eight genera:
- Arum ( Arum L. , syn .: Gymnomesium Schott ): The basic chromosome number is x = 13 or 14. (previously 15) now 29 up to 32 species of Macaronesia over the Mediterranean (center of biodiversity) and Europe to Southwest Asia and Central Asia spread to western China.
-
Biarum Schott (Syn .: Homaid Adans. Nom. Rej.): The basic chromosome number is x = 13 or 14. The 15 to 27 species are distributed in the Mediterranean area and in the Middle East as far as Iran , for example:
- Biarum davisii Turrill : It occurs in Crete.
- Narrow-leaved Biarum ( Biarum tenuifolium (L.) Schott )
-
Dracunculus Mill. (Syn .: Anarmodium Schott ): The basic chromosome number is x = 13 or 14. It contains only about two types:
- Canary Island Dragon Arum ( Dracunculus canariensis Kunth ): It occurs only in Macaronesia on Madeira and the Canaries .
- Common Dragon Arum ( Dracunculus vulgaris Schott ): It occurs only in southern Europe , Turkey and Algeria .
- Eminium (Blume) Schott (Syn .: Helicophyllum Schott ): The nine to ten species are distributed from the eastern Mediterranean to Central Asia.
-
Helicodiceros Schott (Syn .: Megotigea Raf. Nom. Rej.): It contains only one species:
- Helicodiceros muscivorus (L. f.) Engl .: It occurs only in Corsica, Sardinia and the Balearic Islands .
- Sauromatum Schott : The basic chromosome number is x = 13 or 14. The approximately twelve species are distributed in tropical Africa and tropical to subtropical Asia as far as northern China.
- Theriophonum flower (Syn .: Calyptrocoryne Schott , Pauella Ramam. & Sebastine ): The seven or so species occur in India and Sri Lanka . The basic chromosome number is x = 8.
-
Typhonium Schott (Syn .: Jaimenostia Guinea & Gómez Mor. , Lazarum A.Hay ; in some authors including the genus Sauromatum Schott ): The 50 to 60 species (excluding Sauromatum ) are originally distributed from India through Mongolia to Australia and Polynesia . Few species are neophytes in tropical Africa and the Caribbean islands. The basic chromosome number is x = 4 or 8, with 2n = 8 to 65. The extent of this genus is controversial. This is where the species belongs:
- Lizard root ( Typhonium venosum (Aiton) Hett. & PC Boyce , Syn .: Sauromatum venosum (Aiton) Kunth ).
- Tribe Arisaemateae Nakai : It contains only two genera:
- Arisaema Mart. (Syn .: Flagellarisaema Nakai , Heteroarisaema Nakai , Pleuriarum Nakai , Ringentiarum Nakai ): The 170 to 180 species occur mainly in temperate Asia, as well as in other areas of Asia, from eastern North America to Mexico , North Africa, and the Arabian peninsula. There are around 78 species in China, around 45 of them only there.
- Pine trees ( Pinellia Ten. , Syn .: Atherurus flower nom. Rej.): The nine (to eleven) species are mainly found in temperate eastern Asia (China, Japan, Korea). The center of biodiversity is in the Chinese provinces of Anhui , Fujian and Zhejiang . All nine species occur in China, seven of them only there. Two types are neophytes in parts of Australia, Europe, and North America.
- Tribe Arisareae Dumort. : It contains only one genus:
- Arisarum Mill. (Syn .: Balmisa Lag. ): The three to five species are distributed from Macaronesia across the Mediterranean to the western Transcaucasus , for example:
- Tribus Arophyteae A.Lemee ex Bogner : It contains only three genera with about eleven species that only occur in Madagascar :
- Arophyton Jum. (Syn .: Humbertina Buchet , Synandrogyne Buchet ): The seven to nine species occur in Madagascar.
- Carlephyton Jum. : The three or so species occur only in northern Madagascar.
-
Colletogyne Buchet : It contains only one species:
- Colletogyne perrieri Buchet : It occurs only in northern Madagascar.
- Tribus Caladieae Schott : It contains about seven genera mainly in the Neotropic :
- Caladium Vent. (Syn: Phyllotaenium André , Aphyllarum S.Moore ): The 12 to 16 species are common in the Neotropic.
- Chlorospatha Engl. (Syn .: Caladiopsis Engl. ): The 25 to 29 species are common in the Neotropic.
- Hapaline Schott (Syn .: Hapale Schott nom. Rej.): The six to nine species are distributed in Southeast Asia from south-central China to western Malesia .
-
Jasarum G.S.Bunting : It contains only one species:
- Jasarum steyermarkii G.S.Bunting : It occurs in southern Guyana and Venezuela only in the state of Bolívar .
-
Scaphispatha Brongn. ex Schott : It was monotypical and has contained two species in South America since 2005:
- Scaphispatha gracilis Brongn. ex Schott : It is based in Bolivia .
- Scaphispatha robusta E.G. Gonç . : It was newly described in 2005 and occurs in the Brazilian states of Pará to Goiás .
- Syngonium Schott (Syn .: Porphyrospatha Engl. ): The approximately 35 species are common in the Neotropic.
-
Xanthosoma Schott (Syn .: Acontias Schott ): The 60 to 77 species are common in the Neotropic, including:
- Tannia ( Xanthosoma sagittifolium (L.) Schott )
- Tribus Callopsideae Engl .: It contains only one genus:
-
Callopsis Engl .: It contains only one species:
- Callopsis volkensii Engl .: It occurs in tropical East Africa in southeastern Kenya and eastern Tanzania .
-
Callopsis Engl .: It contains only one species:
- Tribe Colocasieae (Schott) Brongn. : It contains about six genera:
- Arrow leaves ( Alocasia (Schott) G.Don , Syn .: Colocasia sect. Alocasia Schott , Ensolenanthe Schott , Panzhuyuia Z.Y.Zhu , Schizocasia Schott ex Engler , Xenophya Schott ): The 70 to 80 species are common in tropical Asia and Malesia .
- Ariopsis Nimmo : The only two species are common in Southeast Asia.
- Colocasia Schott (Syn .: Leucocasia Schott ): The 20 or so species are common in tropical to subtropical Asia. There are six species in China.
-
Protarum Engl .: It contains only one species:
- Protarum Sechellarum Engl .: It isnativeto the Seychelles .
- Remusatia Schott (Syn .: Gonatanthus Klotzsch ): The four or so species are distributed in southern Asia and Southeast Asia, one species also extends to Africa, Madagascar, Australia and the Pacific Islands.
- Steudnera K. Koch : The nine or so species are common in tropical and subtropical Asia.
- Tribus Cryptocoryneae flower : It contains only two genera:
- Water goblets ( Cryptocoryne Fisch. Ex Wydler , Syn .: Myrioblastus Wall. Ex Griff. ): The 50 to 64 species are distributed in tropical to subtropical Asia from India to Malesia.
-
Lagenandra Dalzell : The approximately 15 species are distributed on the Indian subcontinent in India, Bangladesh and Sri Lanka . Here is a selection:
- Lagenandra jacobsenii de Wit : It occurs in southwestern Sri Lanka.
- Lagenandra nairii Ramamurthy & Rajan : It occurs in southwestern India.
- Lagenandra ovata Thwaites : It occurs in southwest India and southwest Sri Lanka.
- Lagenandra praetermissa de Wit It occurs in southwestern Sri Lanka.
- Tribus Culcasieae Engl .: It contains only two genera:
- Cercestis Schott (Syn .: Rhektophyllum N.E.Br. , Alocasiophyllum Engl. ): The ten (to twelve) species are distributed from tropical West Africa to Uganda and Angola .
- Culcasia P.Beauv. (Syn .: Denhamia Schott ): The 24 to 28 species are common in tropical Africa.
- Tribus Dieffenbachieae Engl .: It contains only two genera:
-
Bognera Mayo & Nicolson : It contains only one species:
- Bognera recondita (Madison) Mayo & Nicolson (Syn .: Ulearum reconditum Madison ): It occurs only in the northern Brazilian state of Amazonas.
- Dieffenbachia Schott (Syn .: Maguirea A.D.Hawkes ): The 50 to 59 species are common in the Neotropics.
-
Bognera Mayo & Nicolson : It contains only one species:
- Tribus Homalomeneae M.Hotta : It contains only two genera:
- Furtadoa M.Hotta : The only two species occur in Sumatra and on the Malay Peninsula .
- Homalomena Schott (Syn .: Adelonema Schott , Chamaecladon Miq. , Diandriella Engl. ): The 110 to 132 species are widespread in the Neotropics and in tropical to subtropical Asia and on southwestern Pacific islands.
- Tribus Montrichardieae Engl .: It contains only one genus:
- Montrichardia Crueg. (Syn .: Pleurospa Raf. ): The two types are common in the Neotropic.
- Tribus Nephthytideae Engl .: It contains three genera with about 14 species mainly in tropical Africa:
- Anchomanes Schott : The six or so species are common in tropical Africa.
- Nephthytis Schott (Syn .: Oligogynium Engl. ): The six or so species are mainly distributed in tropical West Africa and one species isnativeto Borneo .
- Pseudohydrosme Engl. (Syn .: Zyganthera N.E.Br. ): There are about two species in Gabon .
- Tribus Peltandreae Engl .: It contains only two genera with only three species:
-
Peltandra Raf. : There are roughly two species in North America (eastern Canada, central and eastern USA) and in Cuba:
- Peltandra sagittifolia (Michx.) Morong
- Peltandra virginica ( L. ) Schott
-
Typhonodorum Schott (Syn .: Arodendron Werth ): It contains only one species:
- Typhonodorum lindleyanum Schott (Syn .: Typhonodorum madagascariense Engl. , Arodendron engleri Werth ): It occurs on the East African islands of Pemba and Zanzibar , in Madagascar, on the Comoros and the Mascarene Islands .
-
Peltandra Raf. : There are roughly two species in North America (eastern Canada, central and eastern USA) and in Cuba:
- Tribus Philodendreae Schott : It contains only one genus:
- Baumfreund ( Philodendron Schott , Syn .: Calostigma Schott nom. Inval., Meconostigma Schott , Sphincterostigma Schott , Arosma Raf. , Telipodus Raf. , Thaumatophyllum Schott , Elopium Schott , Baursea (Rchb.) Hoffmanns. Ex Kuntze ): The approximately 500 species are common in the Neotropic.
- Tribus Philonotieae SYWong & PCBoyce : It contains only one genus:
- Philonotion Schott : The three or so species are common in tropical South America.
- Tribus Pistieae Rich. : It contains only one monotypical genus:
-
Pistia L .: It contains only one species:
- Water lettuce ( Pistia stratiotes L. ): This water plant is widespread in the tropics and subtropics.
-
Pistia L .: It contains only one species:
- Tribe Schismatoglottideae Nakai : It contains about eleven genera with about 150 species, which mainly occur on Borneo (about 95%):
- Apoballis Schott : The twelve or so species are distributed from Indochina to western Malesia.
- Aridarum Ridl. (Syn .: Heteroaridarum M.Hotta ): The ten or so species are native to north-western Borneo.
- Bakoa P.C.Boyce & SYWong : It was set up in 2008. The only three species occur in Borneo.
- Bucephalandra Schott (Syn .: Microcasia Becc. ): There are about two species in Borneo.
-
Hestia S.Y.Wong & PCBoyce : It was set up in 2010 and contains only one species:
- Hestia longifolia (Ridl.) SYWong & PCBoyce : It is common from Perak to Borneo.
- Ooia S.Y.Wong & PCBoyce : It was set up in 2010. There are only two species in Borneo.
-
Phymatarum M.Hotta : It contains only one species:
- Phymatarum borneense M.Hotta : It is native to Borneo.
-
Pichinia S.Y.Wong & PCBoyce : It was set up in 2010 and contains only one species:
- Pichinia disticha S.Y.Wong & PCBoyce : It only occurs in Sarawak .
- Piptospatha N.E.Br. (Syn .: Gamogyne N.E.Br. , Hottarum Bogner & Nicolson , Rhynchopyle Engl. ): The approximately twelve species are distributed in Malesia from Thailand to Borneo.
- Schismatoglottis inches. & Moritzi (Syn .: Apatemone Schott ): The 111 to 120 species are distributed in tropical to subtropical Asia from Myanmar via China to Borneo and on southwestern Pacific islands ( New Guinea , Vanuatu ). The three South American species that were previously classified here have been separated into the genus Philonotion in their own tribe Philonotieaesince 2010.
-
Schottariella P.C.Boyce & SYWong : It was set up in 2009 and contains only one species:
- Schottariella mirifica P.C.Boyce & SYWong : It only occurs in Sarawak .
- Tribe Spathicarpeae Schott : It contains about nine genera in South America since 2012:
- Asterostigma fish. & CAMey. (Syn .: Andromycia A.Rich. , Staurostigma Scheidw. , Rhopalostigma Schott ): The approximately eight species are widespread from Brazil to northeastern Argentina.
-
Gearum N.E.Br. : It contains only one type:
- Gearum brasiliense N.E.Br. : The homeland are the central Brazilian provinces of Goiás , Mato Grosso and Tocantins .
- Gorgonidium Schott : The eight or so species are distributed from Peru and Bolivia to northern Argentina.
-
Lorenzia E.G. Gonç. : It was established in 2012 and contains only one type:
- Lorenzia umbrosa E.G. Gonç. : In 2012 it is only known from the type locality from the Serra do Navio , 2 km to Riozinho in the Brazilian state of Amapá . It thrives in deep shade under trees.
- Mangonia Schott (Syn .: Felipponia Hicken , Felipponiella Hicken ): The roughly two species are distributed from southern Brazil to Uruguay .
- Spathantheum Schott (Syn .: Gamochlamys Baker ): The roughly two species distributed from Peru via Bolivia to northern Argentina.
- Spathicarpa Hook. (Syn .: Aropsis Rojas ): The three or four species are common in tropical South America.
-
Synandrospadix Engl. (Syn .: Lilloa Speg. ): It contains only one species:
- Synandrospadix vermitoxicus (Griseb.) Engl. (Syn .: Asterostigma vermitoxicum Griseb. ): It is in Bolivia, Paraguay , in Peruvian Cuzco and in northern Argentina (Catamarca, Chaco, Córdoba, Corrientes, Formosa, Jujuy, Salta, Santiago del Estero , Tucumán) common.
- Taccarum Brongn. ex Schott (Syn .: Lysistigma Schott , Endera Regel ): The six or so species are distributed from tropical South America to northern Argentina.
- Tribe Stylochaetoneae Schott : It contains only one genus:
- Stylochaeton Lepr. (Syn .: Stylochiton Schott orth. Var., Gueinzia Sond. Ex Schott ): The 19 species since 2012 are distributed from tropical to southern Africa.
- Tribus Thomsonieae flower : It contains only one genus in the tropics worldwide:
- Amorphophallus flower ex Decne. (Syn .: Thomsonia Wall. Nom. Rej., Pythion Mart. Nom. Rej., Candarum Schott nom. Illeg., Pythonium Schott nom. Illeg., Kunda Raf. , Brachyspatha Schott , Conophallus Schott , Plesmonium Schott , Corynophallus Schott , Allopythion Schott , Hansalia Schott , Hydrosme Schott , Rhaphiophallus Schott , Synantherias Schott , Dunalia Montrouz. , Proteinophallus Hook.f. , Tapeinophallus Baill. , Pseudodracontium N.E.Br. ): It has a paleotropic distribution and the (150 to) about 200 species come in West to East Africa, in southern and eastern Asia, in Southeast Asia, in northern Australia and on the Pacific islands.
- Tribe Zamioculcadeae Schott : It contains only two genera:
- Gonatopus Hook. f. ex Engl. (Syn .: Heterolobium Peter , Microculcas Peter ): The five or so species are distributed from tropical East Africa to southern Africa.
-
Zamioculcas Schott : It contains only one species:
- Zamioculcas zamiifolia (Lodd. Et al.) Engl .: It is widespread from Kenya and Tanzania via Malawi, Mozambique and Zimbabwe to the South African province of KwaZulu-Natal.
- Tribus Zantedeschieae Engl .: It contains only one genus:
- Zantedeschia Spreng. (Syn .: Pseudohomalomena A.D. Hawkes , Richardia Kunth ): The eightor sospecies are mainly found in southern Africa, with only one species occurring in Nigeria and Tanzania.
- Tribe Zomicarpeae Schott : It contains four genera with only about six species in South America:
-
Filarum Nicolson : it contains only one species:
- Filarum manserichense Nicolson : It occurs in Peru only in Loreto.
-
Ulearum Engl .: It contains only one species:
- Ulearum sagittatum Engl .: It occurs in western South America in Brazil only in the state of Acre and in Peru only in Loreto.
- Zomicarpa Schott : The only two previously three species since 2012 are common in northeastern Brazil.
- Zomicarpella N.E.Br. : The only two types are common in Colombia, Peru and Brazil.
-
Filarum Nicolson : it contains only one species:
- Tribus Aglaonemateae Engl .: It contains only two genera:
- Subfamily Pothoideae Engl .: It contains two tribes with four genera and about 1000 species:
- Tribus Anthurieae (Schott) Engl .: It contains only one genus:
- Flamingo flowers ( Anthurium Schott , Syn .: Podospadix Raf. , Strepsanthera Raf. ): It probably contains around 1000 species in the Neotropic.
- Tribus Potheae Bartl. : It contains three genera with almost 80 species in the Paleotropic :
-
Pedicellarum M.Hotta : It contains only one species:
- Pedicellarum paiei M.Hotta : It is native to Borneo.
-
Pothoidium Schott: It contains only one species:
- Pothoidium lobbianum Schott : It occurs in southern Taiwan , Celebes , Maluku and Sulawesi and in the Philippines .
- Pothos L. (Syn .: Tapanava Adans. , Batis Blanco , Goniurus C.Presl ): The approximately 75 species are widespread in tropical to subtropical Asia, Madagascar, Australia and Polynesia.
-
Pedicellarum M.Hotta : It contains only one species:
- Tribus Anthurieae (Schott) Engl .: It contains only one genus:
- Subfamily Monsteroideae (Schott) Engl .: It contains four tribes with about twelve genera and about 360 species:
- Tribus Anadendreae Bogner & J.French : It contains only one genus:
- Anadendrum Schott (Syn .: Nothopothos Kuntze ): The nine or so species are distributed in tropical Asia from India to Malaysia.
- Tribus Heteropsideae Engl .: It contains only one genus:
- Heteropsis Kunth : The approximately 13 species distributed in tropical South America.
- Tribus Monstereae (Schott) Engl .: It contains about eight genera:
- Alloschemone Schott : The only two types occur in Brazil.
- Amydrium Schott (Syn .: Epipremnopsis Engler ): The five or so species are common in tropical Asia.
- Efeututen ( Epipremnum Schott ): The 20 or so species are common in tropical Asia.
- Window leaves ( Monstera Adans. ): The 37 or so species are common in the Neotropics.
- Rhaphidophora Hassk. (Syn .: Afrorhaphidophora Engl. ): The approximately 120 species are distributed from tropical Africa to tropical Asia and Australia to islands in the west of the Pacific.
- Rhodospatha Poepp. (Syn .: Anepsias Schott ): The approximately 27 species are common in the Neotropic.
- Scindapsus Schott (Syn .: Cuscuaria Schott ): The approximately 36 species are distributed from tropical to subtropical Asia through northern Australia to islands in the west of the Pacific.
- Stenospermation Schott : The 48 or so species are common in the Neotropics.
- Tribe Spathiphylleae Schott : It contains only two genera:
-
Holochlamys Engl .: It contains only one species:
- Holochlamys beccarii (Engl.) Engl. (Syn .: Holochlamys guineesis Engl. & K.Krause , Holochlamys ornata Alderw. , Spathiphyllum beccarii Engl. ): It occurs in Irian Jaya and Papua New Guinea .
- Spathiphyllum Schott (Syn .: Hydnostachyon Liebm. , Massowia K.Koch , Spathiphyllopsis Teijsm. & Binn. , Amomophyllum Engl. , Leucochlamys Poepp. Ex Engl. ): The approximately 50 species are common in the Neotropic and come in Malesia and the Solomon Islands before.
-
Holochlamys Engl .: It contains only one species:
- Tribus Anadendreae Bogner & J.French : It contains only one genus:
- Subfamily Calloideae Endl. : It contains only one tribe:
- Tribe Calleae Bartl. : It contains only one monotypical genus:
-
Calla L .: It contains only one species:
- Swamp calla or dragon arum ( Calla palustris L. ): It occurs in the temperate zones of the northern hemisphere .
-
Calla L .: It contains only one species:
- Tribe Calleae Bartl. : It contains only one monotypical genus:
- Subfamily Gymnostachydoideae Bogner & Nicolson :
- It contains only one monotypical genus:
-
Gymnostachys R.Br. : It contains only one type:
- Gymnostachys anceps R.Br. : It is common in eastern Australia in New South Wales and Queensland .
-
Gymnostachys R.Br. : It contains only one type:
- It contains only one monotypical genus:
- Subfamily Orontioideae Mayo, Bogner, & Boyce : It contains only one tribe:
- Tribe Orontieae R.Br. ex Dumort. : It contains only three genera with only eight species in Asia and North America:
- Scheinkalla ( Lysichiton Schott ): Of the only two species, one occurs in East Asia only in Kamchatka , Khabarovsk , on the Kuriles , Magadan , Sakhalin and on the Japanese islands of Hokkaidō and Honshu and one in North America from Alaska to the western USA.
-
Orontium L .: It contains only species:
- Orontium aquaticum L .: It is common in the eastern United States and Texas. In southern Sweden she is a neophyte.
-
Symplocarpus Salisb. ex WPCBarton : Of the five or so species, one is widespread in eastern North America, two are widespread in East Asia, one species is endemic to the Primorye region and one species is local to the central Japanese mountain Nabekura . The American way is:
- Symplocarpus foetidus Salisb.
- Tribe Orontieae R.Br. ex Dumort. : It contains only three genera with only eight species in Asia and North America:
- Subfamily Lasioideae Engl .: It contains only one tribe:
- Tribus Lasieae Engl .: It contains about ten genera with about 58 species:
- Anaphyllopsis A.Hay : The only three species are widespread in South America from Venezuela via Guyana to northern Brazil.
- Anaphyllum Schott : The only two species in occur in southern India, for example on the Laccadive Islands and Tamil Nadu.
-
Cyrtosperma handle. (Syn .: Arisacontis Schott ): The approximately twelve species are distributed from Malesia to Polynesia , including:
- Cyrtosperma merkusii (Hassk.) Schott : It occurs from Melanesia to Polynesia, especially on Tuvalu .
-
Dracontioides Engl .: It contains only one species:
- Dracontioides desciscens (Schott) Engl .: It is common in Brazil.
- Dracontium L. (Syn .: Eutereia Raf. , Echidnium Schott , Ophione Schott , Chersydrium Schott , Godwinia Seem. ): The 24 to 29 species are common in the Neotropic.
- Lasia Lour. (Syn .: Lasius Hassk. ): Of the only two species, one occurs only in Kalimantan and the other is common in tropical to subtropical Asia.
-
Lasimorpha Schott : It contains only one species:
- Lasimorpha senegalensis Schott : It is widespread in tropical Africa.
-
Podolasia N.E.Br. : It contains only one type:
- Podolasia stipitata N.E.Br. : It is common in western Malesia in Sumatra and Malaysia.
- Pycnospatha Thorel ex Gagnep. : The only two types are common in Southeast Asia in Thailand, Laos and Vietnam.
- Urospatha Schott (Syn .: Urophyllum K. Koch nom. Illeg., Urospathella G.S.Bunting ): The approximately eleven species are common in the Neotropic.
- Tribus Lasieae Engl .: It contains about ten genera with about 58 species:
- Subfamily Lemna (Lemnoideae Bab. ): It contains only one tribe:
- Tribe Lemneae Rich. ex A. Rich. : It contains five genera with about 37 species:
- Duckweed ( Lemna L. , Syn .: Lenticularia Ség. , Lenticula P.Micheli ex Adans. , Hydrophace Hallier , Telmatophace Schleid. , Staurogeton Rchb. , Thelmatophace Godr. , Lenticularia P.Micheli ex Montandon ): The approximately 13 species are almost spread worldwide.
-
Pond lentils ( Spirodela Schleid. ): It contains only two types:
- Spirodela intermedia (W. Koch : It occurs from Costa Rica to Brazil and Chile.
- Multi-rooted pond lens ( Spirodela polyrhiza (L.) Schleiden ): It occurs worldwide.
-
Landoltia Les & DJCrawford ). It contains only one species ( placed under Spirodela until 1999 ):
- Landoltia punctata (G. Meyer) Les & Crawford
- Dwarf duckweed ( Wolffia Horkel ex Schleid. , Syn .: Horkelia Rchb. Ex Bartl. , Grantia Griff. Ex Voigt , Bruniera Franch. ): The eleven species are distributed almost worldwide.
- Wolffiella (Hegelm.) Hegelm. (Syn .: Pseudowolffia Hartog & Plas , Wolffiopsis Hartog & Plas ): The ten or so species are common in the New World and Africa.
- Tribe Lemneae Rich. ex A. Rich. : It contains five genera with about 37 species:
use
Popular houseplants with decorative leaves or inflorescences come from the genera window leaves ( Monstera ), Baumfreund ( Philodendron ), Dieffenbachia ( Dieffenbachia ), flamingo flowers ( Anthurium ), Kalla ( Zantedeschia ), Syngonium and single leaf ( Spathiphyllum ). Varieties of a number of species of the arum family play a major role in floristry , for example from the genera flamingo flowers ( Anthurium ) and kalla ( Zantedeschia ). Some species of the arum family are also used in the aquarium hobby, for example the dwarf pear leaf ( Anubias barteri var. Nana ) and a number of water goblets ( Cryptocoryne ).
An important food crop is the taro ( Colocasia esculenta ), whose starchy bulbs are used.
history
Arum maculatum and Dracunculus vulgaris
swell
- Antiquity - late antiquity: Dioscorides 1st century --- Pliny 1st century --- Galen 2nd century --- Pseudo-Apuleius 4th century --- Pseudo-Dioscorides de herbis femininis. 6th century
- Arab Middle Ages: Avicenna 11th century --- Constantine 11th century --- Circa instans 12th century --- Pseudo-Serapion 13th century --- Ibn al-Baitar 13th century
- Latin Middle Ages: Pseudo-Macer 11th century --- German Macer 13th century --- Spuria Macri --- Hildegard von Bingen 12th century --- Herbarius Moguntinus 1484 --- Gart der Gesundheit 1485 --- Hortus sanitatis 1491 --- Hieronymus Brunschwig 1500
- Modern times: Otto Brunfels 1532 --- Otto Brunfels 1537 --- Hieronymus Bock 1539 --- Leonhart Fuchs 1543 --- Mattioli - Handsch - Camerarius 1586 --- Nicolas Lémery 1699/1721 --- Onomatologia medica completa 1755 --- William Cullen 1789/90 --- Hecker 1814/15 --- Pereira - Buchheim 1848
Historical illustrations
… Viennese Dioscurides … 6th century
Pseudo-Apuleius Leiden, 6th century
… .. Vitus outlet ..… 1479
Herbarius Moguntinus . Above: Issue Mainz 1484. Below: Issues Vicenza 1491 and Venice 1499
... garden of health ... 1485
… .. Hortus sanitatis ..… 1491
… .. Otto Brunfels ..… 1532
… .. Otto Brunfels …… 1537
Leonhart Fuchs 1543. Left: Aron. Right: snake weed
Hieronymus Bock 1546. Left: Aron. Right: Drachenwurtz
Mattioli , Handsch, Camerarius 1586
swell
- The Araceae family on the AP website. (Sections Description and Systematics)
- CATE Araceae - Information about the eMonocot Team Araceae family . (Sections Systematics and Distribution)
- Anna Haigh: Datasheet Neotropical Araceae In: W. Milliken, B. Klitgård, A. Baracat: Neotropikey - Interactive key and information resources for flowering plants of the Neotropics (from 2009). (Sections Description and Systematics)
- Heng Li, Guanghua Zhu, Peter C. Boyce, Jin Murata, Wilbert LA Hetterscheid, Josef Bogner, Niels Jacobsen: Araceae. In: Wu Zheng-yi, Peter H. Raven, Deyuan Hong (Eds.): Flora of China . Volume 23: Acoraceae through Cyperaceae . Science Press / Missouri Botanical Garden Press, Beijing / St. Louis 2010, ISBN 978-1-930723-99-3 , pp. 3 (English, online - PDF file ). (in the old scope without Lemnaceae; sections description, distribution and systematics).
- Heng Li, Elias Landolt: Lemnaceae. In: Wu Zheng-yi, Peter H. Raven, Deyuan Hong (Eds.): Flora of China . Volume 23: Acoraceae through Cyperaceae . Science Press / Missouri Botanical Garden Press, Beijing / St. Louis 2010, ISBN 978-1-930723-99-3 , pp. 80 (English, online - PDF file ). (still as a separate family; sections description, distribution and systematics).
- L. Nauheimer, D. Metzler, SS Renner: Global history of the ancient monocot family Araceae inferred with models accounting for past continental positions and previous ranges based on fossils. In: New Phytologist. Volume 195, No. 4, 2012, pp. 938-950, doi: 10.1111 / j.1469-8137.2012.04220.x .
- Simon J. Mayo, Josef Bogner, Nathalie Cusimano: Recent Progress in the Phylogenetics and Classification of the Araceae. In: Paul Wilkin, Simon J. Mayo: Early Events in Monocot Evolution. Systematics Association Special Volume Series. Cambridge University Press, 2013, ISBN 978-1-107-01276-9 , pp. 208-242.
- Website of the International Aroid Society. (Sections Description and Systematics).
- The Araceae family without Lemnaceae and the Lemnaceae family at DELTA by L. Watson & MJ Dallwitz. (Sections Description and Systematics).
- PC Boyce, TB Croat: The Überlist of Araceae - Totals for Published and Estimated Number of Species in Aroid Genera - PDF file of January 10, 2012.
Individual evidence
- ↑ a b c d e f g h i j k l m n o p q r s t u Rafaël Govaerts (Ed.): Araceae. In: World Checklist of Selected Plant Families (WCSP) - The Board of Trustees of the Royal Botanic Gardens, Kew . Retrieved April 13, 2020.
- ↑ a b PC Boyce, TB Croat: The Überlist of Araceae - Totals for Published and Estimated Number of Species in Aroid Genera - PDF file of January 10, 2012.
- ↑ a b c d e Anna Haigh, 2009: Datasheet Neotropical Araceae. In: W. Milliken, B. Klitgård, A. Baracat: Neotropikey - Interactive key and information resources for flowering plants of the Neotropics (from 2009).
- ^ Heinrich Wilhelm Schott: Genera Aroidearum exposita. Caroli Ueberreuter, Vienna 1858 (online).
- ^ Heinrich Wilhelm Schott: Prodromus systematis Aroidearum. Typis Congregationis Mechitharisticae, Vienna 1860, p. 214 (online).
- ↑ Michael H. Grayum: Evolution and Phylogeny of the Araceae . In: Annals of the Missouri Botanical Garden . tape 77 , no. 4 , 1990, ISSN 0026-6493 , pp. 628-697 , doi : 10.2307 / 2399668 , JSTOR : 2399668 (English).
- ↑ a b c d e f g h i j k l Araceae in the Germplasm Resources Information Network (GRIN), USDA , ARS , National Genetic Resources Program. National Germplasm Resources Laboratory, Beltsville, Maryland. Retrieved June 21, 2014.
- ↑ The Araceae family on the AP website .
- ↑ a b Angiosperm Phylogeny Group III: An update of the Angiosperm Phylogeny Group classification for the orders and families of flowering plants: APG III . In: Botanical Journal of the Linnean Society . tape 161 , no. 2 , October 2009, ISSN 0024-4074 , p. 105–121 , doi : 10.1111 / j.1095-8339.2009.00996.x (English).
- ^ L. Nauheimer, D. Metzler, Susanne S. Renner : Global history of the ancient monocot family Araceae inferred with models accounting for past continental positions and previous ranges based on fossils. In: New Phytologist. Volume 195, No. 4, 2012, pp. 938-950, doi: 10.1111 / j.1469-8137.2012.04220.x .
- ↑ a b c d e f g h Walter Erhardt , Erich Götz, Nils Bödeker, Siegmund Seybold: The great pikeperch. Encyclopedia of Plant Names. Volume 2. Types and varieties. Eugen Ulmer, Stuttgart (Hohenheim) 2008, ISBN 978-3-8001-5406-7 .
- Jump up ↑ Jeanine Linz, Johannes Stökl, Isabella Urru, Tamara Krügel, Marcus C. Stensmyr, Bill S. Hansson: Molecular phylogeny of the genus Arum (Araceae) inferred from multi-locus sequence data and AFLPs. In: Taxon. Volume 59, No. 2, 2010, pp. 405-415, abstract, ( Memento from September 23, 2016 in the Internet Archive ) JSTOR 25677599 .
- ↑ a b Natalie Cusimano, M. Barrett, Wilbert LA Hetterscheid, Susanne S. Renner : A phylogeny of the Areae (Araceae) implies that Typhonium, Sauromatum, and the Australian species of Typhonium are distinct clades. In: Taxon. Volume 59, 2010, pp. 439-447, abstract, ( Memento from November 1, 2017 in the Internet Archive ) JSTOR 25677602 .
- ↑ Aretuza Sousa, Natalie Cusimano, Susanne S. Renner: Combining FISH and model-based predictions to understand chromosome evolution in Typhonium (Araceae). In: Annals of Botany. Volume 113, No. 4, 2014, pp. 669-680, doi: 10.1093 / aob / mct302 .
- ↑ Christel Kasselmann : aquarium plants. Ulmer Verlag, Stuttgart 1995; 2nd, revised and expanded edition 1999, ISBN 3-8001-7454-5 , pp. 322–326.
- ↑ a b c Sin Yeng Wong, Peter C. Boyce, Ahmad Sofiman bin Othman, Leaw Chui Pin: Molecular phylogeny of tribe Schismatoglottideae (Araceae) based on two plastid markers and recognition of a new tribe, Philonotieae, from the neotropics. In: Taxon. Volume 59, No. 1, 2010, pp. 117–124, abstract, ( page no longer available , search in web archives ) Info: The link was automatically marked as defective. Please check the link according to the instructions and then remove this notice. JSTOR 27757056 .
- ↑ a b c d Lidia I. Cabrera, Gerardo R. Salazar, Mark W. Chase, Simon J. Mayo, Josef Bogner, Patricia Dávila: Phylogenetic relationships of aroids and duck-weeds (Araceae) inferred from coding and noncoding plastid DNA. In: American Journal of Botany. Volume 95, No. 9, 2008, pp. 1153-1165, doi: 10.3732 / ajb.0800073
- ↑ Sin Yeng Wong: Rheophytism in Bornean Schismatoglottideae (Araceae). In: Systematic Botany. Volume 38, No. 1, 2013, pp. 32-45, doi: 10.1600 / 036364413X661908 .
- ↑ Eduardo G. Gonçalves, SJ Mayo, MA Van-Sluy, A. Salatino: Combined genotypic-phenotypic phylogeny of the tribe Spathicarpeae (Araceae) with reference to independent events of invasion to Andean regions. In: Molecular Phylogenetics and Evolution. Volume 43, No. 3, 2007, pp. 1023-1039, doi: 10.1016 / j.ympev.2007.01.008 .
- ↑ Eduardo G. Gonçalves,: Lorenzia (Araceae - Spathicarpeae): A New Genus from Northern Brazil Supported by matK Sequence Data . In: Systematic Botany . tape 37 , no. 1 , March 1, 2012, ISSN 0363-6445 , p. 48-52 , doi : 10.1600 / 036364412X616620 ( online ).
- ↑ a b Heng Li, Guanghua Zhu, Peter C. Boyce, Jin Murata, Wilbert LA Hetterscheid, Josef Bogner, Niels Jacobsen: Araceae. In: Wu Zheng-yi, Peter H. Raven, Deyuan Hong (Eds.): Flora of China . Volume 23: Acoraceae through Cyperaceae . Science Press / Missouri Botanical Garden Press, Beijing / St. Louis 2010, ISBN 978-1-930723-99-3 , pp. 3 (English, online - PDF file ).
- ↑ Peter Boyce, Duangchai Sookchaloem, Wilbert LA Hetterscheid, Guy Gusman, Nirls Jacobsen, Takashike Idei, Nguyen van Du: Flora of Thailand. Volume 11, Part 2, Araceae, Acoraceae , 2012, ISBN 978-974-286-980-9 .
- ^ Eduardo G. Gonçalves: A revision of the small genus Zomicarpa Schott. In: Kew Bulletin. Volume 67, No. 3, 2012, pp. 443-449, doi: 10.1007 / s12225-012-9336-x .
- ^ Josef Bogner: Are the flowers of the duckweeds (Araceae-Lemnoideae) bisexual or unisexual? In: Aroideana. Volume 33, 2010, pp. 178-182.
- ↑ Pedanios Dioscurides . 1st century De Medicinali Materia libri quinque. Translation. Julius Berendes . Pedanius Dioscurides' medicine theory in 5 books. Enke, Stuttgart 1902, p. 245 (Book II, Chapter 197): Aron (digitized version)
- ↑ Pliny the Elder , 1st century. Naturalis historia Book XXIV, Chapter 91 (§ 142–148): Dracontium (digitized version ) ; Translation Külb 1855 (digitized version )
- ↑ Galen , 2nd century De alimentorum facultatibus , Book II, Chapter 63 (based on the Kühn edition, Volume VI, p. 649): Arum (digitized version), Chapter 64 (based on the Kühn edition, Volume VI, p. 651): Dracontium ( Digitized) . De simplicium medicamentorum temperamentis ac facultatibus , Book VI, Chapter I / 61 (after Kühn 1826, Vol. XI, p. 839): Aron (digitalisat) Chapter VI / 9 (after Kühn 1826, Vol. XI, p. 864): Dracontium (digitized version )
- ↑ First printing: Rome 1481, Chapter 15: Dracontea (digitized version )
- ↑ Pseudo-Dioscorides de herbis femininis. 6th century edition: HF Kästner. Pseudo-Dioscorides de herbis femininis. In: Hermes , Vol. 31 (1896), Chapter 44 (pp. 619–620): Dracontea femina (digitized version )
- ↑ Avicenna , 11th century, Canon of Medicine . Translation and adaptation by Gerhard von Cremona , Arnaldus de Villanova and Andrea Alpago (1450–1521). Basel 1556, Volume II, Chapter CCCCXL: Luf - Dracunculus et Aro (digital copy )
- ↑ Constantine the African , 11th century Liber de gradibus simplicium . Pressure. Opera . Basel 1536, p. 350: Aros (digitized version)
- ↑ Circa instans 12th century print. Venice 1497, sheet 200r: Jarus (digitized) ; Sheet 210r: Serpentaria (digitized version )
- ^ Pseudo-Serapion 13th century, print. Venice 1497, sheet 105v (No 43): Luf (digitized)
- ↑ Abu Muhammad ibn al-Baitar , 13th century, Kitāb al-jāmiʿ li-mufradāt al-adwiya wa al-aghdhiya. Translation. Joseph Sontheimer under the title Large compilation on the powers of the well-known simple healing and food. Hallberger, Stuttgart Volume II 1842, p. 446: Luf (digitized)
- ^ Pseudo-Macer Cap. 54: Columbrina . Printed in Basel 1527, sheet 36v: Columbrina (digitized version )
- ^ German Macer . After: Bernhard Schnell, William Crossgrove: The German Macer. Vulgate version. Niemeyer, Tübingen 2003, p. 361 (Cap. 51). Heidelberg, Cpg 226 , Alsace, 1459-1469, sheet 197v: Columbaria (digitized)
- ↑ Ulrike Jansen. Spuria Macri. An appendix to Macer Floridus, De viribus herbarum . (Contributions to antiquity. Volume 314) De Gruyter, Berlin / Boston 2013. Aaron . Text and translation: pp. 189–192; Commentary: pp. 271-275
- ^ Charles Victor Daremberg and Friedrich Anton Reuss (1810-1868). S. Hildegardis Abbatissae Subtilitatum Diversarum Naturarum Creaturarum Libri Novem. Physica , Book I, Chapter 49. Migne, Paris 1855. Sp. 1148 (digitized version ) - Translation: Herbert Reier: Hildegard von Bingen Physica. Translated into German after the text edition by JP Migne, Paris 1882. Kiel 1980, p. 3. The herb Aaron is neither mild nor very strong, but has a balanced temperature, just like the sun has a pleasant warmth after the dawn, just as the dew in the summer before days is pleasant and so everything can be changed can, like a pleasant man who can endure danger and who is lucky enough. If in a person his illness manifests itself in black pustules, in which the death of the person lies, which is called seltega, and if the leaves or roots of this herb are given to him to eat, then they alleviate the excessive heat or cold is in the pustules so that they subside slightly. And when a person is so condemned that all limbs fall from him and he loses his speech, you will soon give him leaves of the herb Aaron to eat with a little salt, and the gout will subside. If it does not yet do so, dip its roots in boiled honey and give it to him to eat immediately so that he can improve. But if a person has slimet fiber in his stomach, from which riddo grows, he boil the root of the herb Aaron in pure wine and let it cool down, then put a red-hot iron in this wine and finally heat it up so that he drinks it hot, and the slim in his belly takes the fiber from him, As the fire melts the snow. A person in whom melancholy rages, who has a rough mind and is always sad, should often drink wine cooked with aaron root; this diminishes in him the melancholy and the overly.
- ↑ Herbarius Moguntinus , Mainz 1484, part, chapter 17: Aaron, Aron (digitized version) ; Chapter 127: Serpentaria, Naterwortz (digitized version )
- ↑ Gart der Gesundheit . Mainz 1485, Chapter 16: (digitized version)
- ↑ Hortus sanitatis 1491, Mainz 1491, Part I, Chapter 1: (digitized version)
- ↑ Hieronymus Brunschwig . Small distilling book , Strasbourg 1500, sheet 19v: Aron (digitized)
- ↑ Otto Brunfels . Contrafayt Kreüterbůch . Johann Schott, Strasbourg 1532, p. 67 (digitized version)
- ↑ Otto Brunfels . Another part of the Teütschen Contrafayten Kreüterbůchs . Johann Schott, Strasbourg 1537, p. 134: Illustration of snake herb (digitized)
- ↑ Hieronymus Bock . New Kreütter Bůch . Wendel Rihel, Strasbourg 1539, Part II, Chapter 78: Aron (digitized version) , Chapter 79: Drachenwurtz (digitized version)
- ↑ Leonhart Fuchs . New Kreütterbuch… Michael Isingrin, Basel 1543, Chapter 22: Aron (digitized) . Chapter 85: Large Snake Weed (digitized version)
- ^ Pietro Andrea Mattioli . Commentarii, in libros sex Pedacii Dioscoridis Anazarbei, de medica materia. Translation by Georg Handsch, edited by Joachim Camerarius the Younger , Johan Feyerabend, Franckfurt am Mayn 1586, sheet 189v – 190v: Drachenwurtz, Schlangenkraut (digitized version ) . Sheet 191v – 193r: Aron (digitized version )
- ↑ Nicolas Lémery . Dictionnaire universel des drogues simples. , Paris 1699, pp. 66-67: Arum (digitalisat) ; Translation. Complete material lexicon. Initially drafted in French, but now after the third edition, which has been enlarged by a large [...] edition, translated into high German / By Christoph Friedrich Richtern, [...]. Leipzig: Johann Friedrich Braun, 1721, Sp. 108-109: Arum (digitized version)
- ^ Albrecht von Haller (editor). Onomatologia medica completa or Medicinisches Lexicon which explains all names and artificial words which are peculiar to the science of medicine and pharmacists art clearly and completely [...]. Gaumische Handlung, Ulm / Frankfurt am Main / Leipzig 1755, Sp. 144–145 (digitized version )
- ^ William Cullen, A treatise of the materia medica. Charles Elliot, Edinburgh 1789. Volume II, pp. 211-214: Acria (digitized) . German. Samuel Hahnemann . Schwickert, Leipzig 1790. Volume II, pp. 241-244 Sharp substances . (Digitized version) . Volume II, p. 554: Diuretics (digital copy ) . German. Samuel Hahnemann . Schwickert, Leipzig 1790. Volume II, p. 613: As a diuretic (digitalized)
- ↑ August Friedrich Hecker's practical medicine theory. Revised and enriched with the latest discoveries by a practicing doctor. Camesius, Vienna, Volume I 1814, p. 648 (digitized version )
- ↑ Jonathan Pereira’s Handbook of Medicines Doctrine. From the point of view of the German Medicin edited by Rudolf Buchheim . Leopold Voß, Leipzig, Volume II 1848, pp. 60–61 (digitized version )
Web links
further reading
- Takanori Ito, Kikukatsu Ito: Nonlinear dynamics of homeothermic temperature control in skunk cabbage, Symplocarpus foetidus. In: Physical Review E. Volume 72, No. 5, 2005, doi: 10.1103 / PhysRevE.72.051909 .
- Richard C. Keating: Vegetative anatomical data and its relationship to a revised classification of the genera of Araceae. In: Annals of the Missouri Botanical Garden. Volume 91, No. 3, 2004, pp. 485-494, JSTOR 3298625 .